Ch9
... Which of the following radioisotopes are most likely to be used in nuclear medicine? Radioisotopes with short half-lives are used in nuclear medicine. ...
... Which of the following radioisotopes are most likely to be used in nuclear medicine? Radioisotopes with short half-lives are used in nuclear medicine. ...
The Band of Stability
... Introduction: Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to rep ...
... Introduction: Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to rep ...
3 Background radiation
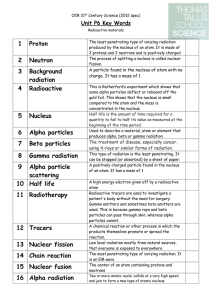
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
I Examen II trim Science
... Protons (P+): Positively charged sub-particles that are located in the nucleus (central part of the atom). Electrons: (e-): Negatively charged sub-particles that move around the nucleus in energy levels. Electrons are found likely in the electron clouds. Neutrons: (n): Sub-particles located in the n ...
... Protons (P+): Positively charged sub-particles that are located in the nucleus (central part of the atom). Electrons: (e-): Negatively charged sub-particles that move around the nucleus in energy levels. Electrons are found likely in the electron clouds. Neutrons: (n): Sub-particles located in the n ...
atomic number.
... Radioactive Atoms mutate by fission or fusion until they have maximum Binding Energy per nucleon which occurs at Iron. ...
... Radioactive Atoms mutate by fission or fusion until they have maximum Binding Energy per nucleon which occurs at Iron. ...
Nuclear Chemistry powerpoint
... minutes. If you start with 20.0 g, how long will it take before only 1.25 g remains? ______________ ...
... minutes. If you start with 20.0 g, how long will it take before only 1.25 g remains? ______________ ...