An Unifying Basis for all the Nuclear Reactions Karunakar
... There is no consistency between the physical aspects such as mass, deficit of mass, binding energy and the volume of an object. All of our assumptions and theories based on the current definition of mass appear to be flawed. The most appropriate description of the observations coherently is that the ...
... There is no consistency between the physical aspects such as mass, deficit of mass, binding energy and the volume of an object. All of our assumptions and theories based on the current definition of mass appear to be flawed. The most appropriate description of the observations coherently is that the ...
Reading 1.4 What Are The Parts Of An Atom and How Are They
... Electrons have a negative charge. As a result, they are attracted to positive objects and repelled from negative objects, including other electrons (illustrated below). To minimize repulsion, each electron is capable of staking out a “territory” and “defending” itself from other electrons. Protons a ...
... Electrons have a negative charge. As a result, they are attracted to positive objects and repelled from negative objects, including other electrons (illustrated below). To minimize repulsion, each electron is capable of staking out a “territory” and “defending” itself from other electrons. Protons a ...
Hi Guys. Today we are going to be talking about the smallest part of
... have? Well, the atomic mass tells me how many protons plus neutrons. And I am going to round it, so I am going to say that the atomic mass of hydrogen here is 1 so if I take the atomic mass minus the atomic number, which is the number of protons, I’m going to get the number of neutrons. So this is h ...
... have? Well, the atomic mass tells me how many protons plus neutrons. And I am going to round it, so I am going to say that the atomic mass of hydrogen here is 1 so if I take the atomic mass minus the atomic number, which is the number of protons, I’m going to get the number of neutrons. So this is h ...
Radiocarbon dating
... dating technique which compares two isotopes of uranium (U) in a sample: 235U and 238U.235U/238U dating is one of several radiometric dating techniques exploiting the uranium radioactive decay series, in which 238U undergoes 14alpha and beta decay events while decaying to the stable isotope 206Pb an ...
... dating technique which compares two isotopes of uranium (U) in a sample: 235U and 238U.235U/238U dating is one of several radiometric dating techniques exploiting the uranium radioactive decay series, in which 238U undergoes 14alpha and beta decay events while decaying to the stable isotope 206Pb an ...
Learning about atoms
... see it with our eye. In fact, it is so small that it has been said that more than one million can fit side by side on the tip of your pencil! Parts of an atom The atom is made up of two parts. The center, which is called the nucleus, and the surrounding part, called the electron cloud. You will find ...
... see it with our eye. In fact, it is so small that it has been said that more than one million can fit side by side on the tip of your pencil! Parts of an atom The atom is made up of two parts. The center, which is called the nucleus, and the surrounding part, called the electron cloud. You will find ...
Chapter 5
... Protons and neutrons are located in the nucleus. Electrons are dispersed throughout the remainder of the atom (mainly open space). Neutral atoms contain the same number of protons and neutrons to maintain charge balance. © 2014 John Wiley & Sons, Inc. All rights reserved. ...
... Protons and neutrons are located in the nucleus. Electrons are dispersed throughout the remainder of the atom (mainly open space). Neutral atoms contain the same number of protons and neutrons to maintain charge balance. © 2014 John Wiley & Sons, Inc. All rights reserved. ...
10/2/2013 1 5 Early Atomic Theory and Structure Chapter Outline
... Dalton’s theory of atoms, proposed in the early 1800s, states: 1. Elements are composed of small, indivisible particles called atoms. 2. Atoms of the same element are identical in mass and size. 3. Atoms of different elements differ in their mass and size. 4. Compounds are formed by combining two or ...
... Dalton’s theory of atoms, proposed in the early 1800s, states: 1. Elements are composed of small, indivisible particles called atoms. 2. Atoms of the same element are identical in mass and size. 3. Atoms of different elements differ in their mass and size. 4. Compounds are formed by combining two or ...
The Atoms Family
... explore the world of electrons and quarks that are at least 9,000 times smaller than a nucleus. • We can not see anything smaller than an atom with our eyes, even with the electron microscope. Physicists study much smaller things without seeing them directly. ...
