Transition Metal Catalyzed Selective Oxidation of Sugars and Polyols
... The NiBr2 mediated oxidation of alcohols and benzoylperoxide was investigated. The selective oxidation of vicinal diols was unsuccessful with this method. However, the oxidation reaction of mono-alcohol substrates was greatly improved using water in the reaction. The reaction mechanism was investiga ...
... The NiBr2 mediated oxidation of alcohols and benzoylperoxide was investigated. The selective oxidation of vicinal diols was unsuccessful with this method. However, the oxidation reaction of mono-alcohol substrates was greatly improved using water in the reaction. The reaction mechanism was investiga ...
Chapter - FIU Faculty Websites
... • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ester we have to protect ketone group in A as acet ...
... • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ester we have to protect ketone group in A as acet ...
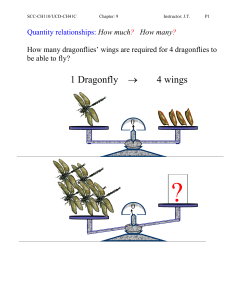
Quantity relationships: How much
... Percent Yield: The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yiel ...
... Percent Yield: The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yiel ...
Acid-Catalyzed Hydration of Alkenes
... Optically inactive reactants produce optically inactive products. (Racemic mixtures are optically inactive) The correlary is that an optically active starting material MAY produce an optically active product depending on the mechanism. ...
... Optically inactive reactants produce optically inactive products. (Racemic mixtures are optically inactive) The correlary is that an optically active starting material MAY produce an optically active product depending on the mechanism. ...
CHAPTER 9 Notes
... c. When 2.50 g of K and 1.00 g Cl2 react together, the mass of KCl produced is _____2.10 g_______________, the limiting reactant is____Cl2____________, and the reactant in excess is _______K__________________. ...
... c. When 2.50 g of K and 1.00 g Cl2 react together, the mass of KCl produced is _____2.10 g_______________, the limiting reactant is____Cl2____________, and the reactant in excess is _______K__________________. ...
Chapter 7. Solubility
... concentration↑ precipitate↑ Light transmission ↓ turning point = solubility ...
... concentration↑ precipitate↑ Light transmission ↓ turning point = solubility ...
Curriculum Vitae
... 106. Deluca, R. J.; Stokes, B. J.; Sigman, M. S. “The strategic generation and interception of palladiumhydrides for use in alkene functionalization reactions,” Pure Appl. Chem. 2014, 86, 395-408. 105. Xu, L.; Hilton, M. J.; Zhang, X.; Norrby, P.-O.*; Wu, Y.-D.*; Sigman, M. S.*; Wiest, O.* “Mechanis ...
... 106. Deluca, R. J.; Stokes, B. J.; Sigman, M. S. “The strategic generation and interception of palladiumhydrides for use in alkene functionalization reactions,” Pure Appl. Chem. 2014, 86, 395-408. 105. Xu, L.; Hilton, M. J.; Zhang, X.; Norrby, P.-O.*; Wu, Y.-D.*; Sigman, M. S.*; Wiest, O.* “Mechanis ...
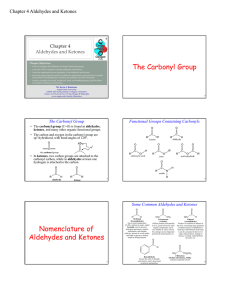
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... • In molecules which have both OH and C=O groups on different carbon atoms, an intramolecular addition reaction can occur, producing a cyclic ...
... • In molecules which have both OH and C=O groups on different carbon atoms, an intramolecular addition reaction can occur, producing a cyclic ...
Alcohols, Phenols and Ethers
... Polarity: Water is a very polar molecule; alcohols and ethers also display polarity. Boiling point: Water has a higher boiling point than predicted on the basis of its molar mass. Alcohols also share this characteristic, but ethers do not. Solubility: Water is miscible with other polar liquids; alco ...
... Polarity: Water is a very polar molecule; alcohols and ethers also display polarity. Boiling point: Water has a higher boiling point than predicted on the basis of its molar mass. Alcohols also share this characteristic, but ethers do not. Solubility: Water is miscible with other polar liquids; alco ...
HIGHLY SELECTIVE RHODIUM–CATALYZED C–H BORYLATIONS IN
... synthesis of new carbon–carbon bonds has increased the demand for efficient routes to organoboron starting materials. C–H borylation (activation) has provided an interesting approach to alleviate the requirement for prefunctionalized molecules such as aryl halides to obtain these desired organoboron ...
... synthesis of new carbon–carbon bonds has increased the demand for efficient routes to organoboron starting materials. C–H borylation (activation) has provided an interesting approach to alleviate the requirement for prefunctionalized molecules such as aryl halides to obtain these desired organoboron ...
On The catalytic Hydrogenation of Co2 and Carboxylic acid esters
... reactions that seemed unfeasible 50 years ago. The majority of substrates are organic compounds that contain a reducible functionality. Depending on their nature, hydrogenation of these functional groups may require different conditions and different types of catalysts. One class of compounds that h ...
... reactions that seemed unfeasible 50 years ago. The majority of substrates are organic compounds that contain a reducible functionality. Depending on their nature, hydrogenation of these functional groups may require different conditions and different types of catalysts. One class of compounds that h ...
Boron and Metal Catalyzed CC and CH Bond Formation
... dialkynylation of aryl aldehydes using dialkynylboron chloride. Numerous applications of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepare ...
... dialkynylation of aryl aldehydes using dialkynylboron chloride. Numerous applications of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepare ...
Palladium(II)-Catalyzed Oxidative Cyclization Strategies Andreas K. Å. Persson
... The use of transition metal catalysts has proven to be one of the most diverse tools for the mild and selective formation of carbon-carbon bonds. In particular palladium-catalyzed cross-coupling reactions have revolutionized the field. The main focus of this thesis has been directed towards preparat ...
... The use of transition metal catalysts has proven to be one of the most diverse tools for the mild and selective formation of carbon-carbon bonds. In particular palladium-catalyzed cross-coupling reactions have revolutionized the field. The main focus of this thesis has been directed towards preparat ...
couverture these PRES Toulouse M ESCARCEGA 2011
... activity and mainly the selectivity induced, however their separation from organic products and their recycling remain often troublesome.6,9 In 1980s, Schwartz pointed an expanded and better consideration for defining a catalysts as “homogeneous”, relating it to its possessing only a single type of ...
... activity and mainly the selectivity induced, however their separation from organic products and their recycling remain often troublesome.6,9 In 1980s, Schwartz pointed an expanded and better consideration for defining a catalysts as “homogeneous”, relating it to its possessing only a single type of ...
Iodine and Lipase Based Green Oxidation Technology
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
STOICHIOMETRY via ChemLog - Small
... Determining the mass percent composition of a compound refers to the proportion of one element expressed as a percentage of the total mass of the compound. Knowing the mass percent composition of a compound can help determine environmental effects from that compound. For example, carbon dioxide (CO2 ...
... Determining the mass percent composition of a compound refers to the proportion of one element expressed as a percentage of the total mass of the compound. Knowing the mass percent composition of a compound can help determine environmental effects from that compound. For example, carbon dioxide (CO2 ...
Document
... of sodium iodide under solvent free conditions. Eur. J. Org. Chem. 1999, 617. 21. S. Alessandrini; G. Bartoli; M. C. Bellucci; M. Bosco; M. Malavolta; E. Marcantoni; L. Sambri: Reversed stereochemical control in the presence of CeCl3 and TiCl4 in the Lewis acid mediated reduction of α-alkyl-β-keto e ...
... of sodium iodide under solvent free conditions. Eur. J. Org. Chem. 1999, 617. 21. S. Alessandrini; G. Bartoli; M. C. Bellucci; M. Bosco; M. Malavolta; E. Marcantoni; L. Sambri: Reversed stereochemical control in the presence of CeCl3 and TiCl4 in the Lewis acid mediated reduction of α-alkyl-β-keto e ...
File
... less than what was expected. They identify and list the most likely errors in the activity. Which is a systematic error? A. B. C. D. ...
... less than what was expected. They identify and list the most likely errors in the activity. Which is a systematic error? A. B. C. D. ...
14A
... – they are so susceptible to oxidation that they must be protected from contact with air during storage – disulfides, in turn, are easily reduced to thiols by several reducing agents. ...
... – they are so susceptible to oxidation that they must be protected from contact with air during storage – disulfides, in turn, are easily reduced to thiols by several reducing agents. ...
Chapter Seven PPT
... • Platinum Catalysts Allow Double Addition of H2 On Alkyne • Can Also Hydrogenate Once to Generate Alkenes • Cis and Trans (E and Z) Stereoisomers are Possible • Can Control Stereochemistry with Catalyst Selection ...
... • Platinum Catalysts Allow Double Addition of H2 On Alkyne • Can Also Hydrogenate Once to Generate Alkenes • Cis and Trans (E and Z) Stereoisomers are Possible • Can Control Stereochemistry with Catalyst Selection ...
Fatty Acids and Derivatives from Coconut Oil
... and direct sulfation because coconut oil lacks the necessary unsaturation to initiate the two reactions. As can be seen from Figure 1, a great number of oleochemicals can be derived from natural fats and oils. These olechemicals find increasing use in various applications. 3. TYPES OF FATTY ACIDS AN ...
... and direct sulfation because coconut oil lacks the necessary unsaturation to initiate the two reactions. As can be seen from Figure 1, a great number of oleochemicals can be derived from natural fats and oils. These olechemicals find increasing use in various applications. 3. TYPES OF FATTY ACIDS AN ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... Partial credit will be given when/where appropriate. Brief answers are most welcome, citing specific experimental support from text or below. Long, rambling answers are not necessary! Note: There is no particular order to questions presented here. Answer first those questions you know best. Partial ...
... Partial credit will be given when/where appropriate. Brief answers are most welcome, citing specific experimental support from text or below. Long, rambling answers are not necessary! Note: There is no particular order to questions presented here. Answer first those questions you know best. Partial ...
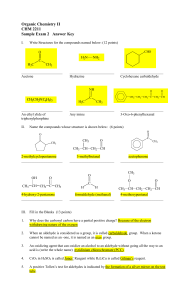
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... IV. Write a multi step reaction mechanism for the reaction of phenylmagnesium bromide ( MgBr) with acetone to form an alcohol. Indicate intermediates that may or may not be isolated. Use curved arrows to indicate electron attack and show charges and intermediates. (9 points) ...
... IV. Write a multi step reaction mechanism for the reaction of phenylmagnesium bromide ( MgBr) with acetone to form an alcohol. Indicate intermediates that may or may not be isolated. Use curved arrows to indicate electron attack and show charges and intermediates. (9 points) ...
Learning Guide for Chapter 23: Amines
... VII. Reactions with acid chlorides - p 11 reduction of amides VIII. Reduction of other groups to form amines - p 12 IX. Reactivity of anilines - p 15 aromatic substutition, use of diazonium ions I. Introduction to amines Amines are considered to be organic derivatives of ammonia. They are classified ...
... VII. Reactions with acid chlorides - p 11 reduction of amides VIII. Reduction of other groups to form amines - p 12 IX. Reactivity of anilines - p 15 aromatic substutition, use of diazonium ions I. Introduction to amines Amines are considered to be organic derivatives of ammonia. They are classified ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.