IN-BEAM SPECTROSCOPY OF EXTREMELY NEUTRON
... to unravel the intrinsic structure of these nuclei is described, and the results are compared with theoretical models. The nucleus can be regarded as being made of neutrons and protons interacting predominantly via the strong force. However, the forces between the nucleons in a nucleus are not due t ...
... to unravel the intrinsic structure of these nuclei is described, and the results are compared with theoretical models. The nucleus can be regarded as being made of neutrons and protons interacting predominantly via the strong force. However, the forces between the nucleons in a nucleus are not due t ...
1 Structure of Atom - Viva Online Learning
... 1. All atoms of a particular element are identical in _____________ and _____________. 2. Cathode rays are produced from the _____________ in a discharge tube. 3. The anode rays consist of the positively-charged particles called _____________. 4. The charge to mass ratio (e/m) of proton is fixed. (T ...
... 1. All atoms of a particular element are identical in _____________ and _____________. 2. Cathode rays are produced from the _____________ in a discharge tube. 3. The anode rays consist of the positively-charged particles called _____________. 4. The charge to mass ratio (e/m) of proton is fixed. (T ...
Introduction to PHY008: Atomic and Nuclear Physics
... Around the same time J.J. Thomson was investigating a long-standing puzzle known as "cathode rays." If the low pressure gas in the glass tube was replaced by a total vacuum then the glow disappeared. However, where the positive high voltage electrode passed through the glass a fluorescent glow was s ...
... Around the same time J.J. Thomson was investigating a long-standing puzzle known as "cathode rays." If the low pressure gas in the glass tube was replaced by a total vacuum then the glow disappeared. However, where the positive high voltage electrode passed through the glass a fluorescent glow was s ...
7.2 Powerpoint
... Topic 7: Atomic, nuclear and particle physics 7.2 – Nuclear reactions Mass defect and nuclear binding energy The binding energy Eb of a nucleus is the amount of work or energy that must be expended to pull it apart. EXAMPLE: What is the binding energy Eb in 4He in the nuclear reaction 2(1H) + 2n ...
... Topic 7: Atomic, nuclear and particle physics 7.2 – Nuclear reactions Mass defect and nuclear binding energy The binding energy Eb of a nucleus is the amount of work or energy that must be expended to pull it apart. EXAMPLE: What is the binding energy Eb in 4He in the nuclear reaction 2(1H) + 2n ...
Atomic Mass
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
Physics, Chapter 45: Natural Radioactivity
... This value can be used as an indication of the age of the earth. From such measurements the age of the earth is known to lie between 3 and 6 billion years. In our discussion of radioactive decay, it was tacitly assumed that a given isotope will have only one mode of disintegration. However, a glance ...
... This value can be used as an indication of the age of the earth. From such measurements the age of the earth is known to lie between 3 and 6 billion years. In our discussion of radioactive decay, it was tacitly assumed that a given isotope will have only one mode of disintegration. However, a glance ...
4.1 Introduction to Atoms
... another particle; the neutron. • This new particle was hard to discover because it had no charge, thus was named the neutron! • The new atomic model explains that at the center of the atom is a tiny, massive nucleus containing protons and neutrons. Surrounding the nucleus is a cloudlike region of mo ...
... another particle; the neutron. • This new particle was hard to discover because it had no charge, thus was named the neutron! • The new atomic model explains that at the center of the atom is a tiny, massive nucleus containing protons and neutrons. Surrounding the nucleus is a cloudlike region of mo ...
Chapter 3—Time and Geology
... 4. The concept of half-life is necessary because it represents a convenient ratio (50:50) of parent and daughter. Furthermore, half-life of most radiogenic elements studied is a convenient duration (several 10s of millions of years) regarding geologic processes. Carbon14 (half-life = 5,730 year) is ...
... 4. The concept of half-life is necessary because it represents a convenient ratio (50:50) of parent and daughter. Furthermore, half-life of most radiogenic elements studied is a convenient duration (several 10s of millions of years) regarding geologic processes. Carbon14 (half-life = 5,730 year) is ...
