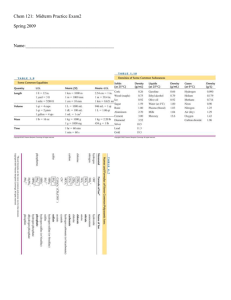
AP Chemistry Review Packet 1 CO2(g) + H2(g) « H2O(g) + CO(g
... constant for the reaction. (c) Determine Kp in terms of Kc for this system. (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to CO2(g). Calculate the value of Kc at this lower temperature. (e) In a different experiment, 0.50 mole of H2(g) ...
... constant for the reaction. (c) Determine Kp in terms of Kc for this system. (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to CO2(g). Calculate the value of Kc at this lower temperature. (e) In a different experiment, 0.50 mole of H2(g) ...
Es 241 P and Chem Pot
... And define the enthalpy of vaporization ~ the energy in the molecular bonds of the liquid ...
... And define the enthalpy of vaporization ~ the energy in the molecular bonds of the liquid ...
Unit 4: Physical Properties and Changes
... Metal – an element that is typically a hard, shiny solid, is malleable, and is a good conductor of heat and electricity Metalloid – an element that shares some properties of metals and some of non-metals Non-metal – an element that is usually a gas or brittle solid at room temperature, is not mallea ...
... Metal – an element that is typically a hard, shiny solid, is malleable, and is a good conductor of heat and electricity Metalloid – an element that shares some properties of metals and some of non-metals Non-metal – an element that is usually a gas or brittle solid at room temperature, is not mallea ...
practice test2
... Which of the following molecules can form hydrogen bonds A) CH4 B) NaH C) NH3 D) BH3 ...
... Which of the following molecules can form hydrogen bonds A) CH4 B) NaH C) NH3 D) BH3 ...
Chapter 13 Gases handout
... Lighter gases diffuse faster than heavier gases. It is not necessary to know individual rates just relative rates ...
... Lighter gases diffuse faster than heavier gases. It is not necessary to know individual rates just relative rates ...
Separation Methods
... type of centrifuge that throws out the liquid by the "centrifugal force" of the rotation. ...
... type of centrifuge that throws out the liquid by the "centrifugal force" of the rotation. ...
Advanced Physical Chemistry Problems (VIII)
... The solution of this equation for p results in a value for the partial pressure of the ammonia. The total pressure will be ptotal = 200 + 2p 9. For the reaction: H2 S(g) + I2 (s) * ) 2HI(g) + S(s,rhombic) Kp is 1.33 × 10−5 atm at 60o C. What will be the mole fraction of HI in the vapor at this tempe ...
... The solution of this equation for p results in a value for the partial pressure of the ammonia. The total pressure will be ptotal = 200 + 2p 9. For the reaction: H2 S(g) + I2 (s) * ) 2HI(g) + S(s,rhombic) Kp is 1.33 × 10−5 atm at 60o C. What will be the mole fraction of HI in the vapor at this tempe ...
Presentation
... Critical temperature—the temp. above which a gas cannot be liquefied at any pressure Critical pressure—the pressure required to liquefy the gas at critical temperature ...
... Critical temperature—the temp. above which a gas cannot be liquefied at any pressure Critical pressure—the pressure required to liquefy the gas at critical temperature ...
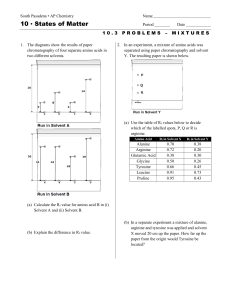
10 States of Matter
... acetone (C3H6O) at 40°C? Assume urea is nonvolatile, and the vapor pressure of pure acetone is 400. torr at 40°C. ...
... acetone (C3H6O) at 40°C? Assume urea is nonvolatile, and the vapor pressure of pure acetone is 400. torr at 40°C. ...
Unit 3: States of Matter Review
... 7. Which type of solid is likely to have the lowest melting point—an ionic solid or a molecular solid? Explain. 8. The normal boiling point of ethanol is 78.5°C. The normal boiling point of water is 100°C. At 75°C, which liquid, ethanol or water, has the greater vapor pressure? Explain. 9. The heigh ...
... 7. Which type of solid is likely to have the lowest melting point—an ionic solid or a molecular solid? Explain. 8. The normal boiling point of ethanol is 78.5°C. The normal boiling point of water is 100°C. At 75°C, which liquid, ethanol or water, has the greater vapor pressure? Explain. 9. The heigh ...