... explore the world of electrons and quarks that are at least 9,000 times smaller than a nucleus. • We can not see anything smaller than an atom with our eyes, even with the electron microscope. Physicists study much smaller things without seeing them directly. ...
- Orangefield ISD
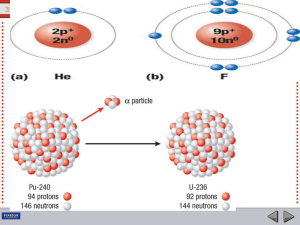
... Isotopes and Mass Number All atoms of a particular element have the same number of protons and electrons but the number of neutrons in the nucleus can differ. Atoms with the same number of protons but different numbers of neutrons are called isotopes. In nature, most elements are found as mixtures ...
... Isotopes and Mass Number All atoms of a particular element have the same number of protons and electrons but the number of neutrons in the nucleus can differ. Atoms with the same number of protons but different numbers of neutrons are called isotopes. In nature, most elements are found as mixtures ...
nuclear physics - Thierry Karsenti
... This module (Nuclear Physics) introduces the basic properties of the atomic nucleus nuclear constituents; the binding energy; isotopes; and nuclear models are concepts dealt in the first activity. Most atoms found in nature are stable and do not emit particles or energy that change form over time. H ...
... This module (Nuclear Physics) introduces the basic properties of the atomic nucleus nuclear constituents; the binding energy; isotopes; and nuclear models are concepts dealt in the first activity. Most atoms found in nature are stable and do not emit particles or energy that change form over time. H ...
Ch 3 Sec 3 Highlighted
... and tells you how many protons it has, and how many electrons it has. If you want to know which element has atomic number 47 look at the periodic table it is silver, Ag. All silver atoms contain 47 protons in their nuclei. Because atoms are neutral, we know from the atomic number that all silver ato ...
... and tells you how many protons it has, and how many electrons it has. If you want to know which element has atomic number 47 look at the periodic table it is silver, Ag. All silver atoms contain 47 protons in their nuclei. Because atoms are neutral, we know from the atomic number that all silver ato ...
Document
... • The periodic table is a list of all of the elements that can build matter. It’s a little like the alphabet of chemistry. • The periodic table tells us ...
... • The periodic table is a list of all of the elements that can build matter. It’s a little like the alphabet of chemistry. • The periodic table tells us ...
IONIZING RADIATION AND RADIONUCLIDS AS THE SOURSES …
... As the atomic mass number increases, the ratio of neutrons to protons increases according to a definite pattern. If isotopes vary from this pattern, they are relatively unstable. The most stable state of a nucleus is called the ‘ground’ state. In an unstable nucleus the nucleons are in an ‘exited’ s ...
... As the atomic mass number increases, the ratio of neutrons to protons increases according to a definite pattern. If isotopes vary from this pattern, they are relatively unstable. The most stable state of a nucleus is called the ‘ground’ state. In an unstable nucleus the nucleons are in an ‘exited’ s ...
13.1 Fundamental Particles and Forces
... too many “Stable” means the nucleus stays together. For complex reasons, the neutrons? nucleus of an atom becomes unstable if it contains too many or too few neutrons relative to the number of protons. If the nucleus is unstable, it breaks apart. Carbon has two stable isotopes, carbon-12 and carbon- ...
... too many “Stable” means the nucleus stays together. For complex reasons, the neutrons? nucleus of an atom becomes unstable if it contains too many or too few neutrons relative to the number of protons. If the nucleus is unstable, it breaks apart. Carbon has two stable isotopes, carbon-12 and carbon- ...
1-4 What Are The Parts Of An Atom and How Are They Arranged
... Thomson thought about his results for a long time. It was almost as if the cathode rays were attracted to the positively charged metal plate and repelled from the negatively charged metal plate. Thomson knew that charged objects are attracted to and repelled from other charged objects according to ...
... Thomson thought about his results for a long time. It was almost as if the cathode rays were attracted to the positively charged metal plate and repelled from the negatively charged metal plate. Thomson knew that charged objects are attracted to and repelled from other charged objects according to ...
Chapter 2 PowerPoint
... Determine (a) the number of C atoms in 0.515 g of carbon, and (b) the mass of helium that contains 6.89×1018 He atoms. Strategy Use the conversions depicted in the previous slide to convert (a) from grams to moles to atoms and (b) from atoms to moles to grams. Think About It A ballpark estimate can ...
... Determine (a) the number of C atoms in 0.515 g of carbon, and (b) the mass of helium that contains 6.89×1018 He atoms. Strategy Use the conversions depicted in the previous slide to convert (a) from grams to moles to atoms and (b) from atoms to moles to grams. Think About It A ballpark estimate can ...
Notes
... elements combine to form 2 or more compounds, the mass of one element that combines with a given mass of the other is in the ratio of small whole numbers ...
... elements combine to form 2 or more compounds, the mass of one element that combines with a given mass of the other is in the ratio of small whole numbers ...
Chapter 21 Nuclear Chemistry
... • Not all atoms of the same element have the same mass due to different numbers of neutrons in those atoms. • There are three naturally occurring isotopes of uranium: Uranium-234 Uranium-235 Uranium-238 ...
... • Not all atoms of the same element have the same mass due to different numbers of neutrons in those atoms. • There are three naturally occurring isotopes of uranium: Uranium-234 Uranium-235 Uranium-238 ...
Full research publication
... monohelat form of 2B also well explained by chain extension interface, in which the electron density on the atoms nitrogen increases, resulting in the screening of the NH protons. The signal value of the two methylene protons monohelatnoy form of 2B shifted to a strong field compared to the same sig ...
... monohelat form of 2B also well explained by chain extension interface, in which the electron density on the atoms nitrogen increases, resulting in the screening of the NH protons. The signal value of the two methylene protons monohelatnoy form of 2B shifted to a strong field compared to the same sig ...
Chapter 25.1 Nuclear Radiation
... Radioactivity: process where materials give off high energy rays Radiation: name give to these penetrating rays and particles Radioisotopes: unstable isotopes that will decay into a different element Alpha particle: high energy helium nuclei containing 2 protons and 2 neutrons beta particle: high en ...
... Radioactivity: process where materials give off high energy rays Radiation: name give to these penetrating rays and particles Radioisotopes: unstable isotopes that will decay into a different element Alpha particle: high energy helium nuclei containing 2 protons and 2 neutrons beta particle: high en ...
Chapter 2 - HCC Learning Web
... Cation is a positively charged ion Anion is a negatively charged ion Ion charge is directly related to the number of electrons lost or gained ...
... Cation is a positively charged ion Anion is a negatively charged ion Ion charge is directly related to the number of electrons lost or gained ...
Theory of the Nuclear Binding Energy
... energy per nucleon about –7.074 MeV. We can see that the theoretical result, –7.069 MeV, is very close to experimental data. 2.4 Nucleus of iron atom According to the extension of supersymmetry in nuclear structure, [5], in atomic nuclei there are groups of nucleons containing following number of pr ...
... energy per nucleon about –7.074 MeV. We can see that the theoretical result, –7.069 MeV, is very close to experimental data. 2.4 Nucleus of iron atom According to the extension of supersymmetry in nuclear structure, [5], in atomic nuclei there are groups of nucleons containing following number of pr ...
DEFINING THE ATOM - BradyMathScience
... ________ 14. The number of neutrons in the nucleus of an atom can be calculated by a. adding together the numbers of electrons and protons. b. subtracting the number of protons from the number of electrons. c. subtracting the number of protons from the mass number. d. adding the mass number to the n ...
... ________ 14. The number of neutrons in the nucleus of an atom can be calculated by a. adding together the numbers of electrons and protons. b. subtracting the number of protons from the number of electrons. c. subtracting the number of protons from the mass number. d. adding the mass number to the n ...
Slide 1
... mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occur for reactions where more mass results. A proton decaying into a neutron can occur only with external energy input. ...
... mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occur for reactions where more mass results. A proton decaying into a neutron can occur only with external energy input. ...