Your Turn
... 1. Elements are composed of minute, indivisible particles called atoms. – Atoms are made up of smaller particles 2. Atoms of the same element are alike in mass and size. – Isotopes of elements exist 3. Atoms of different elements have different masses and sizes. – Isotopes like C-14 and N-14 make th ...
... 1. Elements are composed of minute, indivisible particles called atoms. – Atoms are made up of smaller particles 2. Atoms of the same element are alike in mass and size. – Isotopes of elements exist 3. Atoms of different elements have different masses and sizes. – Isotopes like C-14 and N-14 make th ...
ch05.ppt - James Goodwin
... 1. Elements are composed of minute, indivisible particles called atoms. – Atoms are made up of smaller particles 2. Atoms of the same element are alike in mass and size. – Isotopes of elements exist 3. Atoms of different elements have different masses and sizes. – Isotopes like C-14 and N-14 make th ...
... 1. Elements are composed of minute, indivisible particles called atoms. – Atoms are made up of smaller particles 2. Atoms of the same element are alike in mass and size. – Isotopes of elements exist 3. Atoms of different elements have different masses and sizes. – Isotopes like C-14 and N-14 make th ...
Chapter 18 - Powell County Schools
... and thinkers. What some of them discovered along the way changed the world, influencing not only theoretical spheres such as many of the sciences, philosophy, logic, and other areas, but also those subjects’ practical applications. So many new technological developments of the late 20th century have ...
... and thinkers. What some of them discovered along the way changed the world, influencing not only theoretical spheres such as many of the sciences, philosophy, logic, and other areas, but also those subjects’ practical applications. So many new technological developments of the late 20th century have ...
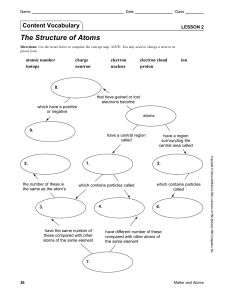
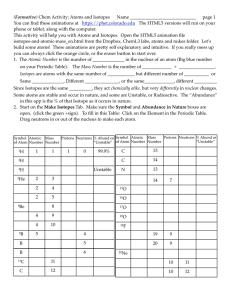
Lesson 2 | The Structure of Atoms
... Directions: Complete the chart with the correct terms or numbers from the word bank on the lines provided. Some terms or numbers may be used more than once or not at all. ...
... Directions: Complete the chart with the correct terms or numbers from the word bank on the lines provided. Some terms or numbers may be used more than once or not at all. ...
Protons for Breakfast
... pullover. The force would be very similar to that on the sausage, but because you are so much heavier, you would accelerate slower. However you would eventually swing around! The largest thing I have accelerated in this way was a guitar weighing around 10 kg. By week 6 will I be able to converse wit ...
... pullover. The force would be very similar to that on the sausage, but because you are so much heavier, you would accelerate slower. However you would eventually swing around! The largest thing I have accelerated in this way was a guitar weighing around 10 kg. By week 6 will I be able to converse wit ...
Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 16
... 11. (a) The strong nuclear force is the stronger of the two forces, by a factor of about 100 but, unlike the electrostatic force, it acts only over very short distances—a few femtometres at most—and acts on both protons and neutrons. The range of the electromagnetic force is infinite, but it acts on ...
... 11. (a) The strong nuclear force is the stronger of the two forces, by a factor of about 100 but, unlike the electrostatic force, it acts only over very short distances—a few femtometres at most—and acts on both protons and neutrons. The range of the electromagnetic force is infinite, but it acts on ...
Deuterium Nucleus Confirms Proton Radius Puzzle
... kind of change in the oscillation mode requires not only parity change, but also charge and time changes (CPT symmetry) resulting a right handed anti-neutrino or a left handed neutrino. The right handed anti-neutrino and the left handed neutrino exist only because changing back the quark flavor coul ...
