中国科学技术大学有机合成讲义-2
... G. J. Am. Chem. Soc. 1965, 87, 5661). Dicyclohexylcarbodiimide (DCC) functions as the electrophilic activating agent in conjunction with a Br?nsted acid promoter. Typically, oxidations are carried out with an excess of DCC at or near 23 °C. Separation of the by-product dicyclohexylurea and MTM ether ...
... G. J. Am. Chem. Soc. 1965, 87, 5661). Dicyclohexylcarbodiimide (DCC) functions as the electrophilic activating agent in conjunction with a Br?nsted acid promoter. Typically, oxidations are carried out with an excess of DCC at or near 23 °C. Separation of the by-product dicyclohexylurea and MTM ether ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... material, the intensity of signals at 1225, 1204, and 1730 cm−1, attributed to C−O (C−OH/C−O−C, hydroxyl/ epoxide) and CO (carbonyl groups), respectively, has significantly decreased. Moreover, complete disappearance of signals at 1413 and 1817 cm−1, attributed to carboxylic acid RCOO-H bending vibr ...
... material, the intensity of signals at 1225, 1204, and 1730 cm−1, attributed to C−O (C−OH/C−O−C, hydroxyl/ epoxide) and CO (carbonyl groups), respectively, has significantly decreased. Moreover, complete disappearance of signals at 1413 and 1817 cm−1, attributed to carboxylic acid RCOO-H bending vibr ...
Oxidation
... electronegative atom than the carbon in question? 3. Give the carbon a +1 oxidation number if to carbon is attached to more electronegative atom than the carbon in question? 4. Give 0 to each other carbon atom attached to the particular carbon. 5. Give 0 to undefined (R) group attached to particular ...
... electronegative atom than the carbon in question? 3. Give the carbon a +1 oxidation number if to carbon is attached to more electronegative atom than the carbon in question? 4. Give 0 to each other carbon atom attached to the particular carbon. 5. Give 0 to undefined (R) group attached to particular ...
Preliminary Screening — Technical and Economic Assessment of Synthesis Gas
... In principle, syngas (primarily consisting of CO and H2) can be produced from any hydrocarbon feedstock, including: natural gas, naphtha, residual oil, petroleum coke, coal, and biomass. The lowest cost routes for syngas production, however, are based on natural gas, the cheapest option being remote ...
... In principle, syngas (primarily consisting of CO and H2) can be produced from any hydrocarbon feedstock, including: natural gas, naphtha, residual oil, petroleum coke, coal, and biomass. The lowest cost routes for syngas production, however, are based on natural gas, the cheapest option being remote ...
Synthesis of a TREN in Which the Aryl Substituents are... Atom Macrocycle ̈ller *
... ligand, (HIPTNHCH2CH2)3N, was present at the end of a catalytic reaction. Protonation of an amido ligand, or strong Hbonding of [LutH]+ to it, was proposed to be the beginning of a process that stripped the ligand off the metal under catalytic conditions and led to irreversible loss of catalytic acti ...
... ligand, (HIPTNHCH2CH2)3N, was present at the end of a catalytic reaction. Protonation of an amido ligand, or strong Hbonding of [LutH]+ to it, was proposed to be the beginning of a process that stripped the ligand off the metal under catalytic conditions and led to irreversible loss of catalytic acti ...
Iodomethylzinc_iodid.. - Groupe Charette
... Reagents derived from ArOZnCH2 I have also been prepared and were found to be quite reactive for the cyclopropanation of alkenes, especially when the aryl group is substituted with halogens.36 The reagent derived from 2,4,6-trichlorophenol converts alkyl- and aryl-substituted alkenes into the corres ...
... Reagents derived from ArOZnCH2 I have also been prepared and were found to be quite reactive for the cyclopropanation of alkenes, especially when the aryl group is substituted with halogens.36 The reagent derived from 2,4,6-trichlorophenol converts alkyl- and aryl-substituted alkenes into the corres ...
Ether - Clayton State University
... For single hydroxy (-OH) group • Step 1: Identify longest chain that includes (-OH) group. Drop –e from hydrocarbon name, and replace with ending –ol. • Step 2: Number this parent chain to give lowest number to carbon with attached (-OH) group. • Step 3: Locate position of (-OH) group. • Step 4: Loc ...
... For single hydroxy (-OH) group • Step 1: Identify longest chain that includes (-OH) group. Drop –e from hydrocarbon name, and replace with ending –ol. • Step 2: Number this parent chain to give lowest number to carbon with attached (-OH) group. • Step 3: Locate position of (-OH) group. • Step 4: Loc ...
Kinetic Assay of Human Pepsin with Albumin
... one-to-one molar ratio. The value of k, the stability constant, was 3.72 x i0, which indicates that very little free dye or albumin is present when the two compounds are allowed to react in unimolar proportions. One of the curves is illustrated in Figure 1. Spectral properties of the complex. In Fig ...
... one-to-one molar ratio. The value of k, the stability constant, was 3.72 x i0, which indicates that very little free dye or albumin is present when the two compounds are allowed to react in unimolar proportions. One of the curves is illustrated in Figure 1. Spectral properties of the complex. In Fig ...
Organometallic Methods for Forming and Cleaving Carbon
... Amanda Sølvhøj for being entertained whenever I was singing or whistling “Barbra Streisand” and I would also like to thank the rest of the Madsen group for a friendly and helpful environment. I appreciate the social and entertaining fellow members of the PhD. ChemClub for the organizing of several e ...
