What`s Inside an Element
... Teacher will explain to students that today they are going to be creating a model of “what’s inside an element.” Teacher will show students pictures or models of an atom. Students will identify the protons and neutrons found in the nucleus and the electrons orbiting the nucleus. Explain that scienti ...
... Teacher will explain to students that today they are going to be creating a model of “what’s inside an element.” Teacher will show students pictures or models of an atom. Students will identify the protons and neutrons found in the nucleus and the electrons orbiting the nucleus. Explain that scienti ...
4 ATOMIC STRUCTURE NOTES __ /__ pts
... ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in an atom of that element. ________ 13. An atom of nitrogen has 7 protons and 7 neutrons. ________ 14. Relative a ...
... ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in an atom of that element. ________ 13. An atom of nitrogen has 7 protons and 7 neutrons. ________ 14. Relative a ...
Introduction to Nuclear Forces
... must absorb the meson it has emitted within a very short time, but then it can emit another one. Similarly, the meson field surrounding an isolated neutron should sometimes contain a ! 0 meson and sometimes a " ! - meson. But the proton files cannot contain a " ! - meson and the neutron field cannot ...
... must absorb the meson it has emitted within a very short time, but then it can emit another one. Similarly, the meson field surrounding an isolated neutron should sometimes contain a ! 0 meson and sometimes a " ! - meson. But the proton files cannot contain a " ! - meson and the neutron field cannot ...
Chapter 4 - The Structure of the Atom Atomic Models PIONEERS OF
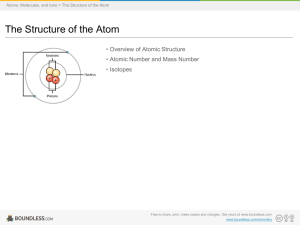
... o Composed of electrons, protons, and neutrons The protons and the neutrons make up the center of the atom. o Center of the atom is called the nucleus. The protons and neutrons occupy the mass of the atom. The electrons surround the nucleus and occupy most of the atom’s volume How, then, are atoms o ...
... o Composed of electrons, protons, and neutrons The protons and the neutrons make up the center of the atom. o Center of the atom is called the nucleus. The protons and neutrons occupy the mass of the atom. The electrons surround the nucleus and occupy most of the atom’s volume How, then, are atoms o ...
Textbook Unit 4 Review Solutions
... half-life: the time it takes for half of the radioactive nuclei in a sample to decay ionization energy: the energy required to remove an electron from an atom isotopes: atoms that have the same number of protons, but different numbers of neutrons lepton: a subatomic particle that does not interact v ...
... half-life: the time it takes for half of the radioactive nuclei in a sample to decay ionization energy: the energy required to remove an electron from an atom isotopes: atoms that have the same number of protons, but different numbers of neutrons lepton: a subatomic particle that does not interact v ...
Module 6 Chemical Reactions
... 2.Count the number of atoms of each element on each side 3.See what elements don’t match in your T-chart 4.Add coefficients in front of one substance at a time, •Start by balancing the most complex compound first, •general rule of thumb is to save Hydrogen and Oxygen until the end to balance. 5.reco ...
... 2.Count the number of atoms of each element on each side 3.See what elements don’t match in your T-chart 4.Add coefficients in front of one substance at a time, •Start by balancing the most complex compound first, •general rule of thumb is to save Hydrogen and Oxygen until the end to balance. 5.reco ...
Nickel-Hydrogen Cold Fusion by Intermediate Rydberg State of
... mechanical (QM) models of atoms are based on the Rutherford-Bohr planetary model of hydrogen. The nuclear size is determined by scattering experiments, while the nuclear shape for all atoms is assumed spherical and solid (without geometrical shape and structure). Using this assumption Rutherford pro ...
... mechanical (QM) models of atoms are based on the Rutherford-Bohr planetary model of hydrogen. The nuclear size is determined by scattering experiments, while the nuclear shape for all atoms is assumed spherical and solid (without geometrical shape and structure). Using this assumption Rutherford pro ...
Atomic Structure_Bohr Flashcards
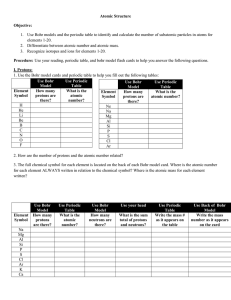
... 1. Use the Bohr model cards and periodic table to help you fill out the following tables: Use Bohr Use Periodic Use Bohr Use Periodic Model Table Model Table ...
... 1. Use the Bohr model cards and periodic table to help you fill out the following tables: Use Bohr Use Periodic Use Bohr Use Periodic Model Table Model Table ...
Symbols of Elements
... negatively charged particles called electrons • proposed the “plum pudding” model of an atom in which protons and electrons were distributed throughout the ...
... negatively charged particles called electrons • proposed the “plum pudding” model of an atom in which protons and electrons were distributed throughout the ...
Boundless Study Slides
... • Each electron has a negative charge equal to the positive charge of a proton. • Neutrons are uncharged particles found within the nucleus. ...
... • Each electron has a negative charge equal to the positive charge of a proton. • Neutrons are uncharged particles found within the nucleus. ...
atomic regents review
... One model of the atom states that atoms are tiny particles composed of a uniform mixture of positive and negative charges. Scientists conducted an experiment where alpha particles were aimed at a thin layer of gold atoms. Most of the alpha particles passed directly through the gold atoms. A few alph ...
... One model of the atom states that atoms are tiny particles composed of a uniform mixture of positive and negative charges. Scientists conducted an experiment where alpha particles were aimed at a thin layer of gold atoms. Most of the alpha particles passed directly through the gold atoms. A few alph ...
Atoms - Chemistry R: 4(AE)
... •Electrons revolve around the nucleus in specific orbits, or energy levels. • An atom has energy levels. Electrons can only exist in these energy levels, not in between. •When an atom is in the ground state, the electrons exist in the energy levels closest to the nucleus. •GROUND STATE: the lowest e ...
... •Electrons revolve around the nucleus in specific orbits, or energy levels. • An atom has energy levels. Electrons can only exist in these energy levels, not in between. •When an atom is in the ground state, the electrons exist in the energy levels closest to the nucleus. •GROUND STATE: the lowest e ...
Lecture 1.
... emits proton(s) to get energetically more stable position. Above of the proton drop-off line the nuclei are not stable, but they emit protons immediately. ...
... emits proton(s) to get energetically more stable position. Above of the proton drop-off line the nuclei are not stable, but they emit protons immediately. ...
Chapter 2
... Determine (a) the number of moles of C in 25.00 g of carbon, (b) the number of moles of He in 10.50 g of helium, and (c) the number of moles of Na in 15.75 g of sodium. Strategy Molar mass of an element is numerically equal to its average atomic mass. Use the molar mass for each element to convert f ...
... Determine (a) the number of moles of C in 25.00 g of carbon, (b) the number of moles of He in 10.50 g of helium, and (c) the number of moles of Na in 15.75 g of sodium. Strategy Molar mass of an element is numerically equal to its average atomic mass. Use the molar mass for each element to convert f ...
atom
... Determine (a) the number of C atoms in 0.515 g of carbon, and (b) the mass of helium that contains 6.89×1018 He atoms. Strategy Use the conversions depicted in the previous slide to convert (a) from grams to moles to atoms and (b) from atoms to moles to grams. Think About It A ballpark estimate can ...
... Determine (a) the number of C atoms in 0.515 g of carbon, and (b) the mass of helium that contains 6.89×1018 He atoms. Strategy Use the conversions depicted in the previous slide to convert (a) from grams to moles to atoms and (b) from atoms to moles to grams. Think About It A ballpark estimate can ...
Beta Decay
... magnetic field. You should tackle one of these three and show quantitatively how it effect on the counts vs. energy plot for Cs (why do we choose Cs?). To help with the analysis, Yang-Hui He (’96) has written a C program. The program may be obtained from L. Page. If you use the program, you are resp ...
... magnetic field. You should tackle one of these three and show quantitatively how it effect on the counts vs. energy plot for Cs (why do we choose Cs?). To help with the analysis, Yang-Hui He (’96) has written a C program. The program may be obtained from L. Page. If you use the program, you are resp ...
Topic 7_2__Radioactive decay
... was caused by some new type of radiation being emitted by the uranium compound. This radiation had sufficient energy to pass through the cardboard storage box and the glass of the ...
... was caused by some new type of radiation being emitted by the uranium compound. This radiation had sufficient energy to pass through the cardboard storage box and the glass of the ...
Physics 2
... A small rocket is launched. At a certain point in the flight, the rocket’s mass is 82kg, and is travelling at a velocity of 30m/s. 10 seconds later, the mass of the rocket has reduced to 72kg, and its velocity has increased to 65 m/s. Calculate the (average) resultant force on the rocket during this ...
... A small rocket is launched. At a certain point in the flight, the rocket’s mass is 82kg, and is travelling at a velocity of 30m/s. 10 seconds later, the mass of the rocket has reduced to 72kg, and its velocity has increased to 65 m/s. Calculate the (average) resultant force on the rocket during this ...
Princeton University, Physics 311/312 Beta Decay, Page 1 BETA
... • What is the magnetic field inside the spectrometer when the current is near 10 amps? • Why shouldn’t you open the knife switch when current is flowing? • Is the Fermi-Kurie plot useful for the Cs decay? • Why is the instrument response to the K-conversion electrons broad? What is that telling you ...
... • What is the magnetic field inside the spectrometer when the current is near 10 amps? • Why shouldn’t you open the knife switch when current is flowing? • Is the Fermi-Kurie plot useful for the Cs decay? • Why is the instrument response to the K-conversion electrons broad? What is that telling you ...
The Atom
... • In 1897, J. J. Thomson performed experiments that detected smaller particles within atoms. • In the early 1900s, Ernest Rutherford and James Chadwick revealed the nature of the dense center of an atom. ...
... • In 1897, J. J. Thomson performed experiments that detected smaller particles within atoms. • In the early 1900s, Ernest Rutherford and James Chadwick revealed the nature of the dense center of an atom. ...
Atoms: The Building Blocks of Matter
... •Electrons revolve around the nucleus in specific orbits, or energy levels. • An atom has energy levels. Electrons can only exist in these energy levels, not in between. •When an atom is in the ground state, the electrons exist in the energy levels closest to the nucleus. •GROUND STATE: the lowest e ...
... •Electrons revolve around the nucleus in specific orbits, or energy levels. • An atom has energy levels. Electrons can only exist in these energy levels, not in between. •When an atom is in the ground state, the electrons exist in the energy levels closest to the nucleus. •GROUND STATE: the lowest e ...
6.7 Explaining the Periodic Table
... 126 neutrons. Such a high number of protons and neutrons makes its nucleus very crowded and therefore unstable. Litvinenko ate polonium nuclei, which released energy and particles that were absorbed into his tissues. This polonium radiation caused extensive tissue damage that killed Litvinenko. ...
... 126 neutrons. Such a high number of protons and neutrons makes its nucleus very crowded and therefore unstable. Litvinenko ate polonium nuclei, which released energy and particles that were absorbed into his tissues. This polonium radiation caused extensive tissue damage that killed Litvinenko. ...
1 What is the frequency, in Hz, of electromagnetic
... 35 Of the three types of radioactivity characterized by Rutherford, which is/are not particles? A ...
... 35 Of the three types of radioactivity characterized by Rutherford, which is/are not particles? A ...