![Covalently Bonded Platinum(II) Complexes of [alpha]](http://s1.studyres.com/store/data/022412983_1-66c66ee18551a43164a79702fd995f95-300x300.png)
View/Open - AURA - Alfred University
... that triflic acid is not responsible for the progress of the reaction, suggests that PEDOT can mediate the Ritter reaction under mild conditions and it may now be possible to use unprotected, acid-sensitive functional groups. In an attempt to determine the scope of the PEDOT-mediated Ritter reactio ...
... that triflic acid is not responsible for the progress of the reaction, suggests that PEDOT can mediate the Ritter reaction under mild conditions and it may now be possible to use unprotected, acid-sensitive functional groups. In an attempt to determine the scope of the PEDOT-mediated Ritter reactio ...
CHM 235 Course Outline and Homework in McMurry (6th ed.)
... Introduction to organic synthesis (the fun part!) Chapter 9: 9.34-9.36, 9..38-9.46, 9.51-9.55, 9.65, 9.67, 9.79, 9.80 Enantiomers, chirality, specific rotation Sequence rules for R and S Diastereomers and meso compounds Racemic mixtures and their resolution (chiral drugs!) Fischer projec ...
... Introduction to organic synthesis (the fun part!) Chapter 9: 9.34-9.36, 9..38-9.46, 9.51-9.55, 9.65, 9.67, 9.79, 9.80 Enantiomers, chirality, specific rotation Sequence rules for R and S Diastereomers and meso compounds Racemic mixtures and their resolution (chiral drugs!) Fischer projec ...
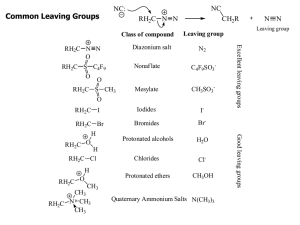
Common Leaving Groups
... •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that will favour one reaction over another, more often than not you will ...
... •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that will favour one reaction over another, more often than not you will ...
Aromatic Compounds
... The nitro-substituted product can be reduced by reagents such as iron or tin metal or to yield an arylamine, ArNH2 Attachment of an amino group to an aromatic ring by the two-step nitration-reduction sequence is a key part of the industrial synthesis of many dyes and pharmaceutical agents ...
... The nitro-substituted product can be reduced by reagents such as iron or tin metal or to yield an arylamine, ArNH2 Attachment of an amino group to an aromatic ring by the two-step nitration-reduction sequence is a key part of the industrial synthesis of many dyes and pharmaceutical agents ...
Full text - Loschmidt Laboratories
... water represents a challenge for reaction engineering in terms of downstream processing and integration into a process chain due to its high boiling point and high heat of vaporization (Ghanem and Aboul-Enein, 2004). The use of organic solvents as reaction media for biocatalytic reactions has proven ...
... water represents a challenge for reaction engineering in terms of downstream processing and integration into a process chain due to its high boiling point and high heat of vaporization (Ghanem and Aboul-Enein, 2004). The use of organic solvents as reaction media for biocatalytic reactions has proven ...
M.Sc. Chemistry - Periyar University
... Thermodynamic and kinetic requirements for reactions, thermodynamic and kinetic control reactions, Hammonds postulate, Microscopic reversibility. Potential energy diagrams, transition states and intermediates, methods of determining mechanisms – identification of products and determination of the pr ...
... Thermodynamic and kinetic requirements for reactions, thermodynamic and kinetic control reactions, Hammonds postulate, Microscopic reversibility. Potential energy diagrams, transition states and intermediates, methods of determining mechanisms – identification of products and determination of the pr ...
organonitrogen compounds i. amines
... seen their structures before. Many of the alkaloids are polycyclic structures and have other functional groups in addition to basic nitrogen. You will see that the nitrogens of alkaloids frequently are tertiary amine functions. All of the alkaloids shown in Figure 23-1 are substances with very prono ...
... seen their structures before. Many of the alkaloids are polycyclic structures and have other functional groups in addition to basic nitrogen. You will see that the nitrogens of alkaloids frequently are tertiary amine functions. All of the alkaloids shown in Figure 23-1 are substances with very prono ...
Organic synthesis and methodology related to the malaria drug artemisinin
... combat this devastating disease. Artemisinin has emerged as the drug of choice for treatment of malaria due to its effectiveness against all strains of the malaria parasite. Access to artemisinin through isolation, bio-engineering, and chemical synthesis will be described. Our attempts to access the ...
... combat this devastating disease. Artemisinin has emerged as the drug of choice for treatment of malaria due to its effectiveness against all strains of the malaria parasite. Access to artemisinin through isolation, bio-engineering, and chemical synthesis will be described. Our attempts to access the ...
Full-Text PDF
... The high reactivity of aldehydes makes them a key functional group in organic chemistry. This group is widespread in Nature, and its use in the synthesis of natural products is noteworthy. Furthermore, as efficient electrophiles, aldehydes can undergo further transformations to be converted into an ...
... The high reactivity of aldehydes makes them a key functional group in organic chemistry. This group is widespread in Nature, and its use in the synthesis of natural products is noteworthy. Furthermore, as efficient electrophiles, aldehydes can undergo further transformations to be converted into an ...
Full Text
... substrate and should be selectively removed in good yield by readily available, preferably nontoxic reagents that do not attack the regenerated functional group.1 One of the most abundant functional groups is the hydroxyl group, which is present in a number of compounds of biological and synthetic i ...
... substrate and should be selectively removed in good yield by readily available, preferably nontoxic reagents that do not attack the regenerated functional group.1 One of the most abundant functional groups is the hydroxyl group, which is present in a number of compounds of biological and synthetic i ...
R - Evans - Harvard University
... investigation.* The established methods for the asymmetric synthesis of a-amino acids can be categorized according to the nature of the stereodifferentiating event. The analysis provided in Scheme I indicates five recognized ways of stereoselectively constructing the n-stereogenic center of a simple ...
... investigation.* The established methods for the asymmetric synthesis of a-amino acids can be categorized according to the nature of the stereodifferentiating event. The analysis provided in Scheme I indicates five recognized ways of stereoselectively constructing the n-stereogenic center of a simple ...
Alkyl halide
... Draw the alkene reactant and identify the allylic positions. Label the two different allylic positions A and B. Now abstract an allylic hydrogen from each position to generate the two corresponding allylic radicals. Each of the two allylic radicals can add a Br atom at either end (A or a; B or b) to ...
... Draw the alkene reactant and identify the allylic positions. Label the two different allylic positions A and B. Now abstract an allylic hydrogen from each position to generate the two corresponding allylic radicals. Each of the two allylic radicals can add a Br atom at either end (A or a; B or b) to ...
Redox reactions_organic - Welcome to SALEM
... • Only a restricted number of simple, relatively cheap starting materials is available • Include: simple haloalkanes and alcohols of not more than four carbon atoms simple aromatic compounds (e.g. benzene and methylbenzene) ...
... • Only a restricted number of simple, relatively cheap starting materials is available • Include: simple haloalkanes and alcohols of not more than four carbon atoms simple aromatic compounds (e.g. benzene and methylbenzene) ...
Analytical Chemistry - University of Delhi
... only on chemistry, but also on physics, biology, information theory and many fields of technology. It is of fundamental importance not only to all branches of chemistry but also to all biological sciences, engineering sciences, health, medicine, pharmaceuticals, environment, industrial processes, qu ...
... only on chemistry, but also on physics, biology, information theory and many fields of technology. It is of fundamental importance not only to all branches of chemistry but also to all biological sciences, engineering sciences, health, medicine, pharmaceuticals, environment, industrial processes, qu ...
Enantioselective synthesis

Enantioselective synthesis, also called chiral synthesis or asymmetric synthesis, is defined by IUPAC as: a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereoisomeric) products in unequal amounts.Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer.Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity.