Name Period Nuclear Study Packet Set 1 1. What subatomic
... 3. Potassium-42 has a half life of 12 hours. At present, a given ore sample contains 34.2 mg of K-42. How much did it contain yesterday at the same time. 4. What percent of a sample of a radioactive element whose half life is 5 years will decay after 25 years? 5. What are some ways that nuclea ...
... 3. Potassium-42 has a half life of 12 hours. At present, a given ore sample contains 34.2 mg of K-42. How much did it contain yesterday at the same time. 4. What percent of a sample of a radioactive element whose half life is 5 years will decay after 25 years? 5. What are some ways that nuclea ...
File
... Promising instant curative and beautifying effects, Tho-Radia gained wide popularity in France during the early 1930's as a range of beauty products and perfumes. The face cream was especially popular and contained of 0.5g thorium chloride and 0.25mg radium bromide per 100g. It was even advertised a ...
... Promising instant curative and beautifying effects, Tho-Radia gained wide popularity in France during the early 1930's as a range of beauty products and perfumes. The face cream was especially popular and contained of 0.5g thorium chloride and 0.25mg radium bromide per 100g. It was even advertised a ...
View Transcript
... Okay, so why magic numbers? Why are these numbers appearing over and over again? For instance, a lot of these have more isotopes than they might have expected. In other words, calcium has a lot of isotopes and it’s because there’s special stability associated with 20 protons. Now why? Well, just as ...
... Okay, so why magic numbers? Why are these numbers appearing over and over again? For instance, a lot of these have more isotopes than they might have expected. In other words, calcium has a lot of isotopes and it’s because there’s special stability associated with 20 protons. Now why? Well, just as ...
Another look at chemical reactions HYDROGEN PEROXIDE WATER
... - slightly more massive than the proton ELECTRON - a small particle that carries an overall unit NEGATIVE CHARGE - about 2000 times LESS massive than either protons or neutrons ...
... - slightly more massive than the proton ELECTRON - a small particle that carries an overall unit NEGATIVE CHARGE - about 2000 times LESS massive than either protons or neutrons ...
nuclear fusion 1112
... • The name of an element is The atomic mass is the number with determined by the number the decimal. It is always larger of protons it has. than the atomic number • Atomic # = # Protons The atomic mass is equal to the • 42 protons it is element 42 number of protons + neutrons. and called Molybdenum ...
... • The name of an element is The atomic mass is the number with determined by the number the decimal. It is always larger of protons it has. than the atomic number • Atomic # = # Protons The atomic mass is equal to the • 42 protons it is element 42 number of protons + neutrons. and called Molybdenum ...
Chapter 29: Nuclear Physics
... The absorbed dose of ionizing radiation is the amount of radiation energy absorbed per unit mass of tissue. Ionizing radiation is radiation with enough energy to ionize an atom or molecule. The SI unit of absorbed dose is the Gray. 1 Gy = 1 J/kg. Another common unit is the rad (radiation absorbed do ...
... The absorbed dose of ionizing radiation is the amount of radiation energy absorbed per unit mass of tissue. Ionizing radiation is radiation with enough energy to ionize an atom or molecule. The SI unit of absorbed dose is the Gray. 1 Gy = 1 J/kg. Another common unit is the rad (radiation absorbed do ...
Section 4.2 The Structure of an Atom
... 4. Circle the letter of the expression that accurately compares the masses of neutrons and protons. a. mass of 1 neutron = mass of 1 proton b. mass of 2000 neutrons = mass of 1 proton c. mass of 1 electron = mass of 1 proton Physical Science Reading and Study Workbook Level B ...
... 4. Circle the letter of the expression that accurately compares the masses of neutrons and protons. a. mass of 1 neutron = mass of 1 proton b. mass of 2000 neutrons = mass of 1 proton c. mass of 1 electron = mass of 1 proton Physical Science Reading and Study Workbook Level B ...
Protons neutrons electrons Charge Positive neutral negative Mass
... Atomic mass = weighted average of the naturally occurring isotopes • Atomic mass IS NOT THE SAME AS mass number!!! • Mass numbers are whole numbers, sum of protons and neutrons • Atomic numbers are averages so can be decimals and are found on the periodic table underneath element symbol • Atomic ...
... Atomic mass = weighted average of the naturally occurring isotopes • Atomic mass IS NOT THE SAME AS mass number!!! • Mass numbers are whole numbers, sum of protons and neutrons • Atomic numbers are averages so can be decimals and are found on the periodic table underneath element symbol • Atomic ...
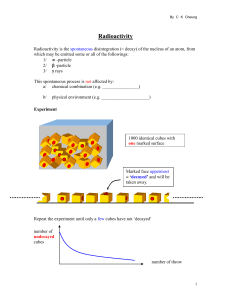
Radioactivity
... Radioactivity is the spontaneous disintegration (= decay) of the nucleus of an atom, from which may be emitted some or all of the followings: 1/ -particle 2/ -particle 3/ rays This spontaneous process is not affected by: a/ chemical combination (e.g. ________________) b/ ...
... Radioactivity is the spontaneous disintegration (= decay) of the nucleus of an atom, from which may be emitted some or all of the followings: 1/ -particle 2/ -particle 3/ rays This spontaneous process is not affected by: a/ chemical combination (e.g. ________________) b/ ...
Nuclear Chem Notes - Warren County Schools
... • Remember, isotopes are: – Atoms with the same atomic number Z, but different neutron numbers N and mass numbers A ...
... • Remember, isotopes are: – Atoms with the same atomic number Z, but different neutron numbers N and mass numbers A ...
