Tech Data Sheet - Direct N
... slowly to prevent fracturing the formation. If the DNP-09MS-800 treatment is mixed in acid, the well should be shut in for a period of time to allow the acid to dissolve solids that are plugging the pore throats. Producing wells should then be returned to production, with back flowing of ...
... slowly to prevent fracturing the formation. If the DNP-09MS-800 treatment is mixed in acid, the well should be shut in for a period of time to allow the acid to dissolve solids that are plugging the pore throats. Producing wells should then be returned to production, with back flowing of ...
Solution to Sample Problems in Recitation 2 - Pruffle
... Sometimes it is useful to draw a diagram representing the system to study: Water ...
... Sometimes it is useful to draw a diagram representing the system to study: Water ...
HONORS CHEMISTRY STUDY GUIDE Thermochemistry
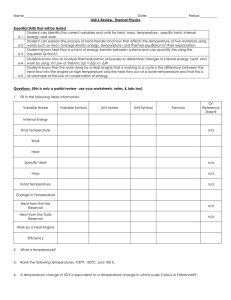
... as well as the names of the heats associated with the phase changes sections. -Be able to do problems determining the heat where the temperature and phase of a substance changes. -Understand that in such problems each section of the heat curve needs its own calculation. The total heat can then be de ...
... as well as the names of the heats associated with the phase changes sections. -Be able to do problems determining the heat where the temperature and phase of a substance changes. -Understand that in such problems each section of the heat curve needs its own calculation. The total heat can then be de ...
Unit 6 Review
... 15. A gas undergoes an isobaric process where it experiences an increase in temperature while expanding. Is heat being added or removed during this process? Analyze the first law of thermodynamics to help you answer this question. ...
... 15. A gas undergoes an isobaric process where it experiences an increase in temperature while expanding. Is heat being added or removed during this process? Analyze the first law of thermodynamics to help you answer this question. ...
Document
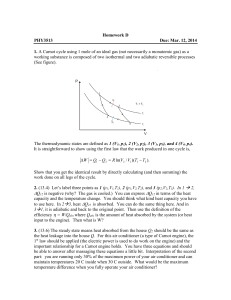
... ∆Q12 is negative (why? The gas is cooled.) You can express ∆Q12 in terms of the heat capacity and the temperature change. You should think what kind heat capacity you have to use here. In 23, heat ∆Q23 is absorbed. You can do the same thing here. And in 31, it is adiabatic and back to the original ...
... ∆Q12 is negative (why? The gas is cooled.) You can express ∆Q12 in terms of the heat capacity and the temperature change. You should think what kind heat capacity you have to use here. In 23, heat ∆Q23 is absorbed. You can do the same thing here. And in 31, it is adiabatic and back to the original ...
Countercurrent exchange

Countercurrent exchange is a mechanism occurring in nature and mimicked in industry and engineering, in which there is a crossover of some property, usually heat or some component, between two flowing bodies flowing in opposite directions to each other. The flowing bodies can be liquids, gases, or even solid powders, or any combination of those. For example, in a distillation column, the vapors bubble up through the downward flowing liquid while exchanging both heat and mass.The maximum amount of heat or mass transfer that can be obtained is higher with countercurrent than co-current (parallel) exchange because countercurrent maintains a slowly declining difference or gradient (usually temperature or concentration difference). In cocurrent exchange the initial gradient is higher but falls off quickly, leading to wasted potential. For example, in the diagram at the right, the fluid being heated (exiting top) has a higher exiting temperature than the cooled fluid (exiting bottom) that was used for heating. With cocurrent or parallel exchange the heated and cooled fluids can only approach one another. The result is that countercurrent exchange can achieve a greater amount of heat or mass transfer than parallel under otherwise similar conditions. See: flow arrangement.Countercurrent exchange when set up in a circuit or loop can be used for building up concentrations, heat, or other properties of flowing liquids. Specifically when set up in a loop with a buffering liquid between the incoming and outgoing fluid running in a circuit, and with active transport pumps on the outgoing fluid's tubes, the system is called a Countercurrent multiplier, enabling a multiplied effect of many small pumps to gradually build up a large concentration in the buffer liquid.Other countercurrent exchange circuits where the incoming and outgoing fluids touch each other are used for retaining a high concentration of a dissolved substance or for retaining heat, or for allowing the external buildup of the heat or concentration at one point in the system.Countercurrent exchange circuits or loops are found extensively in nature, specifically in biologic systems. In vertebrates, they are called a Rete mirabile, originally the name of an organ in fish gills for absorbing oxygen from the water. It is mimicked in industrial systems. Countercurrent exchange is a key concept in chemical engineering thermodynamics and manufacturing processes, for example in extracting sucrose from sugar beet roots.Countercurrent multiplication is a similar but different concept where liquid moves in a loop followed by a long length of movement in opposite directions with an intermediate zone. The tube leading to the loop passively building up a gradient of heat (or cooling) or solvent concentration while the returning tube has a constant small pumping action all along it, so that a gradual intensification of the heat or concentration is created towards the loop. Countercurrent multiplication has been found in the kidneys as well as in many other biological organs.