THESE DOCTORAT DE L`UNIVERSITE DE TOULOUSE ET
... The chemistry of molybdenum and tungsten cyclopentadienyl complexes in higher oxidation states with oxo, imido and sulfide ligands has increased in significance. Interest in Cp* oxo molybdenum and tungsten complexes is particularly motivated by their potential in oxidation catalysis. Most advances i ...
... The chemistry of molybdenum and tungsten cyclopentadienyl complexes in higher oxidation states with oxo, imido and sulfide ligands has increased in significance. Interest in Cp* oxo molybdenum and tungsten complexes is particularly motivated by their potential in oxidation catalysis. Most advances i ...
HYBRID MULTIDENTATE PHOSPHINE
... Stille,9 Suzuki-Miyaura,10 and Sonogashira,11 are well-established methods for the construction of C-C bonds, and widely applied in the synthesis of complex molecules. Over the last few decades considerable large research effort has been placed on the design and synthesis of highly active transition ...
... Stille,9 Suzuki-Miyaura,10 and Sonogashira,11 are well-established methods for the construction of C-C bonds, and widely applied in the synthesis of complex molecules. Over the last few decades considerable large research effort has been placed on the design and synthesis of highly active transition ...
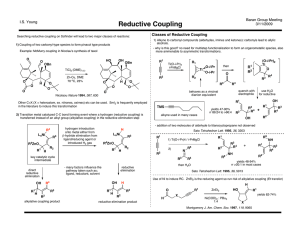
Reductive Couplings
... - degree of regioselectivity influenced by remote alkene - sense of regioselectivity controled by additive - with directing alkene and ligand combined, completely different mechanism ...
... - degree of regioselectivity influenced by remote alkene - sense of regioselectivity controled by additive - with directing alkene and ligand combined, completely different mechanism ...
Iridium(III) and Rhodium(III) compounds of dipyridyl-N
... {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ([(η5 -C5 Me5 ) Rh{(C5 H4 N)2 C=NH}])+ ([M]-PF6 -Cl)+ indicating the reaction also produces rhodium(III ...
... {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ([(η5 -C5 Me5 ) Rh{(C5 H4 N)2 C=NH}])+ ([M]-PF6 -Cl)+ indicating the reaction also produces rhodium(III ...
Learning Guide for Chapter 9 - Alkyl Halides I
... Show how an alkyl halide can react in each of the following ways: arrows? ...
... Show how an alkyl halide can react in each of the following ways: arrows? ...
File
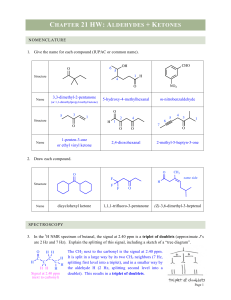
... the carbonyl species (starting reactant energies). The carbonyl carbon is δ+, and EDG lower the energy. The aldehyde has one alkyl group (EDG) attached to the C=O, but the ketone has 2 EDG. Therefore, the ketone stabilizes the δ+ more and starts at a lower energy than the aldehyde. This causes the h ...
... the carbonyl species (starting reactant energies). The carbonyl carbon is δ+, and EDG lower the energy. The aldehyde has one alkyl group (EDG) attached to the C=O, but the ketone has 2 EDG. Therefore, the ketone stabilizes the δ+ more and starts at a lower energy than the aldehyde. This causes the h ...
Photoremovable Protecting Groups
... When the literature is insufficient for providing a comprehensive treatment of applications of a photoprotecting group, then only a brief discussion is provided. An exhaustive list of applications for any of the chromophores is not included; these may be found by consulting other reviews or the orig ...
... When the literature is insufficient for providing a comprehensive treatment of applications of a photoprotecting group, then only a brief discussion is provided. An exhaustive list of applications for any of the chromophores is not included; these may be found by consulting other reviews or the orig ...
Organic Halides (Haloalkanes) (Alkyl Halides)
... • The presence of a halide makes the molecule more polar. • Since water is also polar and “like dissolves like”, alkyl halides are soluble in water. The more halides connected to the parent chain, the more polar the molecule. • The polar nature of the molecule means that boiling and melting points o ...
... • The presence of a halide makes the molecule more polar. • Since water is also polar and “like dissolves like”, alkyl halides are soluble in water. The more halides connected to the parent chain, the more polar the molecule. • The polar nature of the molecule means that boiling and melting points o ...
Alcohols
... compounds that have their –OH or –SH group bonded to a saturated, sp3-hybridized carbon atom • Phenols and thiophenols are compounds with their –OH or ...
... compounds that have their –OH or –SH group bonded to a saturated, sp3-hybridized carbon atom • Phenols and thiophenols are compounds with their –OH or ...
molecules
... It is found that addition of a heterocyclic nitrogen base such as imidazole or 1-methylimidazole to this catalytic system improves the activity of the catalyst. In the presence of imidazole, the Mn(Br8TPPS) –Ad-400 system converts different olefins efficiently to their corresponding epoxides (Table1 ...
