i m. pharm. - Rajiv Gandhi University of Health Sciences
... the practical pyruvic acid production was 42.4 g/L. The determination coefficient (R2) was 0.9483, which ensures adequate credibility of the model. 8. Chanakya Pallem et al 12., carried out solid state fermentation (SSF) for the production of L-glutaminase by the fungal strain Trichoderma koningii ...
... the practical pyruvic acid production was 42.4 g/L. The determination coefficient (R2) was 0.9483, which ensures adequate credibility of the model. 8. Chanakya Pallem et al 12., carried out solid state fermentation (SSF) for the production of L-glutaminase by the fungal strain Trichoderma koningii ...
Assignment 2 Group A and B
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
Answer Key to Assignment #7
... 2. How would you prepare the following molecules? More than one step may be necessary. You may use any reagents/solvents you wish. ...
... 2. How would you prepare the following molecules? More than one step may be necessary. You may use any reagents/solvents you wish. ...
Optical Isomers or Enantiomers
... difference in physical and chemical properties. • Chemical differences can arise when enantiomers interact with other optically active substances. • Chemical reactions which produce a chiral carbon often contain equal amounts of the two enatniomers. This is called a racemic mixture. • The only diffe ...
... difference in physical and chemical properties. • Chemical differences can arise when enantiomers interact with other optically active substances. • Chemical reactions which produce a chiral carbon often contain equal amounts of the two enatniomers. This is called a racemic mixture. • The only diffe ...
Day 16 – Intro to Chemistry
... chemistry and how are they used in the structure of organic macromolecules ...
... chemistry and how are they used in the structure of organic macromolecules ...
Solution-Phase Combinatorial Chemistry
... focus was on solid-phase approaches due to the many advantages. • Solution chemistry was not regarded as being suitable for combinatorial chemistry because of the often tedious isolation and purification. • It was first used for easily synthesized compound classes [amides, sulfonamides, ureas, heter ...
... focus was on solid-phase approaches due to the many advantages. • Solution chemistry was not regarded as being suitable for combinatorial chemistry because of the often tedious isolation and purification. • It was first used for easily synthesized compound classes [amides, sulfonamides, ureas, heter ...
Exam 3 - Organic Chemistry at CU Boulder
... Appendix: Periodic Table, common NMR, IR absorption ...
... Appendix: Periodic Table, common NMR, IR absorption ...
Gizmos: Types of Reactions
... is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Question: How are chemical reactions classified? 1. Match: Most chemical reactions can be classified as one ...
... is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Question: How are chemical reactions classified? 1. Match: Most chemical reactions can be classified as one ...
Synthesis of Platinum Group Metal Complexes for
... • Synthesis of perfluorinated or partially fluorinated organic and organometallic compounds in order to prepare supramolecular arrangements. Study of the physical/chemical properties of multiblocks semifluorinated compounds either as small molecules and gas dissolving agents. • Functionalization of ...
... • Synthesis of perfluorinated or partially fluorinated organic and organometallic compounds in order to prepare supramolecular arrangements. Study of the physical/chemical properties of multiblocks semifluorinated compounds either as small molecules and gas dissolving agents. • Functionalization of ...
Conducting Polymer
... The polyacetylene film forms at the gas-liquid interface when acetylene gas passes through a heptane solution of the Ziegler-Natta catalyst. Cis polymer forms at low temperature (-78 °C). Isomerization to the more stable trans form takes place on rising the temperature of the film. Conductivity of d ...
... The polyacetylene film forms at the gas-liquid interface when acetylene gas passes through a heptane solution of the Ziegler-Natta catalyst. Cis polymer forms at low temperature (-78 °C). Isomerization to the more stable trans form takes place on rising the temperature of the film. Conductivity of d ...
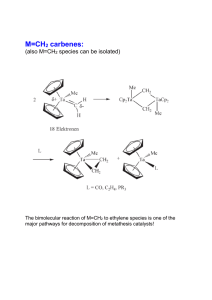
Steric protection of alkylidene is not needed:
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
MS PowerPoint - Catalysis Eprints database
... I. V. Kozhevnikov, Russ. Chem. Rev. 56 (1987) 811 M. Misono, N. Mizuno, K. Katamura, A. Kasai, Y. Konishi, K. Sakata, T. Okuhara, Y. Yoneda, Bull. Chem. Soc. Jpn. 55 (1982) 400 ...
... I. V. Kozhevnikov, Russ. Chem. Rev. 56 (1987) 811 M. Misono, N. Mizuno, K. Katamura, A. Kasai, Y. Konishi, K. Sakata, T. Okuhara, Y. Yoneda, Bull. Chem. Soc. Jpn. 55 (1982) 400 ...
Chemical Reactions
... 1. Begin by taking an element “inventory” for reactants and products. 2. Pick an element that only occurs once on each ...
... 1. Begin by taking an element “inventory” for reactants and products. 2. Pick an element that only occurs once on each ...