... kind of change in the oscillation mode requires not only parity change, but also charge and time changes (CPT symmetry) resulting a right handed anti-neutrino or a left handed neutrino. The right handed anti-neutrino and the left handed neutrino exist only because changing back the quark flavor coul ...
Bellringer Chapter 11 Objectives Chapter 11 How Small Is an Atom
... atoms is the atomic mass unit (amu). Each proton and neutron has a mass of about 1 amu. • Compared with protons and neutrons, electrons are very small in mass. It takes more than 1,800 electrons to equal the mass of 1 proton. ...
... atoms is the atomic mass unit (amu). Each proton and neutron has a mass of about 1 amu. • Compared with protons and neutrons, electrons are very small in mass. It takes more than 1,800 electrons to equal the mass of 1 proton. ...
Science Power Guide
... All other energy levels are multiples of this fundamental quantum German scientist Max Planck first proposed energy quantization in 1900 ...
... All other energy levels are multiples of this fundamental quantum German scientist Max Planck first proposed energy quantization in 1900 ...
1 Introduction to Atoms
... smaller particles called protons, neutrons, and electrons. Understanding the structure of atoms will help you understand the properties of matter. Particles in Atoms An atom consists of a nucleus surrounded by one or more electrons. The nucleus (NOO klee us) (plural nuclei) is the very small center ...
... smaller particles called protons, neutrons, and electrons. Understanding the structure of atoms will help you understand the properties of matter. Particles in Atoms An atom consists of a nucleus surrounded by one or more electrons. The nucleus (NOO klee us) (plural nuclei) is the very small center ...
Chapter 14 Inside an Atom
... • The atomic number of an element is the number of protons in the nucleus of an atom of that element. • Atoms of an element are identified by the number of protons because this number never changes without changing the identity of the element. ...
... • The atomic number of an element is the number of protons in the nucleus of an atom of that element. • Atoms of an element are identified by the number of protons because this number never changes without changing the identity of the element. ...
Document
... Learning Objectives: GLE: PS-M-A2: Students will understand that all matter is made up of particles called atoms and that atoms of different elements are different. Students will describe the structure of atoms and the electrical charge of protons, neutrons, and electrons. ...
... Learning Objectives: GLE: PS-M-A2: Students will understand that all matter is made up of particles called atoms and that atoms of different elements are different. Students will describe the structure of atoms and the electrical charge of protons, neutrons, and electrons. ...
Atoms and nukes packet 2016
... open and have your Teacher sign here _________________________ 7. Nicholas Cage thinks the smaller human skull dates from the Revolutionary War. Is this possible? Why or why not? ...
... open and have your Teacher sign here _________________________ 7. Nicholas Cage thinks the smaller human skull dates from the Revolutionary War. Is this possible? Why or why not? ...
Lab 77 Nuclear Radiation Detection
... Isotopes of certain elements are unstable and therefore are in a process of decay. As they decay they emit unseen radiations. This phenomenon is called radioactivity, and such isotopes are called radioisotopes. All elements with atomic numbers greater than 82 (and some with smaller atomic numbers) p ...
... Isotopes of certain elements are unstable and therefore are in a process of decay. As they decay they emit unseen radiations. This phenomenon is called radioactivity, and such isotopes are called radioisotopes. All elements with atomic numbers greater than 82 (and some with smaller atomic numbers) p ...
2gravity a new concept
... e.g. Proton U – D – U to Neutron D – U – D to Proton U – D – U etc,. It is notable that the Proton always forms the atom base as it is the only atomic component with the capacity to generate positive waves that will capture and maintain an Electron in one of the two trajectories. In a matrix of atom ...
... e.g. Proton U – D – U to Neutron D – U – D to Proton U – D – U etc,. It is notable that the Proton always forms the atom base as it is the only atomic component with the capacity to generate positive waves that will capture and maintain an Electron in one of the two trajectories. In a matrix of atom ...