solid_solutions_mse528
... where in 1958, he was appointed to the first chair in metallurgy. • His research led to some simple and useful rules on the extent to which an element might dissolve in a metal . The rules that he derived are paraphrased here. The rules are still used widely. For example, the miscibility gap in Au-N ...
... where in 1958, he was appointed to the first chair in metallurgy. • His research led to some simple and useful rules on the extent to which an element might dissolve in a metal . The rules that he derived are paraphrased here. The rules are still used widely. For example, the miscibility gap in Au-N ...
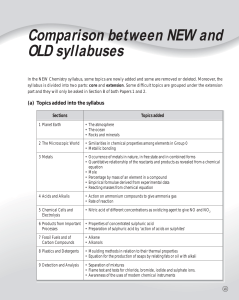
Chapter 23 Metals and Metallurgy
... observed trends in melting point, boiling point, heat of fusion, etc. Suggests these properties should increase with increasing number of valence electrons. ...
... observed trends in melting point, boiling point, heat of fusion, etc. Suggests these properties should increase with increasing number of valence electrons. ...
Biologically Important Inorganic Elements Occurrence and Availability
... • Despite the high abundance of Si, Al and Ti (the 2nd, 3rd and 10th most abundant elements on earth). Why are they are not utilized biologically? • Because of the insolubility of their naturally occurring oxides (SiO2, Al2O3, TiO2) under physiological conditions. A lower oxidation state is unavaila ...
... • Despite the high abundance of Si, Al and Ti (the 2nd, 3rd and 10th most abundant elements on earth). Why are they are not utilized biologically? • Because of the insolubility of their naturally occurring oxides (SiO2, Al2O3, TiO2) under physiological conditions. A lower oxidation state is unavaila ...
chapter 1 - College Test bank - get test bank and solution manual
... 7. This answer can vary between students, but the combustion of gasoline to propel vehicles is an example of a useful chemical reaction. This reaction is a chemical change because the liquid gasoline is converted to heat and gases. Another constructive example is the burning of coal to heat water in ...
... 7. This answer can vary between students, but the combustion of gasoline to propel vehicles is an example of a useful chemical reaction. This reaction is a chemical change because the liquid gasoline is converted to heat and gases. Another constructive example is the burning of coal to heat water in ...
Metallic Crystal Structure
... Most of the elements are chemically reactive, so that we find very few of them in their elemental state in the Earth's crust—oxygen and nitrogen mixed together in the atmosphere are the most common, whilst a few metals such as copper, gold and silver, also occur uncombined. Most of the substances we ...
... Most of the elements are chemically reactive, so that we find very few of them in their elemental state in the Earth's crust—oxygen and nitrogen mixed together in the atmosphere are the most common, whilst a few metals such as copper, gold and silver, also occur uncombined. Most of the substances we ...
the Main-Group Metals - McQuarrie General Chemistry
... polished aluminum has a bright, silvery appearance, but weathered aluminum has a dull tarnish because of the aluminum oxide coating. Structural alloys of aluminum for aircraft, automobiles, and some bicycles contain silicon, copper, magnesium, and other metals, which increase the strength and stiffn ...
... polished aluminum has a bright, silvery appearance, but weathered aluminum has a dull tarnish because of the aluminum oxide coating. Structural alloys of aluminum for aircraft, automobiles, and some bicycles contain silicon, copper, magnesium, and other metals, which increase the strength and stiffn ...
File - Mr Weng`s IB Chemistry
... • The properties of a material based on the degree of covalent, ionic or metallic character in a compound can be deduced from its position on a bonding triangle. • Composites are mixtures in which materials are composed of two distinct phases, a reinforcing phase that is embedded in a matrix phase. ...
... • The properties of a material based on the degree of covalent, ionic or metallic character in a compound can be deduced from its position on a bonding triangle. • Composites are mixtures in which materials are composed of two distinct phases, a reinforcing phase that is embedded in a matrix phase. ...
Fig. 6. Typical circuits with high magnetic permeability
... initial and finished melt state and wishes to each the tc during melting. Some times the melt may be heated only to the anomaly temperatures tan. The melt holding time at the maximal heating temperature is experimentally during the investigation of the time dependence of properties changes at this t ...
... initial and finished melt state and wishes to each the tc during melting. Some times the melt may be heated only to the anomaly temperatures tan. The melt holding time at the maximal heating temperature is experimentally during the investigation of the time dependence of properties changes at this t ...
Formation of binary alloy cluster ions from group
... the class of fusible alloys, and lead is a raw material for leadbased alloys. Because a small change in composition can lead to special properties, many systems such as T/Sn, Au/T/Sn and Sn/Pb2±6 have also been studied. In recent years, the investigations of these systems have focused on the stabili ...
... the class of fusible alloys, and lead is a raw material for leadbased alloys. Because a small change in composition can lead to special properties, many systems such as T/Sn, Au/T/Sn and Sn/Pb2±6 have also been studied. In recent years, the investigations of these systems have focused on the stabili ...
Unit 10
... products formed in the reaction. Write down the correct formulae of reactants on the left hand side of the arrow. Write down the correct formulae of products on the right hand side of the arrow. Balance the equation with simple whole numbers such that the total number of each type of atoms are equal ...
... products formed in the reaction. Write down the correct formulae of reactants on the left hand side of the arrow. Write down the correct formulae of products on the right hand side of the arrow. Balance the equation with simple whole numbers such that the total number of each type of atoms are equal ...
01.CN_Other pages/p1-9
... (b) (i) Which particle(s) is / are the ions? Hint 2 (ii) What is the relationship between P and Q? (iii) Do particles of P and Q have the same chemical properties? Explain your answer. (c) (i) Suggest a term to indicate the relationship between S and T. (ii) Explain why S and T have the same chemica ...
... (b) (i) Which particle(s) is / are the ions? Hint 2 (ii) What is the relationship between P and Q? (iii) Do particles of P and Q have the same chemical properties? Explain your answer. (c) (i) Suggest a term to indicate the relationship between S and T. (ii) Explain why S and T have the same chemica ...
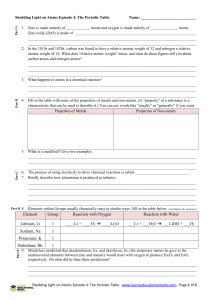
Element Group Reaction with Oxygen Reaction with Water Lithium
... Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2. In the 1810s and 1820s, carbon w ...
... Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2. In the 1810s and 1820s, carbon w ...
FORMATION, COMPOSITION AND SOME PROPERTIES OF
... According to XSA the film site with dispersed particles includes next elements, at %: 1.4 Pt, 22.4 Ti, 11.8 P, 64.4 O, i.e. the concentrations of platinum and titanium in dispersed particles (micrograines) are higher that those in the film. According to (6) conditions for formation of amorphous or ...
... According to XSA the film site with dispersed particles includes next elements, at %: 1.4 Pt, 22.4 Ti, 11.8 P, 64.4 O, i.e. the concentrations of platinum and titanium in dispersed particles (micrograines) are higher that those in the film. According to (6) conditions for formation of amorphous or ...
Chapter 4: Imperfections in Solids Imperfections in Solids
... •The properties of some materials are significantly influenced by the presence of imperfections. •It is important to have a knowledge about the types of imperfections that exist and the roles they play in affecting the behavior of materials. •The mechanical properties of pure metals experience signi ...
... •The properties of some materials are significantly influenced by the presence of imperfections. •It is important to have a knowledge about the types of imperfections that exist and the roles they play in affecting the behavior of materials. •The mechanical properties of pure metals experience signi ...
Low temperature surface hardening of stainless steel
... development of the protective surface film. However, it was demonstrated that the incorporation of nitrogen and/or carbon in austenitic stainless steel brings about the development of expanded austenite (also referred to as S-phase), which spectacularly improves the tribological and wear performance ...
... development of the protective surface film. However, it was demonstrated that the incorporation of nitrogen and/or carbon in austenitic stainless steel brings about the development of expanded austenite (also referred to as S-phase), which spectacularly improves the tribological and wear performance ...
b) Mole
... 21. Which byproduct gas is released when an acid reacts with metal? a) dioxygen b) dehydrogen c) dichlorine d) dinitrogen 22. According to Arrhenius , which ion is released from a substance so that it becomes Arrhenius base? a) H+ b) OH c) N3 d) O2 23. In H3 O+, there is coordinate covalent bond bet ...
... 21. Which byproduct gas is released when an acid reacts with metal? a) dioxygen b) dehydrogen c) dichlorine d) dinitrogen 22. According to Arrhenius , which ion is released from a substance so that it becomes Arrhenius base? a) H+ b) OH c) N3 d) O2 23. In H3 O+, there is coordinate covalent bond bet ...
Introduction
... temperature range, high temperature resistance ( refractoriness), thermal shock resistance, extreme wear resistance, electrical insulation, chemical inertness, corrosion resistance at low and high temperatures, high mechanical strength, hardness properties. Many properties or combinations of propert ...
... temperature range, high temperature resistance ( refractoriness), thermal shock resistance, extreme wear resistance, electrical insulation, chemical inertness, corrosion resistance at low and high temperatures, high mechanical strength, hardness properties. Many properties or combinations of propert ...
Heat Treatment Effect on Multicomponent Nickel Alloys Structure
... metallic carbides (MC) with a tendency to their own degradation during heat treatments. They generate into carbides M23C6 and/or MC6, which segregate to the grain boundaries. Carbides at the grain boundaries have mostly positive influence on the rupture stress at high temperatures [3,4]. Carbides im ...
... metallic carbides (MC) with a tendency to their own degradation during heat treatments. They generate into carbides M23C6 and/or MC6, which segregate to the grain boundaries. Carbides at the grain boundaries have mostly positive influence on the rupture stress at high temperatures [3,4]. Carbides im ...
2. Objectives - McMaster Materials Science and Engineering
... determine the range of validity of Eq. 7. The goal of this step will be to determine regimes where data generated from the simplified model will be a good approximation to that of full model. 3. To develop a similarity solution that predicts the coarseing rate in spinodal decomposition using a simil ...
... determine the range of validity of Eq. 7. The goal of this step will be to determine regimes where data generated from the simplified model will be a good approximation to that of full model. 3. To develop a similarity solution that predicts the coarseing rate in spinodal decomposition using a simil ...
CHEM 120 WEEK 11 LECTURES (INORGANIC WEEK 2) Dr. MD
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
Electroless plating of rhenium-based alloys with nickel, cobalt and iron
... that addition of Ni2+ ions to the plating bath is required to start the induced co-deposition of Re. Moreover, Ni2+ acts as a catalyst for Re reduction. Therefore, it was of interest to know if other iron-group metals (namely, Co and Fe) induce Re electroless plating too. The goal of this work is to ...
... that addition of Ni2+ ions to the plating bath is required to start the induced co-deposition of Re. Moreover, Ni2+ acts as a catalyst for Re reduction. Therefore, it was of interest to know if other iron-group metals (namely, Co and Fe) induce Re electroless plating too. The goal of this work is to ...
DETERMINATION OF ACTIVATION ENERGY IN HOT
... laboratory casting over its height (or thickness 20 mm). Similarly the cylindrical specimens were oriented, where the areas with the as small as possible occurrence of the internal cavities were chosen, which complicated the preparation of specimens and their following hot forming. Uniaxial hot comp ...
... laboratory casting over its height (or thickness 20 mm). Similarly the cylindrical specimens were oriented, where the areas with the as small as possible occurrence of the internal cavities were chosen, which complicated the preparation of specimens and their following hot forming. Uniaxial hot comp ...
MAGNESIUM
... Magnesium is a chemical element with atomic number 12. It is an light alkali metal, medium strong, shiny gray with a density of 1.73 ton per cubic meter with a hexagonal crystal structure making it the lightest metal material that can be used in a practical way (two-thirds of aluminum density and a ...
... Magnesium is a chemical element with atomic number 12. It is an light alkali metal, medium strong, shiny gray with a density of 1.73 ton per cubic meter with a hexagonal crystal structure making it the lightest metal material that can be used in a practical way (two-thirds of aluminum density and a ...
The ocean is a mixture.
... in the B families. These are the metals you are probably most familiar: copper, tin, zinc, iron, nickel, gold, and silver. They are good conductors of heat and electricity. ...
... in the B families. These are the metals you are probably most familiar: copper, tin, zinc, iron, nickel, gold, and silver. They are good conductors of heat and electricity. ...
Alloy

An alloy is a mixture of metals or a mixture of a metal and another element. Alloys are defined by metallic bonding character. An alloy may be a solid solution of metal elements (a single phase) or a mixture of metallic phases (two or more solutions). Intermetallic compounds are alloys with a defined stoichiometry and crystal structure. Zintl phases are also sometimes considered alloys depending on bond types (see also: Van Arkel-Ketelaar triangle for information on classifying bonding in binary compounds).Alloys are used in a wide variety of applications. In some cases, a combination of metals may reduce the overall cost of the material while preserving important properties. In other cases, the combination of metals imparts synergistic properties to the constituent metal elements such as corrosion resistance or mechanical strength. Examples of alloys are steel, solder, brass, pewter, duralumin, phosphor bronze and amalgams.The alloy constituents are usually measured by mass. Alloys are usually classified as substitutional or interstitial alloys, depending on the atomic arrangement that forms the alloy. They can be further classified as homogeneous (consisting of a single phase), or heterogeneous (consisting of two or more phases) or intermetallic.