BellWork 2/16/2015
... Mg2+ ClFe2+ O2These elements are written to describe what? Grab a textbook and look it up if needed. ...
... Mg2+ ClFe2+ O2These elements are written to describe what? Grab a textbook and look it up if needed. ...
The Atom Chapter 2
... Antimony (Sb) has two stable isotopes, 121Sb and 123Sb with masses of 120.9038 u and 122.9042 u, respectively. Calculate the percent abundances of these two isotopes ...
... Antimony (Sb) has two stable isotopes, 121Sb and 123Sb with masses of 120.9038 u and 122.9042 u, respectively. Calculate the percent abundances of these two isotopes ...
Atoms have a structure that determines their properties.
... • The Periodic Table organizes elements in the following way: • Metals are found on the left side, non-metals on the right, and metalloids in between. • Chemical families are arranged in vertical groups. • The table can also display the chemical symbol, atomic number, atomic mass, ion charge, densit ...
... • The Periodic Table organizes elements in the following way: • Metals are found on the left side, non-metals on the right, and metalloids in between. • Chemical families are arranged in vertical groups. • The table can also display the chemical symbol, atomic number, atomic mass, ion charge, densit ...
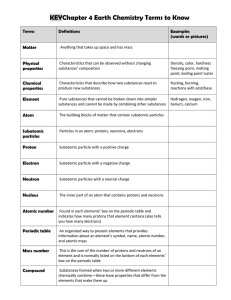
Chapter 4 Atomic Structure Notes
... increasing atomic mass no. 1. The elements fell into 7 columns based on chemical & physical properties 2. He left spaces for undiscovered elements B. Henry Mosely (1913) (British) publishes the “modern” periodic table based on increasing atomic no.. ...
... increasing atomic mass no. 1. The elements fell into 7 columns based on chemical & physical properties 2. He left spaces for undiscovered elements B. Henry Mosely (1913) (British) publishes the “modern” periodic table based on increasing atomic no.. ...
Ch. 5 Outline Notes
... 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________________ Atomic Theory 1. All matter is made of ________________ 2. Atoms are ____________________ ...
... 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________________ Atomic Theory 1. All matter is made of ________________ 2. Atoms are ____________________ ...
The Atom - Taylorsville
... • Helped moved alchemy to be chemistry • Mass cannot be created nor destroyed in normal chemical or physical processes. ...
... • Helped moved alchemy to be chemistry • Mass cannot be created nor destroyed in normal chemical or physical processes. ...
2.9 Use the helium-4 isotope to define atomic number and mass
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
Chapter 3 – Atomic Structure - Mercer Island School District
... – Law of Constant Composition – compounds contain the same elements always in the same proportions. ...
... – Law of Constant Composition – compounds contain the same elements always in the same proportions. ...
Structure of the Atom
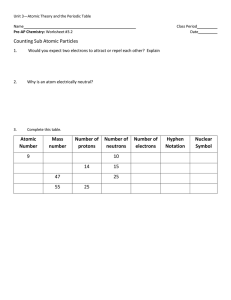
... • The number of protons tells us the atomic number of the atom. • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
... • The number of protons tells us the atomic number of the atom. • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
Atomic Structure and the Periodic Table
... Proton – positively charged particle found in the nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
... Proton – positively charged particle found in the nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
Ions and isotopes
... Quick Review • Atoms are made up of three particles: • Protons • Neutrons • Electrons • Question: Which of the three particles identifies what element an atom is? • The PROTON! (very important) ...
... Quick Review • Atoms are made up of three particles: • Protons • Neutrons • Electrons • Question: Which of the three particles identifies what element an atom is? • The PROTON! (very important) ...
and View
... d. Atomic mass—mass of protons with neutrons. C. IVPAC—organization that chooses permanent name for element. ...
... d. Atomic mass—mass of protons with neutrons. C. IVPAC—organization that chooses permanent name for element. ...
11 what are elements made of
... When scientists make new elements they make new kinds of atoms. It is difficult to imagine anything as small as an atom. 100 million of them side by side will only measure about 1cm3. A single atom is so small that it cannot be weighed on a balance. There has to be a special scale to measure the mas ...
... When scientists make new elements they make new kinds of atoms. It is difficult to imagine anything as small as an atom. 100 million of them side by side will only measure about 1cm3. A single atom is so small that it cannot be weighed on a balance. There has to be a special scale to measure the mas ...
john dalton!! - Hawk Chemistry
... • John Dalton made an impact on the world as we know it. • According to Dalton, an element is composed of only one kind of atom, and a compound is composed of particles that are chemical combinations of different kinds of atoms. ...
... • John Dalton made an impact on the world as we know it. • According to Dalton, an element is composed of only one kind of atom, and a compound is composed of particles that are chemical combinations of different kinds of atoms. ...
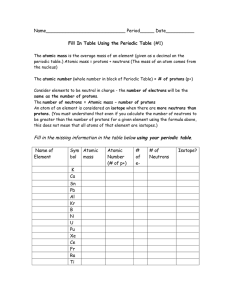
Periodic Table Fill in Table 1
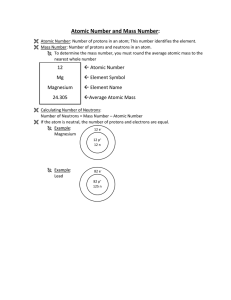
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
8.P.1.1 Warm-Up Questions for Website
... is made up of one type of atom. B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
... is made up of one type of atom. B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
MrsB-Chemistry
... Lesson 14: Isotopia (You will need the isotope graph handed out during lesson 14) 1. Would an atom with 50 protons and a mass of 155 amu be stable? ...
... Lesson 14: Isotopia (You will need the isotope graph handed out during lesson 14) 1. Would an atom with 50 protons and a mass of 155 amu be stable? ...
Intro to Atoms Clicker Questions 1. "atomos" means? 2. Atoms of one
... 2. Atoms of one kind of element _______ be changed into a different element with ordinary chemical means. (can, can’t) 3. Every compound is composed of atoms of different elements combined how? 4. In Thompson's model of the atom, the negatively charged electrons were located how in the atom? 5. In R ...
... 2. Atoms of one kind of element _______ be changed into a different element with ordinary chemical means. (can, can’t) 3. Every compound is composed of atoms of different elements combined how? 4. In Thompson's model of the atom, the negatively charged electrons were located how in the atom? 5. In R ...
Chapter One Outline
... The kinetic-molecular theory states that all matter consists of extremely small particles that are in constant motion. The higher the temperature the greater the speed of the particles The Atomic Theory An atom is the smallest particle of an element that embodies the chemical properties of an elemen ...
... The kinetic-molecular theory states that all matter consists of extremely small particles that are in constant motion. The higher the temperature the greater the speed of the particles The Atomic Theory An atom is the smallest particle of an element that embodies the chemical properties of an elemen ...
Understanding the Atom GN
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
Chemistry Notes
... • Are of the same element and have the same atomic number but different masses because they have different numbers of neutrons. ...
... • Are of the same element and have the same atomic number but different masses because they have different numbers of neutrons. ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.