... It is found that addition of a heterocyclic nitrogen base such as imidazole or 1-methylimidazole to this catalytic system improves the activity of the catalyst. In the presence of imidazole, the Mn(Br8TPPS) –Ad-400 system converts different olefins efficiently to their corresponding epoxides (Table1 ...
HOMOLOGATION OF HETEROCYCLES BY A SEQUENTIAL REDUCTIVE OPENING LITHIATION – S
... 04CRV2667) are of great interest in organic synthesis because polyfunctionalized molecules are obtained in a single synthetic operation by reaction with electrophilic reagents.(95MI5, 02MI6) Functionalized organolithium compounds can be prepared by halogen-lithium exchange, metal-lithium exchange, d ...
... 04CRV2667) are of great interest in organic synthesis because polyfunctionalized molecules are obtained in a single synthetic operation by reaction with electrophilic reagents.(95MI5, 02MI6) Functionalized organolithium compounds can be prepared by halogen-lithium exchange, metal-lithium exchange, d ...
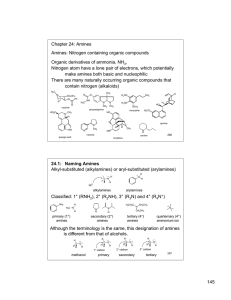
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Symmetrical secondary and tertiary amines are named by adding the prefix di- or tri- to the alkyl group Unsymmetrical secondary and tertiary amines are named as N-substituted primary amines. The largest alkyl group is the parent name, and other alkyl groups are considered N-substituents. H H3CH2C ...
... Symmetrical secondary and tertiary amines are named by adding the prefix di- or tri- to the alkyl group Unsymmetrical secondary and tertiary amines are named as N-substituted primary amines. The largest alkyl group is the parent name, and other alkyl groups are considered N-substituents. H H3CH2C ...
Mock Exam One
... a.) The carbon atom of a carbonyl group is electrophilic. b.) In general, aldehydes are more reactive than ketones. c.) Nucleophilic addition to carbonyl groups can be catalyzed by acid or base. d.) Addition of a nucleophile to a carbonyl group changes the hybridization of the carbonyl carbon from s ...
... a.) The carbon atom of a carbonyl group is electrophilic. b.) In general, aldehydes are more reactive than ketones. c.) Nucleophilic addition to carbonyl groups can be catalyzed by acid or base. d.) Addition of a nucleophile to a carbonyl group changes the hybridization of the carbonyl carbon from s ...
Abdullah F. Eid
... The octahedra are arranged in four M3O13 groups. Each group is formed by three edges sharing octahedra and having a common oxygen atom which is also shared with the central tetrahedron XO4 . Among a wide variety of HPAs, the Keggin’s are the most stable and more easily available; these are the most ...
... The octahedra are arranged in four M3O13 groups. Each group is formed by three edges sharing octahedra and having a common oxygen atom which is also shared with the central tetrahedron XO4 . Among a wide variety of HPAs, the Keggin’s are the most stable and more easily available; these are the most ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... the ions that each contains. We then correlate these charged ionic species with the ones shown in the diagram. Solve: The diagram shows twice as many cations as anions, consistent with the formulation K 2SO4. Aqueous Check: Notice that the total net charge in the diagram is zero, as it must be if it ...
... the ions that each contains. We then correlate these charged ionic species with the ones shown in the diagram. Solve: The diagram shows twice as many cations as anions, consistent with the formulation K 2SO4. Aqueous Check: Notice that the total net charge in the diagram is zero, as it must be if it ...
Organic Chemistry Fifth Edition
... But the method doesn't work well in practice. Usually gives a mixture of primary, secondary, and tertiary amines, plus the quaternary salt. ...
... But the method doesn't work well in practice. Usually gives a mixture of primary, secondary, and tertiary amines, plus the quaternary salt. ...
Chapter 11
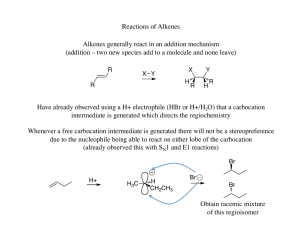
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
Alcohols
... compounds • Thiols, also called mercaptans, are named using the same system as for alcohols, with the suffix –thiol in place of –ol • The –SH group is sometimes referred to as the mercapto ...
... compounds • Thiols, also called mercaptans, are named using the same system as for alcohols, with the suffix –thiol in place of –ol • The –SH group is sometimes referred to as the mercapto ...
aq - Haverford Alchemy
... the ions that each contains. We then correlate these charged ionic species with the ones shown in the diagram. Solve: The diagram shows twice as many cations as anions, consistent with the formulation K 2SO4. Aqueous Check: Notice that the total net charge in the diagram is zero, as it must be if it ...
... the ions that each contains. We then correlate these charged ionic species with the ones shown in the diagram. Solve: The diagram shows twice as many cations as anions, consistent with the formulation K 2SO4. Aqueous Check: Notice that the total net charge in the diagram is zero, as it must be if it ...
Enantioselective synthesis

Enantioselective synthesis, also called chiral synthesis or asymmetric synthesis, is defined by IUPAC as: a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereoisomeric) products in unequal amounts.Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer.Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity.