Cookies and Chemistry…Huh!?!?
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
Chapter+12
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... 2 cups semisweet chocolate chips Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
Heterogeneous Catalysis and Solid Catalysts
... the catalyst or a reactant during the reaction. This can take place in a homogeneous or heterogeneous system. One example is the utilization of semiconductor catalysts (titanium, zinc, and iron oxides) for photochemical degradation of organic substances, e.g., on selfcleaning surfaces. In biocatalys ...
... the catalyst or a reactant during the reaction. This can take place in a homogeneous or heterogeneous system. One example is the utilization of semiconductor catalysts (titanium, zinc, and iron oxides) for photochemical degradation of organic substances, e.g., on selfcleaning surfaces. In biocatalys ...
Lorell Thesis Final Version in PDF S
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
1H-Imidazol-4(5H)-ones and thiazol-4(5H)
... prior trimethylsilyl enol ether formation which is necessary to avoid O-alkylation. The starting thiohydantoins, in turn, are obtained by heating a mixture of the corresponding N-substituted amino acid or its methyl ester 3 and thiourea at 195 °C [59] (Scheme 1b). Following this protocol different 1 ...
... prior trimethylsilyl enol ether formation which is necessary to avoid O-alkylation. The starting thiohydantoins, in turn, are obtained by heating a mixture of the corresponding N-substituted amino acid or its methyl ester 3 and thiourea at 195 °C [59] (Scheme 1b). Following this protocol different 1 ...
Project Overview
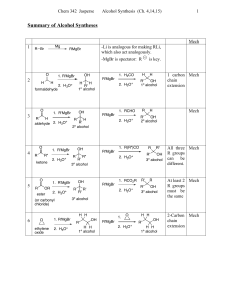
... ● Tertiary alcohols are usually so easily dehydrated that extremely mild conditions can be used. tert-Butyl alcohol, for example, dehydrates in 20% aqueous sulfuric acid at a temperature of 85°C ...
... ● Tertiary alcohols are usually so easily dehydrated that extremely mild conditions can be used. tert-Butyl alcohol, for example, dehydrates in 20% aqueous sulfuric acid at a temperature of 85°C ...
2 - cloudfront.net
... run out of first. Determines how much product you can make! 2. The excess reactant is the one you have left over. • Often referred to as limiting reagent or excess reagent ...
... run out of first. Determines how much product you can make! 2. The excess reactant is the one you have left over. • Often referred to as limiting reagent or excess reagent ...
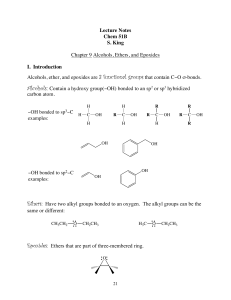
Lecture Notes Chem 51B S. King Chapter 9 Alcohols, Ethers, and
... alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make it a better LG: ...
... alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make it a better LG: ...
Title Several Reactions of Isocyanide and Related Compounds
... strong coordinations of these special alcohols with CuCl have been demonstrated also by IR spectroscopy. In Chapter 3, reactions of thiols and isocyanide have been investigated. The reaction of thiol with isocyanide proceeds in two directions. ...
... strong coordinations of these special alcohols with CuCl have been demonstrated also by IR spectroscopy. In Chapter 3, reactions of thiols and isocyanide have been investigated. The reaction of thiol with isocyanide proceeds in two directions. ...
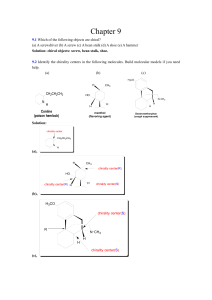
Chapter 9 - ComSizo.com.br
... The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is not equally likely, so an unequal mix of (2S,3R) or (2R,3S) ...
... The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is not equally likely, so an unequal mix of (2S,3R) or (2R,3S) ...
Module I Oxidation Reactions
... under stoichiometric conditions have been shown that imidotrioxo osmium(VIII) complexes transfer the nitrogen atom and one of the oxygen atoms into the substrate. The major regioisomer normally has the nitrogen placed distal to the most electron withdrawing group of the substrate. ...