Exam 3 Review
... What is the name for this alcohol / ether / aldehyde / ketone? Is an alcohol 1°, 2°, or 3°? Describe hydrogen bonding in alcohols, and compare alcohol polarity to ether polarity. What are the acid/base properties of alcohols? Rank these compounds in order of acidity. How are Grignard reagents prepar ...
... What is the name for this alcohol / ether / aldehyde / ketone? Is an alcohol 1°, 2°, or 3°? Describe hydrogen bonding in alcohols, and compare alcohol polarity to ether polarity. What are the acid/base properties of alcohols? Rank these compounds in order of acidity. How are Grignard reagents prepar ...
Organometallic Reactions and Catalysis
... diphosphane, prochiral unsaturated molecules can be hydrogenated to chiral products (enantiomeric selectivity). • L-Dopa (treatment of Parkinson’s disease). ...
... diphosphane, prochiral unsaturated molecules can be hydrogenated to chiral products (enantiomeric selectivity). • L-Dopa (treatment of Parkinson’s disease). ...
Carbohydrates Typical formula: C (H O) , eg glucose: C H O
... Aldoses and ketoses may form internal hemiacetals (the carbonyl reacts with an -OH in the same molecule), resulting in a ring. If the ring is 5-membered, it is called a furanose; if it is 6-membered, it is called a pyranose. [Note that when the ring is formed, the former carbonyl carbon becomes tetr ...
... Aldoses and ketoses may form internal hemiacetals (the carbonyl reacts with an -OH in the same molecule), resulting in a ring. If the ring is 5-membered, it is called a furanose; if it is 6-membered, it is called a pyranose. [Note that when the ring is formed, the former carbonyl carbon becomes tetr ...
Topic 20 IB Chemistry Definitions
... Chemistry: Definitions Topic 20 – Organic Chemistry Chiral center: ...
... Chemistry: Definitions Topic 20 – Organic Chemistry Chiral center: ...
Dr. Amit Basak - Department of Chemistry, IIT Kgp
... Stereogenic Centre: An atom, again usually carbon, in which interchanging the position of two ligands gives rise to a stereoisomer. This includes the carbon atom attached to four different ligands as also the sp2 carbon which can ...
... Stereogenic Centre: An atom, again usually carbon, in which interchanging the position of two ligands gives rise to a stereoisomer. This includes the carbon atom attached to four different ligands as also the sp2 carbon which can ...
The first practical method for asymmetric epoxidation
... (9) Due to the small scale, we have chosen to ignore the excess TBHP. If one wishes to remove it, a number of reductive procedures are available.8 (IO) Do not expose the reaction mixture to this base treatment for longer than ‘ / 2 h as base-catalyzed rearrangements of the epoxy alcohol may occur: G ...
... (9) Due to the small scale, we have chosen to ignore the excess TBHP. If one wishes to remove it, a number of reductive procedures are available.8 (IO) Do not expose the reaction mixture to this base treatment for longer than ‘ / 2 h as base-catalyzed rearrangements of the epoxy alcohol may occur: G ...
Efficient one pot synthesis of N-alkyl and N-aryl imides
... of their potential utility, some of the reported methods suffer from drawbacks such as long reaction times, cumbersome product isolation procedures. Thus there is still need for development of new method for synthesis of imides. In the present work we have reported use of sulphamic acid for synthesi ...
... of their potential utility, some of the reported methods suffer from drawbacks such as long reaction times, cumbersome product isolation procedures. Thus there is still need for development of new method for synthesis of imides. In the present work we have reported use of sulphamic acid for synthesi ...
SCH4U Organic Chemistry Portfolio Name: This portfolio is due on
... 5. Nomex is a polymer used to make flame-resistant clothing for firefighters. A portion of its structure is provided below. Write a polymerization reaction showing its production from monomers. What type of reaction is this? ...
... 5. Nomex is a polymer used to make flame-resistant clothing for firefighters. A portion of its structure is provided below. Write a polymerization reaction showing its production from monomers. What type of reaction is this? ...
Catalytic Enantioselective Dibromination of Allylic Alcohols
... the introduction of polar functionality capable of binding to the metal might lead to improvements in the reactivity, selectivity, and product utility. Indeed, cinnamyl alcohol (6) was converted to dibromide (−)-7 with 43% ee using (R,R)-TADDOL 8 (entry 3). Allylic ethers, amides, and sulfonamides w ...
... the introduction of polar functionality capable of binding to the metal might lead to improvements in the reactivity, selectivity, and product utility. Indeed, cinnamyl alcohol (6) was converted to dibromide (−)-7 with 43% ee using (R,R)-TADDOL 8 (entry 3). Allylic ethers, amides, and sulfonamides w ...
Enantioselective synthesis

Enantioselective synthesis, also called chiral synthesis or asymmetric synthesis, is defined by IUPAC as: a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereoisomeric) products in unequal amounts.Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer.Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity.