Atoms, Ions, and Molecules File
... Atoms of different elements are different. • Atoms are not changed into different atoms in a chemical reaction. • Compounds are formed when atoms of two or more elements combine. ...
... Atoms of different elements are different. • Atoms are not changed into different atoms in a chemical reaction. • Compounds are formed when atoms of two or more elements combine. ...
Chem 11 Study Guide SCH3U Unit 1 Definitions: SATP: Standard
... Arrhenius acids and bases According to the Arrhenius definition, an acid is any substance, which when dissolved in water, tends to increase the amount of . An example is HCl: ...
... Arrhenius acids and bases According to the Arrhenius definition, an acid is any substance, which when dissolved in water, tends to increase the amount of . An example is HCl: ...
10 TEST 2 (of 3)
... volume were reduced to 0.5 L and the temperature increased to 250 ºC. What would the new pressure be? ...
... volume were reduced to 0.5 L and the temperature increased to 250 ºC. What would the new pressure be? ...
California Chemistry Standards Test
... 1. covalent bonds, metallic bonds and ionic bonds 2. know examples of bonds 3. crystals and structure 4. intermolecular forces 5. Lewis dot structures Conservation of Matter & Stoichiometry-(10) 1. balanced equation 2. carbon12 as the mole standard 3. what a mole equals 4. molar mass 5. stoichiometr ...
... 1. covalent bonds, metallic bonds and ionic bonds 2. know examples of bonds 3. crystals and structure 4. intermolecular forces 5. Lewis dot structures Conservation of Matter & Stoichiometry-(10) 1. balanced equation 2. carbon12 as the mole standard 3. what a mole equals 4. molar mass 5. stoichiometr ...
Acid K a
... 2.0 moles of NH3 gas are introduced into a previously evacuated 1.0 L container. At a certain temperature the NH3 partially dissociates by the following equation. 2 NH3(g) ...
... 2.0 moles of NH3 gas are introduced into a previously evacuated 1.0 L container. At a certain temperature the NH3 partially dissociates by the following equation. 2 NH3(g) ...
Chem Sheets to Memorize SOLUBILITY CHART
... single displacement/replacement). Any ion has an aqueous state of matter. For acid-base reactions, strong acids (HCl, HBr, HI, H2SO4, HClO4, and HNO3) and strong bases (metal ions in groups 1 and 2 paired with hydroxide (OH-) completely dissociate. Weak acids and bases do not. For precipitation (and ...
... single displacement/replacement). Any ion has an aqueous state of matter. For acid-base reactions, strong acids (HCl, HBr, HI, H2SO4, HClO4, and HNO3) and strong bases (metal ions in groups 1 and 2 paired with hydroxide (OH-) completely dissociate. Weak acids and bases do not. For precipitation (and ...
Chapter 3
... 1) Substitution- one group replaces another 2) Additions- All parts of the adding agent appear in the product. Two compounds become one. 3) Eliminations- One molecule loses the elements of another small molecule 4) Rearrangements-A molecule undergoes a reorganization of its constituent parts ...
... 1) Substitution- one group replaces another 2) Additions- All parts of the adding agent appear in the product. Two compounds become one. 3) Eliminations- One molecule loses the elements of another small molecule 4) Rearrangements-A molecule undergoes a reorganization of its constituent parts ...
Notes
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble substance formed by the reaction of ...
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble substance formed by the reaction of ...
ATOMS, MOLECULES, AND IONS
... kg. Later experiments by Rutherford determined that at the center of an atom is a positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic number of the atom). The fundamental unit of positive charge in the nucleus is the proton. ♦ Chemical identity of an ...
... kg. Later experiments by Rutherford determined that at the center of an atom is a positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic number of the atom). The fundamental unit of positive charge in the nucleus is the proton. ♦ Chemical identity of an ...
Preview Sample 1
... 1. List several differences between ionic and covalent bonds. Ionic bonds occur when ions of opposite charge are mutually attracted. Acids and bases are examples of ionic compounds. Covalent bonds are strong chemical bonds that occur when atoms share electrons. Methane and sugar are examples of cova ...
... 1. List several differences between ionic and covalent bonds. Ionic bonds occur when ions of opposite charge are mutually attracted. Acids and bases are examples of ionic compounds. Covalent bonds are strong chemical bonds that occur when atoms share electrons. Methane and sugar are examples of cova ...
1. Cl2 + 2Br- ® 2Cl- + Br2 formulae correct for elements 1 correct
... (do not allow bonds or no forces, allow inter molecular forces are weak, do not allow they have weak forces / bonds) so little heat / energy is required before they can overcome forces / move freely / break out of solid structure / lattice (N.B. second point can be gained even if first is not) ...
... (do not allow bonds or no forces, allow inter molecular forces are weak, do not allow they have weak forces / bonds) so little heat / energy is required before they can overcome forces / move freely / break out of solid structure / lattice (N.B. second point can be gained even if first is not) ...
File
... B. balance oxygen using H2O C. balance hydrogen using H+ D. balance the charge using electrons 3. if necessary, multiply one or both half-reactions by an integer to equalize the number of electrons transferred in the two half reactions 4. add the half-reactions, and cancel identical species 5. check ...
... B. balance oxygen using H2O C. balance hydrogen using H+ D. balance the charge using electrons 3. if necessary, multiply one or both half-reactions by an integer to equalize the number of electrons transferred in the two half reactions 4. add the half-reactions, and cancel identical species 5. check ...
Vocabulary CHEM121
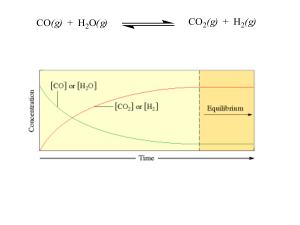
... Bases: anything giving OH- when dissolved in water Salts: all other ionic materials Formulas of molecular/covalent compounds: Non-metals can combine with other non-metals to form molecules. Molecules are covalently bonded groups of atoms—they do not have a charge like polyatomic ions, but are neutra ...
... Bases: anything giving OH- when dissolved in water Salts: all other ionic materials Formulas of molecular/covalent compounds: Non-metals can combine with other non-metals to form molecules. Molecules are covalently bonded groups of atoms—they do not have a charge like polyatomic ions, but are neutra ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Separate the reactions into oxidation and reduction processes Work with one (ox or red) first Balance number of non-oxygen, non-hydrogen atoms first. Then balance oxygen with water Then balance hydrogen with H+ Then balance charge with electrons. Then balance other half-reaction using steps 3 throug ...
... Separate the reactions into oxidation and reduction processes Work with one (ox or red) first Balance number of non-oxygen, non-hydrogen atoms first. Then balance oxygen with water Then balance hydrogen with H+ Then balance charge with electrons. Then balance other half-reaction using steps 3 throug ...
namimg compounds
... As the number of named compounds increased it was obvious that if such common names were used, confusion would result. In 1787, in order to solve the problem, a scientist named Lavoisier established the principles for a systematic naming process. There are, however, some familiar compounds that are ...
... As the number of named compounds increased it was obvious that if such common names were used, confusion would result. In 1787, in order to solve the problem, a scientist named Lavoisier established the principles for a systematic naming process. There are, however, some familiar compounds that are ...
Chapter 5
... • one of the best solvents is water, because it is a polar molecule • Electrolytes are substances whose aqueous solutions conduct electricity • When an ionic substance is broken apart into the solvated cations and anions, the process is properly called dissociation. However, when the ions are formed ...
... • one of the best solvents is water, because it is a polar molecule • Electrolytes are substances whose aqueous solutions conduct electricity • When an ionic substance is broken apart into the solvated cations and anions, the process is properly called dissociation. However, when the ions are formed ...
EXAM # 1
... the electrode. Crystalline Membrane Electrode, Liquid Membrane Electrode or Enzyme electrodes are common examples ...
... the electrode. Crystalline Membrane Electrode, Liquid Membrane Electrode or Enzyme electrodes are common examples ...
PH

In chemistry, pH (/piːˈeɪtʃ/) is a numeric scale used to specify the acidity or alkalinity of an aqueous solution. It is the negative of the logarithm to base 10 of the activity of the hydrogen ion. Solutions with a pH less than 7 are acidic and solutions with a pH greater than 7 are alkaline or basic. Pure water is neutral, being neither an acid nor a base. Contrary to popular belief, the pH value can be less than 0 or greater than 14 for very strong acids and bases respectively.pH measurements are important in medicine, biology, chemistry, agriculture, forestry, food science, environmental science, oceanography, civil engineering, chemical engineering, nutrition, water treatment & water purification, and many other applications. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.Primary pH standard values are determined using a concentration cell with transference, by measuring the potential difference between a hydrogen electrode and a standard electrode such as the silver chloride electrode.The pH of aqueous solutions can be measured with a glass electrode and a pH meter, or indicator.pH is the negative of the logarithm to base 10 of the activity of the (solvated) hydronium ion, more often (albeit somewhat inaccurately) expressed as the measure of the hydronium ion concentration.The rest of this article uses the technically correct word ""base"" and its inflections in place of ""alkaline"", which specifically refers to a base dissolved in water, and its inflections.