Chapter 6
... • Sugars and amino acids with same relative configuration as (+)-glyceraldehyde were assigned D and same as (-)glyceraldehyde were assigned L. • With X-ray crystallography, now know absolute configurations: D is (R) and L is (S). • No relationship to dextro- or levorotatory. ...
... • Sugars and amino acids with same relative configuration as (+)-glyceraldehyde were assigned D and same as (-)glyceraldehyde were assigned L. • With X-ray crystallography, now know absolute configurations: D is (R) and L is (S). • No relationship to dextro- or levorotatory. ...
Visible Light Photoredox Catalysis with Transition
... commercial household light bulb, a significant advantage over the specialized equipment required for processes employing high-energy ultraviolet (UV) light. Additionally, because organic molecules generally do not absorb visible light, there is little potential for deleterious side reactions that mig ...
... commercial household light bulb, a significant advantage over the specialized equipment required for processes employing high-energy ultraviolet (UV) light. Additionally, because organic molecules generally do not absorb visible light, there is little potential for deleterious side reactions that mig ...
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic
... reaction kinetics. Leaving groups and nucleophiles are often the same for both mechanisms, and the structure of the reactant with the leaving group (the substrate) usually determines the reaction mechanism. The relative reactivities of nucleophiles (nucleophilicity) and leaving groups (leaving group ...
... reaction kinetics. Leaving groups and nucleophiles are often the same for both mechanisms, and the structure of the reactant with the leaving group (the substrate) usually determines the reaction mechanism. The relative reactivities of nucleophiles (nucleophilicity) and leaving groups (leaving group ...
13C NMR - Creighton Chemistry Webserver
... C - 13C spin-spin splitting is never observed almost no molecules have $ 2 13C in them: for example, the probability of hexane having two adjacent 13C is 0.011 x 0.011 x 0.29 = 3.5 x 10 -6 ...
... C - 13C spin-spin splitting is never observed almost no molecules have $ 2 13C in them: for example, the probability of hexane having two adjacent 13C is 0.011 x 0.011 x 0.29 = 3.5 x 10 -6 ...
Synthesis And Antifungal Activity Of Terbinafine Analogues
... Preparation of substituted naphthalene ethers (1-14) General procedure: In a 250 mL RBF, 1.49 mL (0.01 mole) of 1-chloromethyl naphthalene (1.49 mL, 0.01 mole) was dissolved in 30 mL of dry acetone. To this solution, 0.01 mole of aryl alcohols and 0.01 mol of anhydrous potassium carbonate were added ...
... Preparation of substituted naphthalene ethers (1-14) General procedure: In a 250 mL RBF, 1.49 mL (0.01 mole) of 1-chloromethyl naphthalene (1.49 mL, 0.01 mole) was dissolved in 30 mL of dry acetone. To this solution, 0.01 mole of aryl alcohols and 0.01 mol of anhydrous potassium carbonate were added ...
Recent developments in the synthesis of functional poly(olefin)s
... M.J. Yanjarappa, S. Sivaram / Prog. Polym. Sci. 27 (2002) 1347±1398 3.1.1. Copolymerization of ethylene with v-chloro-a-ole®ns . . . . . . . . . . . . . . . . . . . . . . . . .1369 3.1.2. Copolymerization of ole®ns with amine functionalized ole®ns . . . . . . . . . . . . . . . . . . . .1369 3.1.3. C ...
... M.J. Yanjarappa, S. Sivaram / Prog. Polym. Sci. 27 (2002) 1347±1398 3.1.1. Copolymerization of ethylene with v-chloro-a-ole®ns . . . . . . . . . . . . . . . . . . . . . . . . .1369 3.1.2. Copolymerization of ole®ns with amine functionalized ole®ns . . . . . . . . . . . . . . . . . . . .1369 3.1.3. C ...
Organic Chemistry with a Biological Emphasis Volume I
... Composed of the four elements carbon, hydrogen, oxygen and nitrogen, capsaicin is produced by the pepper plant for the purpose of warding off hungry mammals. The molecule binds to and activates a mammalian receptor protein called TrpV1, which in normal circumstances has the job of detecting high tem ...
... Composed of the four elements carbon, hydrogen, oxygen and nitrogen, capsaicin is produced by the pepper plant for the purpose of warding off hungry mammals. The molecule binds to and activates a mammalian receptor protein called TrpV1, which in normal circumstances has the job of detecting high tem ...
STOICHIOMETRY via ChemLog - Small
... When carrying out a chemical reaction, we may use the exact amount of each reactant needed. Or, we may use an excess of some reactants and a limited amount of others. We may do this if one reactant is very expensive and others are inexpensive so that we can use all of the expensive compound. It can ...
... When carrying out a chemical reaction, we may use the exact amount of each reactant needed. Or, we may use an excess of some reactants and a limited amount of others. We may do this if one reactant is very expensive and others are inexpensive so that we can use all of the expensive compound. It can ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
Alkyl Halides SN and E reactions
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
... Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs with 2° or 3° substrates. Once the C+ forms, both SN1 and E1 oc ...
Silylation Overview - Sigma
... used since the late fifties in gas chromatography and mass spectrometry, for the derivatisation of a wide variety of products and functional groups. Silylation of a polar compound results in reduced polarity, enhanced volatility and increased thermal stability, and enables the GC-MS analysis of many ...
... used since the late fifties in gas chromatography and mass spectrometry, for the derivatisation of a wide variety of products and functional groups. Silylation of a polar compound results in reduced polarity, enhanced volatility and increased thermal stability, and enables the GC-MS analysis of many ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
Handout VI
... The attack of hydride ion on the carbonyl group, i.e., the reduction of aldehydes and ketones may be seen as nucleophilic addition but the actual pathway is quite complicated. Lithium aluminium hydride is a very powerful reducing agent and easily reduces both aldehydes and ketones to their correspon ...
... The attack of hydride ion on the carbonyl group, i.e., the reduction of aldehydes and ketones may be seen as nucleophilic addition but the actual pathway is quite complicated. Lithium aluminium hydride is a very powerful reducing agent and easily reduces both aldehydes and ketones to their correspon ...
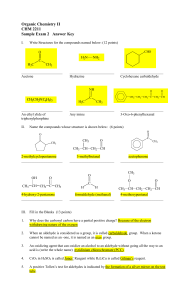
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
Chemistry
... pair repulsion theory (VSEPR), shapes of the following simple molecules and ions containing lone pairs and bond pairs of electrons: H2O, NH3, PCl3, PCl5, SF6, ClF3, I3-, BrF2+, PCl6-, ICl2ICl4- and SO42-. Multiple bonding (ζ and π bond approach) and bond lengths. Covalent character in ionic compound ...
... pair repulsion theory (VSEPR), shapes of the following simple molecules and ions containing lone pairs and bond pairs of electrons: H2O, NH3, PCl3, PCl5, SF6, ClF3, I3-, BrF2+, PCl6-, ICl2ICl4- and SO42-. Multiple bonding (ζ and π bond approach) and bond lengths. Covalent character in ionic compound ...
UNIVERSITY OF DELHI FACULTY OF SCIENCE SYLLABUS OF COURSES TO BE OFFERED
... there is a need to devise a sensible system for awarding the grades based on the performance of students. Presently the performance of the students is reported using the conventional system of marks secured in the examinations or grades or both. The conversion from marks to letter grades and the let ...
... there is a need to devise a sensible system for awarding the grades based on the performance of students. Presently the performance of the students is reported using the conventional system of marks secured in the examinations or grades or both. The conversion from marks to letter grades and the let ...
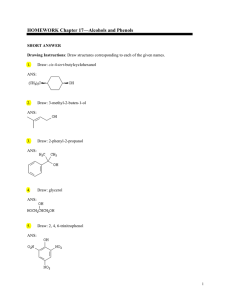
HOMEWORK Chapter 17—Alcohols and Phenols
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
Alkyl Halides02
... nature of the solvent. In general, factors that lower G*, either by stabilizing C+ intermediate or by raising the reactant energy, favor faster SN1 reactions. Conversely, factors that raise G*, either by destabilizing the C+ intermediate or by lowering reactant energy level, slow down SN1 reaction ...
... nature of the solvent. In general, factors that lower G*, either by stabilizing C+ intermediate or by raising the reactant energy, favor faster SN1 reactions. Conversely, factors that raise G*, either by destabilizing the C+ intermediate or by lowering reactant energy level, slow down SN1 reaction ...
Enantioselective synthesis

Enantioselective synthesis, also called chiral synthesis or asymmetric synthesis, is defined by IUPAC as: a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereoisomeric) products in unequal amounts.Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer.Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity.