Metal Questions
... Which statement best describes the attraction present in metallic bonding? A. the attraction between nuclei and electrons B. the attraction between positive ions and electrons C. the attraction between positive ions and negative ions D. the attraction between protons and electrons Which is the best ...
... Which statement best describes the attraction present in metallic bonding? A. the attraction between nuclei and electrons B. the attraction between positive ions and electrons C. the attraction between positive ions and negative ions D. the attraction between protons and electrons Which is the best ...
Chemistry Claims Unit 1: Alchemy: Matter, Atomic Structure, and
... Hydrogen/Carbon bonds are hard/easy bonds to break. Steam engines/Internal combustion engines are the best way to convert chemical energy to work. [Example element] is more/less active than [example element]. Pairing Tin and Gold/Iron and Lead will produce the most/least amount of energy. ...
... Hydrogen/Carbon bonds are hard/easy bonds to break. Steam engines/Internal combustion engines are the best way to convert chemical energy to work. [Example element] is more/less active than [example element]. Pairing Tin and Gold/Iron and Lead will produce the most/least amount of energy. ...
Membrane Permeability A. Permeability If you take a pure solution of
... So how do ions and other substance pass through the membrane? Many transmembrane proteins exist, that form an aqueous channel in the lipid bilayer. How does a protein cross the membrane, if membranes are nonpolar and peptides are polar? It turns out that almost all protein stretches that cross the m ...
... So how do ions and other substance pass through the membrane? Many transmembrane proteins exist, that form an aqueous channel in the lipid bilayer. How does a protein cross the membrane, if membranes are nonpolar and peptides are polar? It turns out that almost all protein stretches that cross the m ...
Answers - Scioly.org
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
AP Reactions - Georgetown ISD
... rest is reduced. The same chemical substance undergoes both oxidation and reduction. NO2 and H2O2 are classic chemicals that have this ability. Example: 3NO2(g) + H2O ...
... rest is reduced. The same chemical substance undergoes both oxidation and reduction. NO2 and H2O2 are classic chemicals that have this ability. Example: 3NO2(g) + H2O ...
Activity Name: Polyatomic Ion Bingo
... has a charge because of the loss or gain of electrons. A polyatomic ion is an ion that consists of at least two different elements. Polyatomic ions are common ingredients in many foods and household products. Phosphate, nitrate, sulfate, and sulfite are just a few examples. Memorizing the chemical f ...
... has a charge because of the loss or gain of electrons. A polyatomic ion is an ion that consists of at least two different elements. Polyatomic ions are common ingredients in many foods and household products. Phosphate, nitrate, sulfate, and sulfite are just a few examples. Memorizing the chemical f ...
Chemistry Nomenclature Notes
... making an ionic compound. All rules for making formulas are followed. Notice that most of the polyatomic ions are negatively charged except for ammonium. Also notice that the names for most of the complex ions end in ‘ate’. This is your clue that the compound consists of a polyatomic ion. ...
... making an ionic compound. All rules for making formulas are followed. Notice that most of the polyatomic ions are negatively charged except for ammonium. Also notice that the names for most of the complex ions end in ‘ate’. This is your clue that the compound consists of a polyatomic ion. ...
2. Chemistry of Living Things Outline
... place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present for the reaction to occur. Some enzymes have a __________________ part called a _______________ ...
... place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present for the reaction to occur. Some enzymes have a __________________ part called a _______________ ...
Chemistry of Living Things Outline
... An ________ is an atom that has ________ or _______ an ____________. Ions are formed during chemical bonding that involves the ________ of electrons. When electrons are transferred from one atom to another, _______ atoms become electrically ____________. The atom that __________ electrons be ...
... An ________ is an atom that has ________ or _______ an ____________. Ions are formed during chemical bonding that involves the ________ of electrons. When electrons are transferred from one atom to another, _______ atoms become electrically ____________. The atom that __________ electrons be ...
Electrode Potentials hw - A
... Use data from the table of standard electrode potentials to deduce the cell which would have a standard e.m.f. of 0.93 V. Represent this cell using the convention shown in part (c). ...
... Use data from the table of standard electrode potentials to deduce the cell which would have a standard e.m.f. of 0.93 V. Represent this cell using the convention shown in part (c). ...
Review Unit - hrsbstaff.ednet.ns.ca
... Remember that the unit for energy is the Joule (J). b) Physical State - Chemical reactions often depend on the physical state of the chemicals involved. This information can be included in an equation by using these symbols: (s) = solid (l) = liquid (g) = gas (a) = aqueous (dissolved in water) For e ...
... Remember that the unit for energy is the Joule (J). b) Physical State - Chemical reactions often depend on the physical state of the chemicals involved. This information can be included in an equation by using these symbols: (s) = solid (l) = liquid (g) = gas (a) = aqueous (dissolved in water) For e ...
