CHM134 General Chemistry I Semester Review – Dr. Steel This list
... Semester Review – Dr. Steel This list represents the most important topics we covered in CHM134 this semester. It is not a complete list of every topic that might appear on the final exam. Formulas and Constants K = °C + 273 M D= 1 in = 2.54 cm V metric conversions ...
... Semester Review – Dr. Steel This list represents the most important topics we covered in CHM134 this semester. It is not a complete list of every topic that might appear on the final exam. Formulas and Constants K = °C + 273 M D= 1 in = 2.54 cm V metric conversions ...
How to Make a Collage
... 19. An organic compound which has the empirical formula CHO has a molar mass of 232. Its molecular formula is: a) ...
... 19. An organic compound which has the empirical formula CHO has a molar mass of 232. Its molecular formula is: a) ...
1999 Advanced Placement Chemistry Exam
... 37. The ionization energies for element X are listed in (C) O2 the table above. On the basis of the data, element X is most likely to be (D) HF (E) F2 (A) Na (B) Mg 2 SO3(g) _ 2 SO2(g) + O2(g) (C) Al 41. After the equilibrium represented above is (D) Si (E) P established, some pure O2(g) is injected ...
... 37. The ionization energies for element X are listed in (C) O2 the table above. On the basis of the data, element X is most likely to be (D) HF (E) F2 (A) Na (B) Mg 2 SO3(g) _ 2 SO2(g) + O2(g) (C) Al 41. After the equilibrium represented above is (D) Si (E) P established, some pure O2(g) is injected ...
Chemistry MSL Practical Style Review 1. What is the nuclear
... pressure of N2 is 4 atm, and the partial pressure of O2 is 6 atm, what is the partial pressure of CO2? A B C D ...
... pressure of N2 is 4 atm, and the partial pressure of O2 is 6 atm, what is the partial pressure of CO2? A B C D ...
Document
... Complete the table that shows the reaction, if any, of the oxides with acid and alkali. Indicate a reaction with "R" and no reaction with "NR". ...
... Complete the table that shows the reaction, if any, of the oxides with acid and alkali. Indicate a reaction with "R" and no reaction with "NR". ...
Topic 2275 Ionic Mobilities: Aqueous Solutions. A classic subject in
... heading ‘thermodynamic properties’, these conductivities have played a major part in the task of understanding the thermodynamic properties of salt solutions. Generally however interest in the electric conductivities of salt solutions has waned as spectroscopic properties in all its form have moved ...
... heading ‘thermodynamic properties’, these conductivities have played a major part in the task of understanding the thermodynamic properties of salt solutions. Generally however interest in the electric conductivities of salt solutions has waned as spectroscopic properties in all its form have moved ...
Unit A Remediation Review
... example of each. 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustio ...
... example of each. 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustio ...
SC 119 PRACTICE Assessment:
... SC 119 PRACTICE Assessment: 1. Outdoor grilling is a very popular method of cooking. Propane is the gas that is commonly used in grills. Three things are required for a gas grill to ignite: gas, oxygen from the air and a spark.When the grill is turned on, propane is delivered to the igniter, where i ...
... SC 119 PRACTICE Assessment: 1. Outdoor grilling is a very popular method of cooking. Propane is the gas that is commonly used in grills. Three things are required for a gas grill to ignite: gas, oxygen from the air and a spark.When the grill is turned on, propane is delivered to the igniter, where i ...
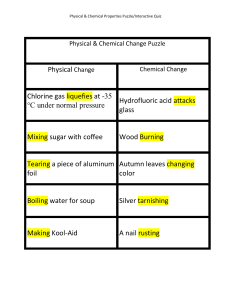
Physical Change Chlorine gas liquefies at
... The boiling point of a certain Carbon and oxygen yielding alcohol is 75 degrees Celsius carbon dioxide Perfume evaporating on your skin Table salt dissolves in water ...
... The boiling point of a certain Carbon and oxygen yielding alcohol is 75 degrees Celsius carbon dioxide Perfume evaporating on your skin Table salt dissolves in water ...
Molarity A measure of the concentration of a solution in moles per
... 1. Find molarity of a solution that has 3g MgCl2 dissolved in 50mL water. 3g MgCl2/95g/mol MgCl2 = 0.0315mol MgCl2 0.0315 mol MgCl2/0.05L = 0.632 M MgCl2 2. A student has 190mL of 1.1M MgCl2 solution. Find the mass of MgCl2 dissolved in solution. M(L)= mol (1.1)(0.19)=0.209 mol MgCl2 0.209 mol MgCl2 ...
... 1. Find molarity of a solution that has 3g MgCl2 dissolved in 50mL water. 3g MgCl2/95g/mol MgCl2 = 0.0315mol MgCl2 0.0315 mol MgCl2/0.05L = 0.632 M MgCl2 2. A student has 190mL of 1.1M MgCl2 solution. Find the mass of MgCl2 dissolved in solution. M(L)= mol (1.1)(0.19)=0.209 mol MgCl2 0.209 mol MgCl2 ...
Chemistry Notes
... Example of Bonds Let's look at Sodium Hydroxide (Na-OH)... You can see that on the left is the Sodium part and the right has the Oxygen/Hydrogen part. The bond which binds the Hydrogen to the Oxygen is covalent. The Sodium is bonded to the HYDROXIDE part of the compound with an ionic bond. This is a ...
... Example of Bonds Let's look at Sodium Hydroxide (Na-OH)... You can see that on the left is the Sodium part and the right has the Oxygen/Hydrogen part. The bond which binds the Hydrogen to the Oxygen is covalent. The Sodium is bonded to the HYDROXIDE part of the compound with an ionic bond. This is a ...
Electrochemistry questions for midterm or semi-final exam
... The magnitude of activity is given properly, if the applied concentration unit is also known. On diluting the solution, i.e. c→0 ...
... The magnitude of activity is given properly, if the applied concentration unit is also known. On diluting the solution, i.e. c→0 ...
Gas Laws
... 15. If excess hydrochloric acid is added to 13.5 grams of Al, what volume of hydrogen gas will be produced if the gas is collected at a temperature of 80.0 oC and a pressure of 750. torr? 22.1 L 16. At a certain temperature, the velocity of chlorine molecules is 0.0410 m/s. What is the velocity of s ...
... 15. If excess hydrochloric acid is added to 13.5 grams of Al, what volume of hydrogen gas will be produced if the gas is collected at a temperature of 80.0 oC and a pressure of 750. torr? 22.1 L 16. At a certain temperature, the velocity of chlorine molecules is 0.0410 m/s. What is the velocity of s ...
2. Electrodics
... Obviously, the faster the ion, the greater its contribution to the total current. If, and only if, the mobilities of anions and cations are exactly the same, the current will be transported in the same proportion (50%) by each species. To calculate the transference number one does not need the absol ...
... Obviously, the faster the ion, the greater its contribution to the total current. If, and only if, the mobilities of anions and cations are exactly the same, the current will be transported in the same proportion (50%) by each species. To calculate the transference number one does not need the absol ...
PH

In chemistry, pH (/piːˈeɪtʃ/) is a numeric scale used to specify the acidity or alkalinity of an aqueous solution. It is the negative of the logarithm to base 10 of the activity of the hydrogen ion. Solutions with a pH less than 7 are acidic and solutions with a pH greater than 7 are alkaline or basic. Pure water is neutral, being neither an acid nor a base. Contrary to popular belief, the pH value can be less than 0 or greater than 14 for very strong acids and bases respectively.pH measurements are important in medicine, biology, chemistry, agriculture, forestry, food science, environmental science, oceanography, civil engineering, chemical engineering, nutrition, water treatment & water purification, and many other applications. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.Primary pH standard values are determined using a concentration cell with transference, by measuring the potential difference between a hydrogen electrode and a standard electrode such as the silver chloride electrode.The pH of aqueous solutions can be measured with a glass electrode and a pH meter, or indicator.pH is the negative of the logarithm to base 10 of the activity of the (solvated) hydronium ion, more often (albeit somewhat inaccurately) expressed as the measure of the hydronium ion concentration.The rest of this article uses the technically correct word ""base"" and its inflections in place of ""alkaline"", which specifically refers to a base dissolved in water, and its inflections.