Chapter #14 Newest CD
... CnH2n+2 Compounds containing only carbon and hydrogen with only single bonds and no multiple bonds, but a ring structure - Saturated hydrocarbons - Cycloalkanes CnH2n Compounds containing only carbon and hydrogen with double bonds - Unsaturated hydrocarbons - Alkenes CnH2n Compounds containing only ...
... CnH2n+2 Compounds containing only carbon and hydrogen with only single bonds and no multiple bonds, but a ring structure - Saturated hydrocarbons - Cycloalkanes CnH2n Compounds containing only carbon and hydrogen with double bonds - Unsaturated hydrocarbons - Alkenes CnH2n Compounds containing only ...
Learning Guide for Chapter 23: Amines
... Once an amine has been protonated, it is an ionic compound. Amine salts are solids, and are usually soluble in water but insoluble in organic solvents. Most biologically active amines are used as their amine salts, which dissolve to make injectable or drinkable water solutions, and are also less pro ...
... Once an amine has been protonated, it is an ionic compound. Amine salts are solids, and are usually soluble in water but insoluble in organic solvents. Most biologically active amines are used as their amine salts, which dissolve to make injectable or drinkable water solutions, and are also less pro ...
Chapter 11
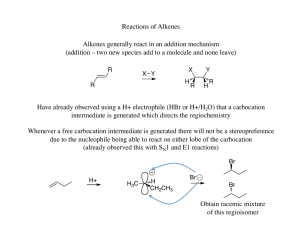
... (do both the X and Y groups add to the same side of the double bond or opposite sides?) ...
... (do both the X and Y groups add to the same side of the double bond or opposite sides?) ...
Organic Chemistry for Highschool Biology students
... Shown below are two short-hand versions of Odor 7, the one on the right includes all of the hydrogen atoms. Look at the arrows pointing at two different carbon atoms in the representation on the left. Now observe in the representation on the right that each of those carbon atoms has a different amou ...
... Shown below are two short-hand versions of Odor 7, the one on the right includes all of the hydrogen atoms. Look at the arrows pointing at two different carbon atoms in the representation on the left. Now observe in the representation on the right that each of those carbon atoms has a different amou ...
Review Graphite | SpringerLink
... It should be noted that pyrolytic graphite available before ∼1960 was not highly-oriented, so that experimental results on such materials should be treated with caution. 3. Bonding An isolated carbon atom has an electronic configuration of 1s2 2s2 2p2 . The 1s2 electrons belong to the ion core and t ...
... It should be noted that pyrolytic graphite available before ∼1960 was not highly-oriented, so that experimental results on such materials should be treated with caution. 3. Bonding An isolated carbon atom has an electronic configuration of 1s2 2s2 2p2 . The 1s2 electrons belong to the ion core and t ...
Organic Chemistry - UCR Chemistry
... atom becomes more oxidized. As the oxidation number becomes less positive (more negative) the C atom becomes more reduced. Do not memorize these oxidation numbers since they will change depending on the R group. But do learn the relative locations of compounds in each row, in order to understand whi ...
... atom becomes more oxidized. As the oxidation number becomes less positive (more negative) the C atom becomes more reduced. Do not memorize these oxidation numbers since they will change depending on the R group. But do learn the relative locations of compounds in each row, in order to understand whi ...
Chapter 3: Ionic and Covalent Compounds Chapter 3: Ionic and
... 80. Anions are formed when a neutral atom gains one or more electrons. A) True B) False Ans: A Difficulty: Easy 81. The (II) in the name of the ionic compound lead (II) acetate specifically indicates that there are two lead ions present in the compound. A) True B) False Ans: B Difficulty: Medium 82. ...
... 80. Anions are formed when a neutral atom gains one or more electrons. A) True B) False Ans: A Difficulty: Easy 81. The (II) in the name of the ionic compound lead (II) acetate specifically indicates that there are two lead ions present in the compound. A) True B) False Ans: B Difficulty: Medium 82. ...
Modern Organic Chemistry
... Inorganic, Organic, and Biological Chemistry Denniston, Topping, and Caret 4th ed ...
... Inorganic, Organic, and Biological Chemistry Denniston, Topping, and Caret 4th ed ...
Nuclear Magnetic Resonance and Potentiometric
... The protonation constants of the macrocyclic ligand 1,4,7-triazacyclononane-N,N’,N”-triacetic acid (NOTA) have been measured by potentiometry, and the protonation sequence of the various amino and carboxylate groups of NOTA has been studied in DzO as a function of pD from the chemical shifts of the ...
... The protonation constants of the macrocyclic ligand 1,4,7-triazacyclononane-N,N’,N”-triacetic acid (NOTA) have been measured by potentiometry, and the protonation sequence of the various amino and carboxylate groups of NOTA has been studied in DzO as a function of pD from the chemical shifts of the ...
Critical thoughts on computing atom condensed Fukui functions Bultinck and Stijn Fias
... view that molecules consist of atoms held together by chemical bonds. Many aspects of chemical reactivity are often traced back to the atoms and the bonds that compose the molecule, as this language facilitates constructing predictive models that only require information about the composition of a r ...
... view that molecules consist of atoms held together by chemical bonds. Many aspects of chemical reactivity are often traced back to the atoms and the bonds that compose the molecule, as this language facilitates constructing predictive models that only require information about the composition of a r ...
N-oxidation - WordPress.com
... several aromatic amines, including aniline and dapsone, and is a result of the bioconversion of the aromatic amine to its N-hydroxy derivative. • The N-hydroxylamine oxidizes the Fe2+ form of hemoglobin to its Fe3+ form. This oxidized (Fe3+) state of hemoglobin (called methemoglobin or ferrihemoglob ...
... several aromatic amines, including aniline and dapsone, and is a result of the bioconversion of the aromatic amine to its N-hydroxy derivative. • The N-hydroxylamine oxidizes the Fe2+ form of hemoglobin to its Fe3+ form. This oxidized (Fe3+) state of hemoglobin (called methemoglobin or ferrihemoglob ...
Unit 5: Oragnic Chemistry Notes (answers)
... 1. Most Alkenes and Alkynes are Non-Polar. They only have London Dispersion Forces. 2. Their boiling points are comparable to alkanes. The double bonds and triple bonds do NOT have any effect on their physical properties. 3. They do give off more heat (more exothermic) when burned (combusted) compar ...
... 1. Most Alkenes and Alkynes are Non-Polar. They only have London Dispersion Forces. 2. Their boiling points are comparable to alkanes. The double bonds and triple bonds do NOT have any effect on their physical properties. 3. They do give off more heat (more exothermic) when burned (combusted) compar ...
2 - Humble ISD
... restricted set of reactions. You can: 1. burn them - destroying the whole molecule 2. react them with some of the halogens, breaking carbon-hydrogen bonds; 3. crack them, breaking carbon-carbon bonds. ...
... restricted set of reactions. You can: 1. burn them - destroying the whole molecule 2. react them with some of the halogens, breaking carbon-hydrogen bonds; 3. crack them, breaking carbon-carbon bonds. ...
File
... The chemical properties (and toxicity) of organic compounds are determined by the compound as a whole. As a substitute in a molecule, a phenyl group ring does not have the same properties as benzene. ...
... The chemical properties (and toxicity) of organic compounds are determined by the compound as a whole. As a substitute in a molecule, a phenyl group ring does not have the same properties as benzene. ...
Sect 5 NMR Trends
... so alkenes and aromatics (and other p-bonds) are not isotropic – they have effects that are different in different directions – we call them ANISOTROPIC Circulation of p the -electrons around the ring ...
... so alkenes and aromatics (and other p-bonds) are not isotropic – they have effects that are different in different directions – we call them ANISOTROPIC Circulation of p the -electrons around the ring ...
Document
... Carbon Skeletons and Hydrogen Skins When determining the number of different skeletons, remember that Each C can form a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of C atoms determines the skeleton, so a straight chain and a bent c ...
... Carbon Skeletons and Hydrogen Skins When determining the number of different skeletons, remember that Each C can form a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of C atoms determines the skeleton, so a straight chain and a bent c ...
Extra Organic chemistry
... Carbon Skeletons and Hydrogen Skins When determining the number of different skeletons, remember that Each C can form a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of C atoms determines the skeleton, so a straight chain and a bent c ...
... Carbon Skeletons and Hydrogen Skins When determining the number of different skeletons, remember that Each C can form a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of C atoms determines the skeleton, so a straight chain and a bent c ...
Naming Organic Compounds
... CH3-CH2Cl + KOH CH3-CH2OH + KCl CH3-CH2OH + HCl CH3-CH2Cl + H2O CH3CH3 + Cl2 CH3CH2Cl + HCl ...
... CH3-CH2Cl + KOH CH3-CH2OH + KCl CH3-CH2OH + HCl CH3-CH2Cl + H2O CH3CH3 + Cl2 CH3CH2Cl + HCl ...
Aromaticity

In organic chemistry, the term aromaticity is formally used to describe an unusually stable nature of some flat rings of atoms. These structures contain a number of double bonds that interact with each other according to certain rules. As a result of their being so stable, such rings tend to form easily, and once formed, tend to be difficult to break in chemical reactions. Since one of the most commonly encountered aromatic system of compounds in organic chemistry is based on derivatives of the prototypical aromatic compound benzene (common in petroleum), the word “aromatic” is occasionally used to refer informally to benzene derivatives, and this is how it was first defined. Nevertheless, many non-benzene aromatic compounds exist. In living organisms, for example, the most common aromatic rings are the double-ringed bases in RNA and DNA.The earliest use of the term “aromatic” was in an article by August Wilhelm Hofmann in 1855. Hofmann used the term for a class of benzene compounds, many of which do have odors (unlike pure saturated hydrocarbons). Today, there is no general relationship between aromaticity as a chemical property and the olfactory properties of such compounds, although in 1855, before the structure of benzene or organic compounds was understood, chemists like Hofmann were beginning to understand that odiferous molecules from plants, such as terpenes, had chemical properties we recognize today are similar to unsaturated petroleum hydrocarbons like benzene.In terms of the electronic nature of the molecule, aromaticity describes the way a conjugated ring of unsaturated bonds, lone pairs of electrons, or empty molecular orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone. Aromaticity can be considered a manifestation of cyclic delocalization and of resonance. This is usually considered to be because electrons are free to cycle around circular arrangements of atoms that are alternately single- and double-bonded to one another. These bonds may be seen as a hybrid of a single bond and a double bond, each bond in the ring identical to every other. This commonly seen model of aromatic rings, namely the idea that benzene was formed from a six-membered carbon ring with alternating single and double bonds (cyclohexatriene), was developed by August Kekulé (see History section below). The model for benzene consists of two resonance forms, which corresponds to the double and single bonds superimposing to produce six one-and-a-half bonds. Benzene is a more stable molecule than would be expected without accounting for charge delocalization.