Chemical Reaction and Matter Review
... ** When acids and bases are added to each other they react to neutralize each other if an equal number of hydrogen and hydroxide ions are present. When this reaction occurs -salt and water are formed. HCl + NaOH NaCl + H2O (Acid) (Base)---(Salt) (Water) What are some useful applications of this reac ...
... ** When acids and bases are added to each other they react to neutralize each other if an equal number of hydrogen and hydroxide ions are present. When this reaction occurs -salt and water are formed. HCl + NaOH NaCl + H2O (Acid) (Base)---(Salt) (Water) What are some useful applications of this reac ...
2C - Edexcel
... (c) The student carried out the experiment three times. The table shows his results. Temperature in °C ...
... (c) The student carried out the experiment three times. The table shows his results. Temperature in °C ...
AP Chemistry Review Packet 1 CO2(g) + H2(g) « H2O(g) + CO(g
... (c) Without performing calculations, describe how the resulting data should be used to obtain the standard molar enthalpy of neutralization. (d) When a class of students performed this experiment, the average of the results was -55.0 kilojoules per mole. The accepted value for the standard molar ent ...
... (c) Without performing calculations, describe how the resulting data should be used to obtain the standard molar enthalpy of neutralization. (d) When a class of students performed this experiment, the average of the results was -55.0 kilojoules per mole. The accepted value for the standard molar ent ...
solutions quiz
... A student has two identical beakers with equal volumes of water. The student adds three grams of sugar to each beaker. The student places beaker B on a hot plate and heats the solution while stirring. Solution A remains at room temperature and is not stirred. The student records the time it takes fo ...
... A student has two identical beakers with equal volumes of water. The student adds three grams of sugar to each beaker. The student places beaker B on a hot plate and heats the solution while stirring. Solution A remains at room temperature and is not stirred. The student records the time it takes fo ...
+ H 2
... Writing REDOX Reactions Write the net ionic equation for the reaction of solid zinc in a solution of hydrochloric acid. ...
... Writing REDOX Reactions Write the net ionic equation for the reaction of solid zinc in a solution of hydrochloric acid. ...
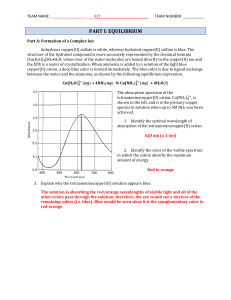
Final competitions (29.03.2008) Competing equilibria Complex
... percentage of methanol decomposed to hydrogen when the reaction reaches equilibrium. a4. Reaction of methanol and water is endothermic. Calculate the percentage of methanol to be oxidized to water for the thermal effect of the reaction to be zero, knowing that when 1.00 mole of oxygen reacts with 2. ...
... percentage of methanol decomposed to hydrogen when the reaction reaches equilibrium. a4. Reaction of methanol and water is endothermic. Calculate the percentage of methanol to be oxidized to water for the thermal effect of the reaction to be zero, knowing that when 1.00 mole of oxygen reacts with 2. ...
buffers - sbhschemistry
... Definition: A buffer solution is one in which there is very little change in its pH when a relatively large amount of strong acid or strong bases is added to it. How to make a buffer A buffer contains a mixture of a weak acid and its conjugate base OR a weak base and its conjugate acid. e.g. (1) wea ...
... Definition: A buffer solution is one in which there is very little change in its pH when a relatively large amount of strong acid or strong bases is added to it. How to make a buffer A buffer contains a mixture of a weak acid and its conjugate base OR a weak base and its conjugate acid. e.g. (1) wea ...
PH

In chemistry, pH (/piːˈeɪtʃ/) is a numeric scale used to specify the acidity or alkalinity of an aqueous solution. It is the negative of the logarithm to base 10 of the activity of the hydrogen ion. Solutions with a pH less than 7 are acidic and solutions with a pH greater than 7 are alkaline or basic. Pure water is neutral, being neither an acid nor a base. Contrary to popular belief, the pH value can be less than 0 or greater than 14 for very strong acids and bases respectively.pH measurements are important in medicine, biology, chemistry, agriculture, forestry, food science, environmental science, oceanography, civil engineering, chemical engineering, nutrition, water treatment & water purification, and many other applications. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.Primary pH standard values are determined using a concentration cell with transference, by measuring the potential difference between a hydrogen electrode and a standard electrode such as the silver chloride electrode.The pH of aqueous solutions can be measured with a glass electrode and a pH meter, or indicator.pH is the negative of the logarithm to base 10 of the activity of the (solvated) hydronium ion, more often (albeit somewhat inaccurately) expressed as the measure of the hydronium ion concentration.The rest of this article uses the technically correct word ""base"" and its inflections in place of ""alkaline"", which specifically refers to a base dissolved in water, and its inflections.