... Amanda Sølvhøj for being entertained whenever I was singing or whistling “Barbra Streisand” and I would also like to thank the rest of the Madsen group for a friendly and helpful environment. I appreciate the social and entertaining fellow members of the PhD. ChemClub for the organizing of several e ...
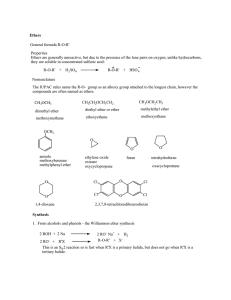
Ethers General formula R-O-R` Properties Ethers are generally
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
OC 2/e Ch 15
... • Step 3: proton transfer from the alcohol to O- gives the hemiacetal and generates a new base catalyst O:– CH3 - C-CH3 + H–OR ...
... • Step 3: proton transfer from the alcohol to O- gives the hemiacetal and generates a new base catalyst O:– CH3 - C-CH3 + H–OR ...
Oxo-molybdenum(VI) - Repositório da Universidade Nova de Lisboa
... Chapter 5 describes the application of the new molybdenum complexes in epoxidation of cyclooctene, (R)-limonene and trans--methylstyrene in organic medium as well in imidazolium- and pyrrolidinium-based ionic liquids. The new molybdenum complexes were found to be efficient catalysts affording high ...
... Chapter 5 describes the application of the new molybdenum complexes in epoxidation of cyclooctene, (R)-limonene and trans--methylstyrene in organic medium as well in imidazolium- and pyrrolidinium-based ionic liquids. The new molybdenum complexes were found to be efficient catalysts affording high ...
Latest Publication (still not complete)
... increased steric hindrance. In the first reported synthesis of such a complex, the alkylation was facilitated through the use of an alkyl halide, typically an iodide or bromide. However, in more recent times, the use of trialkyloxonium tetrafluoroborate salts has become common. These reagents offer ...
... increased steric hindrance. In the first reported synthesis of such a complex, the alkylation was facilitated through the use of an alkyl halide, typically an iodide or bromide. However, in more recent times, the use of trialkyloxonium tetrafluoroborate salts has become common. These reagents offer ...
PDF - Nanyang Technological University
... We used a chiral amine combined with CuBr2 as a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselec ...
... We used a chiral amine combined with CuBr2 as a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselec ...
Esterification and Esters
... hydrogen and hydroxide ions (19,20), a base-catalyzed esterification is not known. A number of mechanisms for acid- and base-catalyzed esterification have been proposed (4). One possible mechanism for the bimolecular acid-catalyzed ester hydrolysis and esterification is shown in equation 2 (6). ...
... hydrogen and hydroxide ions (19,20), a base-catalyzed esterification is not known. A number of mechanisms for acid- and base-catalyzed esterification have been proposed (4). One possible mechanism for the bimolecular acid-catalyzed ester hydrolysis and esterification is shown in equation 2 (6). ...
The Mole
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... TABLE OF CONTENTS List of Tables .......................................................................................................... vii List of Figures ........................................................................................................ viii Abstract .................... ...
... TABLE OF CONTENTS List of Tables .......................................................................................................... vii List of Figures ........................................................................................................ viii Abstract .................... ...
Chapter 1 Chemical Bonding and Chemical Structure
... • Low molecular weight amines tend to be water soluble whether they are primary, secondary or tertiary • Fewer than 5 carbons usually = solubility in water ...
... • Low molecular weight amines tend to be water soluble whether they are primary, secondary or tertiary • Fewer than 5 carbons usually = solubility in water ...
CH221 CLASS 13
... Hydration of Alkenes Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can b ...
... Hydration of Alkenes Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can b ...
12_chemistry_impq_CH10_haloalkanes_and_haloarenes_02
... Q8. Why Grignard reagent should be prepared under an hydrous conditions.? Ans. Grignard reagent react with H2O to form alkanes , therefore they are prepared under anhydrous condition. Q9. Why is Sulphuric acid not used during the reaction of alcohols with KI ? ...
... Q8. Why Grignard reagent should be prepared under an hydrous conditions.? Ans. Grignard reagent react with H2O to form alkanes , therefore they are prepared under anhydrous condition. Q9. Why is Sulphuric acid not used during the reaction of alcohols with KI ? ...
Diastereoselective Allylation of Carbonyl Compounds and Imines:
... crotylboropinacolate, respectively. The reactions should be performed in the presence of a Lewis acid. Thus, the SnCl4 catalyzed addition of allylboropinacolate to the chiral aldehyde gave minimal selectivity (46:54) favoring the syn-diastereomer. However, better diastereoselectivity was obtained wh ...
... crotylboropinacolate, respectively. The reactions should be performed in the presence of a Lewis acid. Thus, the SnCl4 catalyzed addition of allylboropinacolate to the chiral aldehyde gave minimal selectivity (46:54) favoring the syn-diastereomer. However, better diastereoselectivity was obtained wh ...
C 1 hapter
... The catalytic asymmetric hydroboration reaction has proved to be one of the most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide ran ...
... The catalytic asymmetric hydroboration reaction has proved to be one of the most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide ran ...
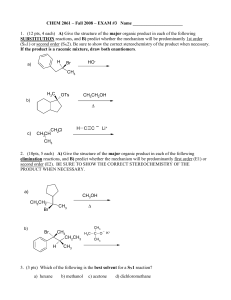
Exam #3
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.