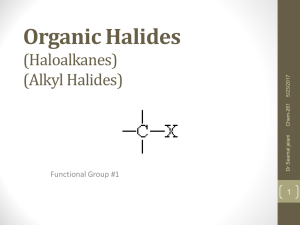CHEMISTRY 211 FINAL EXAM Wed., December 4, 2002 Name
... briefly, but clearly, what type of reaction is taking place in each case and why this would lead to the ...
... briefly, but clearly, what type of reaction is taking place in each case and why this would lead to the ...
Review of Organic Chem II
... 1. Reactant Stability/Reactivity: The more stable the reactant, the less reactive it will be. In terms of rates, this means that the more stable the reactant, the slower it will react. (The concept here is that the more stable the reactant, the more content it is to stay as is, and the less motivate ...
... 1. Reactant Stability/Reactivity: The more stable the reactant, the less reactive it will be. In terms of rates, this means that the more stable the reactant, the slower it will react. (The concept here is that the more stable the reactant, the more content it is to stay as is, and the less motivate ...
Organic Chemistry II with Dr Roche
... Ethers will often form complexes with molecules that have vacant orbitals, enabling ‘unstable’ molecules to be used as reagents. E.g. Hydroboration uses BH3.THF ...
... Ethers will often form complexes with molecules that have vacant orbitals, enabling ‘unstable’ molecules to be used as reagents. E.g. Hydroboration uses BH3.THF ...
Chapter 10:Alcohols, Phenols and Ethers
... Much of the chemistry of phenols is like that of aliphatic alcohols. For example, phenols can be acylated to give esters, and phenoxide ions can serve as nucleophiles in the Williamson ether synthesis. Formation of phenoxide ions is particularly easy because phenols are more acidic than water, aqueo ...
... Much of the chemistry of phenols is like that of aliphatic alcohols. For example, phenols can be acylated to give esters, and phenoxide ions can serve as nucleophiles in the Williamson ether synthesis. Formation of phenoxide ions is particularly easy because phenols are more acidic than water, aqueo ...
Organic Chemistry
... by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)benzene (PS-DIB) as the oxidant under mild conditions. PS-DIB, a yellow powder, was prepared from iodinatio ...
... by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)benzene (PS-DIB) as the oxidant under mild conditions. PS-DIB, a yellow powder, was prepared from iodinatio ...
Alcohols - Angelo State University
... • The O—H bond is therefore a polar bond, and any molecule which contains an O—H bond (like an alcohol) is a polar molecule. ...
... • The O—H bond is therefore a polar bond, and any molecule which contains an O—H bond (like an alcohol) is a polar molecule. ...
研 究 業 績 リ ス ト
... (92) Ruthenium-Catalyzed Intramolecular Cyclization of 3-Butyne-1,2-diols into Furans Y. Yada, Y. Miyake, and Y. Nishibayashi Org. Lett., to be submitted. (93) Novel Monophosphido-Bridged Diruthenium Complexes: Efficient Preparative Method and Their Catalytic Activity toward Condensation of Aldehyde ...
... (92) Ruthenium-Catalyzed Intramolecular Cyclization of 3-Butyne-1,2-diols into Furans Y. Yada, Y. Miyake, and Y. Nishibayashi Org. Lett., to be submitted. (93) Novel Monophosphido-Bridged Diruthenium Complexes: Efficient Preparative Method and Their Catalytic Activity toward Condensation of Aldehyde ...
Reactions of Alcohols
... bromide. • Add ZnCl2, which bonds strongly with -OH, to promote the reaction. • The chloride product is insoluble. • Lucas test: ZnCl2 in conc. HCl 1° alcohols react slowly or not at all. 2 alcohols react in 1-5 minutes. 3 alcohols react in less than 1 minute. ...
... bromide. • Add ZnCl2, which bonds strongly with -OH, to promote the reaction. • The chloride product is insoluble. • Lucas test: ZnCl2 in conc. HCl 1° alcohols react slowly or not at all. 2 alcohols react in 1-5 minutes. 3 alcohols react in less than 1 minute. ...
Orbitals
... • If neutral, the nucleophile usually carries a hydrogen atom that can be subsequently eliminated ...
... • If neutral, the nucleophile usually carries a hydrogen atom that can be subsequently eliminated ...
Full Text
... functional groups is the hydroxyl group, which is present in a number of compounds of biological and synthetic interest, including nucleosides, carbohydrates, steroids, macrolides, polyethers, and the side chain of some amino acids or in large numbers of intermediates in total syntheses of complex n ...
... functional groups is the hydroxyl group, which is present in a number of compounds of biological and synthetic interest, including nucleosides, carbohydrates, steroids, macrolides, polyethers, and the side chain of some amino acids or in large numbers of intermediates in total syntheses of complex n ...
Development of Catalytic Ester Condensations and Hydrolysis of
... yield) along with the ester of hydrophilic acetic acid in 5% yield (Scheme 8). In contrast, the same reaction in heptane preferentially gave the acetate (85% yield). This selectivity was based on the difference in hydrophobicity of the substrates. ...
... yield) along with the ester of hydrophilic acetic acid in 5% yield (Scheme 8). In contrast, the same reaction in heptane preferentially gave the acetate (85% yield). This selectivity was based on the difference in hydrophobicity of the substrates. ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... experimental observations. Together, these provide a thorough understanding of the mechanism. In search of novel molecular rhenium complexes as catalysts for the dehydration reaction, we have synthesized a series of rhenium complexes bearing a bis(phosphinomethyl)pyridine PNP-pincer ligand. These co ...
... experimental observations. Together, these provide a thorough understanding of the mechanism. In search of novel molecular rhenium complexes as catalysts for the dehydration reaction, we have synthesized a series of rhenium complexes bearing a bis(phosphinomethyl)pyridine PNP-pincer ligand. These co ...
Chapter 11 Reactions of Alcohols Types of Alcohol Reactions
... • Add ZnCl2, which bonds strongly with -OH, to promote the reaction. • The chloride product is insoluble. • Lucas test: ZnCl2 in conc. HCl ¾1° alcohols react slowly or not at all. ¾2° alcohols react in 1-5 minutes. ¾3° alcohols react in less than 1 minute. ...
... • Add ZnCl2, which bonds strongly with -OH, to promote the reaction. • The chloride product is insoluble. • Lucas test: ZnCl2 in conc. HCl ¾1° alcohols react slowly or not at all. ¾2° alcohols react in 1-5 minutes. ¾3° alcohols react in less than 1 minute. ...
Cerium(IV) Ammonium Nitrate as a Catalyst in
... to other cations, and for this reason its salts, and especially the commercially available cerium(IV) ammonium nitrate (CAN), have found widespread use as one-electron oxidants.1 More specifically, CAN has been found to be chemically superior in many respects to the widely employed manganese triacet ...
... to other cations, and for this reason its salts, and especially the commercially available cerium(IV) ammonium nitrate (CAN), have found widespread use as one-electron oxidants.1 More specifically, CAN has been found to be chemically superior in many respects to the widely employed manganese triacet ...
Copper-catalysed selective hydroamination reactions of alkynes Please share
... In addition to enamine synthesis, we speculated on the possibility that conditions could be developed to convert alkynes to enantioenriched α-branched alkylamines and/or linear alkylamines in one synthetic operation (Fig. 1b, B). Such cascade processes are highly desirable in organic synthesis, sinc ...
... In addition to enamine synthesis, we speculated on the possibility that conditions could be developed to convert alkynes to enantioenriched α-branched alkylamines and/or linear alkylamines in one synthetic operation (Fig. 1b, B). Such cascade processes are highly desirable in organic synthesis, sinc ...
Pincer Complexes. Applications in Catalysis
... of these complexes have shown activity in the dehydrogenation of alkanes to alkenes. However, the extremely low reaction rates and the low turnover numbers or the instability of the employed catalysts under the reaction conditions16 has limited the use of these species. In 1976 Moulton and Shaw repo ...
... of these complexes have shown activity in the dehydrogenation of alkanes to alkenes. However, the extremely low reaction rates and the low turnover numbers or the instability of the employed catalysts under the reaction conditions16 has limited the use of these species. In 1976 Moulton and Shaw repo ...
Chapter 16
... The alkoxide will reform a carbonyl (strong bond) with the good leaving group present ...
... The alkoxide will reform a carbonyl (strong bond) with the good leaving group present ...
Group Meeting Special Topic: EJ Corey
... tetrahydropyran (THP) • More stable silyl ether than TMS and dimethylisopropylsilyl ether • First time used imidazole and DMF ...
... tetrahydropyran (THP) • More stable silyl ether than TMS and dimethylisopropylsilyl ether • First time used imidazole and DMF ...
Efficient hydrogenation of organic carbonates, carbamates and
... NATURE CHEMISTRY | VOL 3 | AUGUST 2011 | www.nature.com/naturechemistry ...
... NATURE CHEMISTRY | VOL 3 | AUGUST 2011 | www.nature.com/naturechemistry ...
fulltext
... In nature such molecules are predominantly existent as one of the possible versions. In chemistry, the common way of denoting the enantiomers are with an (S) (left handed) or (R) (right handed). Amino acids are usually classified under a different nomenclature which allows the majority of the in nat ...
... In nature such molecules are predominantly existent as one of the possible versions. In chemistry, the common way of denoting the enantiomers are with an (S) (left handed) or (R) (right handed). Amino acids are usually classified under a different nomenclature which allows the majority of the in nat ...
Organic Halides (Haloalkanes) (Alkyl Halides)
... (their use is now restricted due to the fact that some of them damage the ozone layer) • polymers such as Teflon (non-stick surfaces as in cookware) and PVC (polyvinylchloride) used plumbing • DDT (dichlorodiphenyltrichloroethane) – now banned pesticide • PCB’s (polychlorinated biphenyls) – electric ...
... (their use is now restricted due to the fact that some of them damage the ozone layer) • polymers such as Teflon (non-stick surfaces as in cookware) and PVC (polyvinylchloride) used plumbing • DDT (dichlorodiphenyltrichloroethane) – now banned pesticide • PCB’s (polychlorinated biphenyls) – electric ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.