Chapter 2 – Atoms, Ions, and the Periodic Table
... The atomic number of an atom is equal to the number of protons. If you know the name of the element, you can find the atomic number by finding the element on the periodic table. For example, for iron (Fe), you can find the atomic number, 26, listed with the element symbol on the fourth period of the ...
... The atomic number of an atom is equal to the number of protons. If you know the name of the element, you can find the atomic number by finding the element on the periodic table. For example, for iron (Fe), you can find the atomic number, 26, listed with the element symbol on the fourth period of the ...
Theoretical Nuclear Physics
... 11.5.1 Thomas expression for the width . . . . . . . . . 11.6 Homework problems . . . . . . . . . . . . . . . . . . . . ...
... 11.5.1 Thomas expression for the width . . . . . . . . . 11.6 Homework problems . . . . . . . . . . . . . . . . . . . . ...
Wednesday, Oct. 5, 2016
... Weak Interactions • The transition probability per unit time, the width, can be calculated from perturbation theory using Fermi’s ...
... Weak Interactions • The transition probability per unit time, the width, can be calculated from perturbation theory using Fermi’s ...
Chapter 4 and 5
... Demo mole amounts of various elements? Note how they are NOT the same amount!! ...
... Demo mole amounts of various elements? Note how they are NOT the same amount!! ...
Teacher Materials - Scope, Sequence, and Coordination
... particles into energy, and they can release much greater amounts of energy than atomic interactions. Fission is the splitting of a large nucleus into smaller pieces. Fusion is the joining of two nuclei at extremely high temperature and pressure, and is the process responsible for the energy of the s ...
... particles into energy, and they can release much greater amounts of energy than atomic interactions. Fission is the splitting of a large nucleus into smaller pieces. Fusion is the joining of two nuclei at extremely high temperature and pressure, and is the process responsible for the energy of the s ...
4 Radioactive Elements
... radioactive decay, the atomic nuclei of unstable isotopes release fast-moving particles and energy. Discovery of Radioactivity In 1896, the French scientist Henri Becquerel discovered the effects of radioactive decay quite by accident while studying a mineral containing uranium. He observed that wit ...
... radioactive decay, the atomic nuclei of unstable isotopes release fast-moving particles and energy. Discovery of Radioactivity In 1896, the French scientist Henri Becquerel discovered the effects of radioactive decay quite by accident while studying a mineral containing uranium. He observed that wit ...
C 4 The Atomic Theory
... So how could the Greek philosophers have known that Democritus had a good idea with his theory of atomos? The best way would have been to take some careful observation and conduct a few experiments. Recall, however, that the early Greek philosophers tried to understand the nature of the world throug ...
... So how could the Greek philosophers have known that Democritus had a good idea with his theory of atomos? The best way would have been to take some careful observation and conduct a few experiments. Recall, however, that the early Greek philosophers tried to understand the nature of the world throug ...
atoms - WordPress.com
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
rutherford-bohr atomic model untenable in nuclear reactions
... If you think about it, Hydrogen at 1.00794 is more than 1/12 of the weight of carbon-12 (as you can see from the above table, if you multiply 12 times the mass of a single hydrogen atom it comes to more than 12); the reason for this effect is nuclear binding energy. After all, the protons in the nuc ...
... If you think about it, Hydrogen at 1.00794 is more than 1/12 of the weight of carbon-12 (as you can see from the above table, if you multiply 12 times the mass of a single hydrogen atom it comes to more than 12); the reason for this effect is nuclear binding energy. After all, the protons in the nuc ...
Precise Measurement of the Neutron Beta Decay Parameters “a
... allow us to examine the spectrometer response function and verify that the fields have been measured correctly. The main requirements on a suitable magnetic spectrometer are: 1. The spectrometer and its magnetic and electrical fields must be azimuthally symmetric about the central axis, z. 2. Neutro ...
... allow us to examine the spectrometer response function and verify that the fields have been measured correctly. The main requirements on a suitable magnetic spectrometer are: 1. The spectrometer and its magnetic and electrical fields must be azimuthally symmetric about the central axis, z. 2. Neutro ...
The Atomic Theory
... really wanted was a theory that explained all three laws with a simple, plausible model. One way to explain the relationships that Dalton and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called ‘‘atom ...
... really wanted was a theory that explained all three laws with a simple, plausible model. One way to explain the relationships that Dalton and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called ‘‘atom ...
Introductory Chemistry, 2nd Edition Nivaldo Tro - Tutor
... Atomic Structures of Ions Metals form cations For each positive charge the ion has 1 less electron than the neutral atom – Na atom = 11 p+ and 11 e-, Na+ ion = 11 p+ and 10 e– Ca atom = 20 p+ and 20 e-, Ca2+ ion = 20 p+ and 18 e- ...
... Atomic Structures of Ions Metals form cations For each positive charge the ion has 1 less electron than the neutral atom – Na atom = 11 p+ and 11 e-, Na+ ion = 11 p+ and 10 e– Ca atom = 20 p+ and 20 e-, Ca2+ ion = 20 p+ and 18 e- ...
First direct mass measurements on nobelium and lawrencium with
... manmade species of this kind were produced by successive neutron capture and subsequent β − decay starting from the uranium isotope 238 U. This chain of production was, however, interrupted. At this time it was not found obvious that spontaneous fission was the limiting factor. From 1940 until 1955 ...
... manmade species of this kind were produced by successive neutron capture and subsequent β − decay starting from the uranium isotope 238 U. This chain of production was, however, interrupted. At this time it was not found obvious that spontaneous fission was the limiting factor. From 1940 until 1955 ...
1411 Practice Exam 1
... (3 pts) A set of measurements has good precision if a) the average is close to the true value b) the measurements are in close agreement with each other but are not necessarily correct c) a large number of measurements are made d) a small number of measurements are made ...
... (3 pts) A set of measurements has good precision if a) the average is close to the true value b) the measurements are in close agreement with each other but are not necessarily correct c) a large number of measurements are made d) a small number of measurements are made ...
Chemistry 1411 Practice Exam 1, Chapters 1
... (3 pts) A set of measurements has good precision if a) the average is close to the true value b) the measurements are in close agreement with each other but are not necessarily correct c) a large number of measurements are made d) a small number of measurements are made ...
... (3 pts) A set of measurements has good precision if a) the average is close to the true value b) the measurements are in close agreement with each other but are not necessarily correct c) a large number of measurements are made d) a small number of measurements are made ...
Shedding Light on Atoms Episode 5: Protons, Neutrons, and Electrons
... However, atoms can often lose or gain electrons when they chemically react. We’ll have a look at how and when this happens in a later episode. In previous episodes, we were illustrating atoms as solid spheres, but it turns out that they’re not really solid at all. They’re mostly empty space. However ...
... However, atoms can often lose or gain electrons when they chemically react. We’ll have a look at how and when this happens in a later episode. In previous episodes, we were illustrating atoms as solid spheres, but it turns out that they’re not really solid at all. They’re mostly empty space. However ...
Introduction to NMR spectroscopy Nuclei of isotopes which possess
... Nuclei of isotopes which possess an odd number of protons, an odd number of neutrons, or both, have a nuclear spin quantum number, I, such that, I = 1/2n, where n is an integer 0,1,2,3...etc. Since atoms have charge, a spinning nucleus generates a small electric current which in turn creates a finit ...
... Nuclei of isotopes which possess an odd number of protons, an odd number of neutrons, or both, have a nuclear spin quantum number, I, such that, I = 1/2n, where n is an integer 0,1,2,3...etc. Since atoms have charge, a spinning nucleus generates a small electric current which in turn creates a finit ...
Name - Films On Demand
... from atoms of other elements. Atoms may stick together in well-defined molecules or may be packed together in large arrays. Different arrangements of atoms into groups compose all substances. • Scientific ideas about elements were borrowed from some Greek philosophers of 2,000 years earlier, who bel ...
... from atoms of other elements. Atoms may stick together in well-defined molecules or may be packed together in large arrays. Different arrangements of atoms into groups compose all substances. • Scientific ideas about elements were borrowed from some Greek philosophers of 2,000 years earlier, who bel ...
Modern
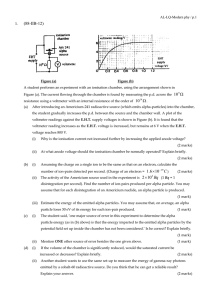
... Describe the energy change of an electron when, after emitting from the hot cathode C, it accelerates towards the grid G and undergoes an inelastic collision with a xenon atom, and finally reaches anode A. (3 marks) ...
... Describe the energy change of an electron when, after emitting from the hot cathode C, it accelerates towards the grid G and undergoes an inelastic collision with a xenon atom, and finally reaches anode A. (3 marks) ...
C5-Early-Atomic-Theory-and-Structure-Comp
... John Wiley & Sons, Inc Morris Hein and Susan Arena ...
... John Wiley & Sons, Inc Morris Hein and Susan Arena ...
Teacher`s Guide Middle School
... from atoms of other elements. Atoms may stick together in well-defined molecules or may be packed together in large arrays. Different arrangements of atoms into groups compose all substances. • Scientific ideas about elements were borrowed from some Greek philosophers of 2,000 years earlier, who bel ...
... from atoms of other elements. Atoms may stick together in well-defined molecules or may be packed together in large arrays. Different arrangements of atoms into groups compose all substances. • Scientific ideas about elements were borrowed from some Greek philosophers of 2,000 years earlier, who bel ...
Atoms and their structure
... 1. The neutral particle found within an atom is the ______________________________. 2. The sum of the protons and neutrons in the nucleus of an atom is called the ______________________________ of that atom. 3. The space around the nucleus of an atom where the atom's electrons are found is called th ...
... 1. The neutral particle found within an atom is the ______________________________. 2. The sum of the protons and neutrons in the nucleus of an atom is called the ______________________________ of that atom. 3. The space around the nucleus of an atom where the atom's electrons are found is called th ...
PDF format
... Figure 1 shows data from a typical NOAA satellite pass during the recovery phase of a storm in March 1998. Protons with angles 10° (solid line) and 80° (dashed line) to the local vertical are each measured in three energy channels 30-80, 80-250 and 250-800 keV. Both the 10° and 80° protons have a we ...
... Figure 1 shows data from a typical NOAA satellite pass during the recovery phase of a storm in March 1998. Protons with angles 10° (solid line) and 80° (dashed line) to the local vertical are each measured in three energy channels 30-80, 80-250 and 250-800 keV. Both the 10° and 80° protons have a we ...
- Cypress HS
... • Note that the initial amount may be in units of mass or number of particles. • A more versatile form of the equation can be written if the exponent n is replaced by the equivalent quantity t/T, where t is the elapsed time and T is the duration of the half-life. ...
... • Note that the initial amount may be in units of mass or number of particles. • A more versatile form of the equation can be written if the exponent n is replaced by the equivalent quantity t/T, where t is the elapsed time and T is the duration of the half-life. ...
Unit 2 Lesson 3
... determined? • Radioactive isotopes, also called radioisotopes, are isotopes that are unstable and break down into other, stable isotopes by a process called radioactive decay. As they break down, they release excess energy by emitting radiation in the form of alpha, beta and gamma rays. • The radioa ...
... determined? • Radioactive isotopes, also called radioisotopes, are isotopes that are unstable and break down into other, stable isotopes by a process called radioactive decay. As they break down, they release excess energy by emitting radiation in the form of alpha, beta and gamma rays. • The radioa ...