INTRODUCTION TO THE PERIODIC TABLE
... random letters and numbers, right? Well it's actually one of the most important tools for understanding chemistry! Let's go through it piece by piece and soon enough you'll not only understand how it works, you'll be able to use it to figure out how and why elements interact to make the world you se ...
... random letters and numbers, right? Well it's actually one of the most important tools for understanding chemistry! Let's go through it piece by piece and soon enough you'll not only understand how it works, you'll be able to use it to figure out how and why elements interact to make the world you se ...
Atomic Number, Mass Number, and Isotopes
... protons determines the identity of the atom. For example, a carbon atom always has six protons. If it has seven protons, it’s nitrogen, not carbon. The number of protons is called the atomic number (Z). ISOTOPES: Although all atoms of an element have the same number of protons, they don’t all have t ...
... protons determines the identity of the atom. For example, a carbon atom always has six protons. If it has seven protons, it’s nitrogen, not carbon. The number of protons is called the atomic number (Z). ISOTOPES: Although all atoms of an element have the same number of protons, they don’t all have t ...
Overview Properties of Atoms and the Periodic Table
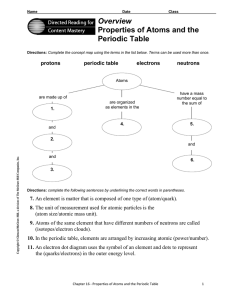
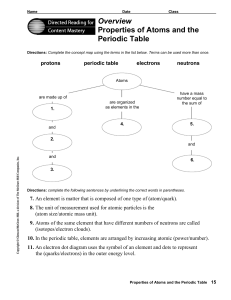
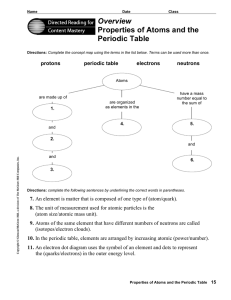
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. ...
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. ...
Overview Properties of Atoms and the Periodic Table
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. ...
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. ...
Atomic Structure of hydrogen
... Cancer treatment – A weak beam of radiation will kill cancer cells more readily than healthy cells. Carbon dating – All living things contain a known proportion of radioactive carbon-14 atoms. When an organism dies it stops taking in new carbon atoms so the proportion of carbon-14 atoms slowly drops ...
... Cancer treatment – A weak beam of radiation will kill cancer cells more readily than healthy cells. Carbon dating – All living things contain a known proportion of radioactive carbon-14 atoms. When an organism dies it stops taking in new carbon atoms so the proportion of carbon-14 atoms slowly drops ...
Properties of Atoms and the Periodic Table
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. electron cloud six number electrons isotopes six protons neutron(s) proton(s) mass quarks The electron has vcry little mass compared to the 1. ...
... Directions: Use the terms below to complete the following paragraphs about atoms, atomic mass, and isotopes. Terms may be used more than once. electron cloud six number electrons isotopes six protons neutron(s) proton(s) mass quarks The electron has vcry little mass compared to the 1. ...
AP Chem Test 5-7 Practice Exam - mvhs
... 3. Name the element corresponding to each characteristic below. the element with the electron configuration 1s22s22p63s23p3 the alkaline earth element with the smallest atomic radius the element with the largest ionization energy in Group 5A the element whose 2+ ion has the configuration [Kr] 4d5 th ...
... 3. Name the element corresponding to each characteristic below. the element with the electron configuration 1s22s22p63s23p3 the alkaline earth element with the smallest atomic radius the element with the largest ionization energy in Group 5A the element whose 2+ ion has the configuration [Kr] 4d5 th ...
Introduction to Atoms - Mother Teresa Regional School
... are made up of smaller particles called protons, neutrons and electrons. An atom consists of a nucleus surrounded by one or more electrons. The nucleus is the very small center core of an atoms. Protons have a positive electric charge. Neutrons have no charge – they are neutral Electrons m ...
... are made up of smaller particles called protons, neutrons and electrons. An atom consists of a nucleus surrounded by one or more electrons. The nucleus is the very small center core of an atoms. Protons have a positive electric charge. Neutrons have no charge – they are neutral Electrons m ...
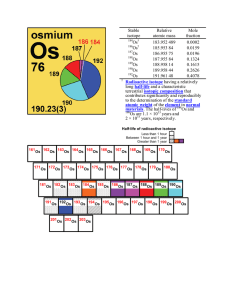
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
Chapter 4: Atomic Structure
... the region the nucleus. (a tiny central core of an atom and is composed of protons and neutrons) Rutherford’s atomic model is known as the nuclear atom. In the nuclear atom, the protons and neutrons are located in the nucleus. The electrons are distributed around the nucleus and occupy almost all of ...
... the region the nucleus. (a tiny central core of an atom and is composed of protons and neutrons) Rutherford’s atomic model is known as the nuclear atom. In the nuclear atom, the protons and neutrons are located in the nucleus. The electrons are distributed around the nucleus and occupy almost all of ...
Lesson 13 - Highline Public Schools
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
The Size of the Atom Atomic Numbers Atomic Mass Numbers
... diagram on page 201, picturing the basic structure of the atom, is not drawn to scale. If the nucleus were as large as shown, the electron cloud would extend far beyond your classroom. The negative electrons stick around the nucleus because they are attracted to the positively charged protons. Elect ...
... diagram on page 201, picturing the basic structure of the atom, is not drawn to scale. If the nucleus were as large as shown, the electron cloud would extend far beyond your classroom. The negative electrons stick around the nucleus because they are attracted to the positively charged protons. Elect ...
Reading Assignment Worksheet on Atoms - District 196 e
... 2. In this table, where are the metals located? 3. Where are the nonmetals located? 4. What are the elements in Groups 3 through 12 called? 5. What are the elements called that are next to the stairstep-shaped line on the right side of the table? 6. What do we call the letter or letters that represe ...
... 2. In this table, where are the metals located? 3. Where are the nonmetals located? 4. What are the elements in Groups 3 through 12 called? 5. What are the elements called that are next to the stairstep-shaped line on the right side of the table? 6. What do we call the letter or letters that represe ...
periodic table
... What is something you thought was cool/unique about it? What is periodic? What is periodic about the table? ...
... What is something you thought was cool/unique about it? What is periodic? What is periodic about the table? ...
ATOMS and PERIODIC TABLE - John Q. Adams Middle School
... Family IVA is half full making it more stable than IIIA or VA on either side of it. ...
... Family IVA is half full making it more stable than IIIA or VA on either side of it. ...
800 - Paint Valley Local Schools
... with other elements very easily. This makes them _______ _________ especially with group 17 because they have 7 valence electrons and when the two combine they make the magic number ...
... with other elements very easily. This makes them _______ _________ especially with group 17 because they have 7 valence electrons and when the two combine they make the magic number ...
and View
... atom. * Number never changes* b. Isotopes—atoms of same element that have different numbers of neutrons. Ex: carbon-12, carbon-13, carbon-14 c. Mass number—number of neutrons plus protons in an atom. i. Neutron number is found by--Mass number - Atomic number _______________ Number of neutrons ...
... atom. * Number never changes* b. Isotopes—atoms of same element that have different numbers of neutrons. Ex: carbon-12, carbon-13, carbon-14 c. Mass number—number of neutrons plus protons in an atom. i. Neutron number is found by--Mass number - Atomic number _______________ Number of neutrons ...
The Periodic Table OL Page 1 of 2 G. Galvin Name: Periodic Table
... 1. Mendeleev: Arranged the elements in order of increasing weight. Defn: Mendeleev’s Periodic Law: When elements are arranged in order of increasing atomic weight, the properties of the elements recur periodically, i.e. the properties displayed by the element are repeated at regular intervals in oth ...
... 1. Mendeleev: Arranged the elements in order of increasing weight. Defn: Mendeleev’s Periodic Law: When elements are arranged in order of increasing atomic weight, the properties of the elements recur periodically, i.e. the properties displayed by the element are repeated at regular intervals in oth ...
Periodic Table Review Key
... H 3. Which non-metal has the smallest atomic mass? F 4. Which metal has the largest atomic mass? D 5. Which elements are considered good conductors? C,E,D, 6. Which element is considered semi-conductors? G 7. Which elements are considered poor conductors? F, B, A,H 8. Which element is considered met ...
... H 3. Which non-metal has the smallest atomic mass? F 4. Which metal has the largest atomic mass? D 5. Which elements are considered good conductors? C,E,D, 6. Which element is considered semi-conductors? G 7. Which elements are considered poor conductors? F, B, A,H 8. Which element is considered met ...
Atomic Structure Worksheet
... 4. Atoms of the same element that differ in their number of neutrons in the nucleus are called isotopes. 5. The total number of nucleons (particles in the nucleus) in the atom make up the mass number. 6. A neutral nuclear particle having a mass of about 1 AMU is called the neutron. 7. The proton is ...
... 4. Atoms of the same element that differ in their number of neutrons in the nucleus are called isotopes. 5. The total number of nucleons (particles in the nucleus) in the atom make up the mass number. 6. A neutral nuclear particle having a mass of about 1 AMU is called the neutron. 7. The proton is ...
Atoms and the Periodic Table
... • Malleable material can be pounded into shapes. • Ductile material can be pulled out or drawn into a long wire. ...
... • Malleable material can be pounded into shapes. • Ductile material can be pulled out or drawn into a long wire. ...
ps-5-1-and-5-2-ws
... Moving across the periodic table from left to right, one finds, in order, metals, then gases, then nonmetals. ...
... Moving across the periodic table from left to right, one finds, in order, metals, then gases, then nonmetals. ...
Name Date Class Period ______
... Name ______________________________________ Date __________________ Class Period _________ Atoms, Elements, and Compound Test Study Guide I. ...
... Name ______________________________________ Date __________________ Class Period _________ Atoms, Elements, and Compound Test Study Guide I. ...