Nuclear Chemistry
... • is the process in which an unstable atomic nucleus emits charged particles & energy • Any atom containing an unstable nucleus is called a radioactive isotope or radioisotope ...
... • is the process in which an unstable atomic nucleus emits charged particles & energy • Any atom containing an unstable nucleus is called a radioactive isotope or radioisotope ...
Periodic Table Quiz
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
Homework 1B1 - 3 - Uddingston Grammar School
... 10. In the manufacture of glass, other chemicals can be added to alter the properties of the glass. The element boron can be added to glass to make ovenproof dishes. (a) Information about an atom of boron is given in the table below. ...
... 10. In the manufacture of glass, other chemicals can be added to alter the properties of the glass. The element boron can be added to glass to make ovenproof dishes. (a) Information about an atom of boron is given in the table below. ...
Chapter 19 Radioactive Material An Isotope is an element with a
... uranium atoms split. The hot water turns into steam which then spins a turbine that is hooked u to a generator. To prevent the uranium from creating to much heat that might melt the walls ...
... uranium atoms split. The hot water turns into steam which then spins a turbine that is hooked u to a generator. To prevent the uranium from creating to much heat that might melt the walls ...
(null): 096.AtomReview
... a. Each specific color tells us about the structure of specific electrons inside the atom b. Zinc spectrum s different from ANY other element (compare to sulfur and helium on same slide) E. Atomic structure basics (see AtomOverview.ppt) – what we’ve learned by seeing without seeing … 1. PROTONS: a. ...
... a. Each specific color tells us about the structure of specific electrons inside the atom b. Zinc spectrum s different from ANY other element (compare to sulfur and helium on same slide) E. Atomic structure basics (see AtomOverview.ppt) – what we’ve learned by seeing without seeing … 1. PROTONS: a. ...
The Atom
... • Oil drop experiment determined the charge (e=1.602 x 10 -19 coulomb) and the mass (m = 9.11 x 10 -28 gram) of an electron. ...
... • Oil drop experiment determined the charge (e=1.602 x 10 -19 coulomb) and the mass (m = 9.11 x 10 -28 gram) of an electron. ...
Chapter 25.2 Nuclear Transformations
... More than 1,500 different nuclei are known. Of those, only 264 are stable and do not decay or change with time. These nuclei are in a region called the ...
... More than 1,500 different nuclei are known. Of those, only 264 are stable and do not decay or change with time. These nuclei are in a region called the ...
Atomic Structure
... Electrons in the atom are to be found 90% of the time in 3D regions called orbitals THE BOHR ATOM When an electron transitions from the excited to the ground state, the atom loses energy When an electron transitions from the ground to the excited state, the atom absorbs energy Ground State Electron ...
... Electrons in the atom are to be found 90% of the time in 3D regions called orbitals THE BOHR ATOM When an electron transitions from the excited to the ground state, the atom loses energy When an electron transitions from the ground to the excited state, the atom absorbs energy Ground State Electron ...
Chapter 2 Matter is Made up of Atoms
... Find the number of protons, neutrons, and electrons in Sodium (Na). The mass number for our sodium atom is 25. ...
... Find the number of protons, neutrons, and electrons in Sodium (Na). The mass number for our sodium atom is 25. ...
Nuclear Physics SL - Hockerill Students
... The time taken for the number (or mass) of radioactive nuclei present to fall to half its value. This length of time is constant at any point in time - showing that radioactive decay is exponential. ...
... The time taken for the number (or mass) of radioactive nuclei present to fall to half its value. This length of time is constant at any point in time - showing that radioactive decay is exponential. ...
20161018145157
... Know that atoms of different elements have different numbers of protons. The atomic number tells you the number of what two subatomic particles? o Protons and electrons What is the mass number? o Protons plus neutrons How do you find the number of neutrons? o Atomic mass minus atomic number What are ...
... Know that atoms of different elements have different numbers of protons. The atomic number tells you the number of what two subatomic particles? o Protons and electrons What is the mass number? o Protons plus neutrons How do you find the number of neutrons? o Atomic mass minus atomic number What are ...
Atomic Models
... 1 amu = 1.66 X 10 –27 kg 1 mole 12C = 12X10-3 kg = 6.0228X1023 atoms 12C Avogadro’s number NA = 6.0228 X 10 23 ...
... 1 amu = 1.66 X 10 –27 kg 1 mole 12C = 12X10-3 kg = 6.0228X1023 atoms 12C Avogadro’s number NA = 6.0228 X 10 23 ...
Early Atomic Theory
... • For example: Water and Earth = Mud • Mud might just seem like a nuisance to us, but back then it was a building material. • Fire and Earth made Lava. • If you are curious check out the Little Alchemy app (game) that can be found in Google Apps. It is a fun game. ...
... • For example: Water and Earth = Mud • Mud might just seem like a nuisance to us, but back then it was a building material. • Fire and Earth made Lava. • If you are curious check out the Little Alchemy app (game) that can be found in Google Apps. It is a fun game. ...
Chapter 4: The Structure of the Atom Early Ideas about Matter Name
... 2. Atoms are indivisible and indestructible 3. Atoms of a given element are identical in size, mass, and chemical properties 4. Atoms of a specific element are different from those of another element 5. Different atoms combine in simple whole-number ratios to form compounds 6. In a chemical reaction ...
... 2. Atoms are indivisible and indestructible 3. Atoms of a given element are identical in size, mass, and chemical properties 4. Atoms of a specific element are different from those of another element 5. Different atoms combine in simple whole-number ratios to form compounds 6. In a chemical reaction ...