... under stoichiometric conditions have been shown that imidotrioxo osmium(VIII) complexes transfer the nitrogen atom and one of the oxygen atoms into the substrate. The major regioisomer normally has the nitrogen placed distal to the most electron withdrawing group of the substrate. ...
Full-Text PDF
... relevant ligand features in catalysts for alkene epoxidation are corroles (3, structurally similar to porphyrins) [12] and bis-amino-phenolate ligands (4, structurally related to salen) [13], which are also represented in Figure 1. The prominence of porphyrin and salen scaffolds in the design of Mn ...
... relevant ligand features in catalysts for alkene epoxidation are corroles (3, structurally similar to porphyrins) [12] and bis-amino-phenolate ligands (4, structurally related to salen) [13], which are also represented in Figure 1. The prominence of porphyrin and salen scaffolds in the design of Mn ...
Chiral Enolate Equivalents
... Despite this impressive track record, the weak reactivity of enolates in general confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophile ...
... Despite this impressive track record, the weak reactivity of enolates in general confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophile ...
Class Notes
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
- University of Bath Opus
... sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs for patient safety. It follows that any methodology that avoids the need to convert alcohols to halides or sulfonates will offer significant benefits to both ...
... sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs for patient safety. It follows that any methodology that avoids the need to convert alcohols to halides or sulfonates will offer significant benefits to both ...
Alcohols, Phenols, Thiols and Ethers
... Learning Check Select the compound that would result for each reaction of CH3—CH2—CH2—OH. 1) CH3—CH=CH2 A. H+, heat ...
... Learning Check Select the compound that would result for each reaction of CH3—CH2—CH2—OH. 1) CH3—CH=CH2 A. H+, heat ...
Chapter 13
... Solubility of Alcohols and Ethers in Water Alcohols and ethers are more soluble in water than alkanes because the oxygen atom hydrogen bonds with water. Alcohols with 1-4 C atoms are soluble, but alcohols with 5 or more C atoms are not. ...
... Solubility of Alcohols and Ethers in Water Alcohols and ethers are more soluble in water than alkanes because the oxygen atom hydrogen bonds with water. Alcohols with 1-4 C atoms are soluble, but alcohols with 5 or more C atoms are not. ...
Process for the preparation of ethene and/or propene
... catalyst used for the first stage is a ZSM-5 containing zeolite catalyst. The second stage catalyst is a 10membered ring zeolite, and ZSM-22, ZSM-23, ZSM-35, ZSM-48 are mentioned. ZSM-35 is preferred. Various embodiments of reaction systems with first and second stage catalyst ...
... catalyst used for the first stage is a ZSM-5 containing zeolite catalyst. The second stage catalyst is a 10membered ring zeolite, and ZSM-22, ZSM-23, ZSM-35, ZSM-48 are mentioned. ZSM-35 is preferred. Various embodiments of reaction systems with first and second stage catalyst ...
The Carbonyl Group - Angelo State University
... original aldehyde or ketone from the acetal or ketal. • This is an example of a hydrolysis reaction, in which water causes a compound to be split into its ...
... original aldehyde or ketone from the acetal or ketal. • This is an example of a hydrolysis reaction, in which water causes a compound to be split into its ...
Ch14_PT MULTIPLE CHOICE. Choose the one alternative that best
... B C Alcohols and ethers can be considered to be derivatives of water. Similar physical properties include: Polarity: Water is a very polar molecule; alcohols and ethers also display polarity. Boiling point: Water has a higher boiling point than predicted on the basis of its molar mass. Alcohols also ...
... B C Alcohols and ethers can be considered to be derivatives of water. Similar physical properties include: Polarity: Water is a very polar molecule; alcohols and ethers also display polarity. Boiling point: Water has a higher boiling point than predicted on the basis of its molar mass. Alcohols also ...
Drawing Organic Structures Functional Groups Constitutional Isomers
... I. Nomenclature (Review) 1. Parent chain is longest containing C bonded to –N 2. Change suffix “-e” to “-amine” 3. Number from end closest to –N. Show location of –N. 4. Name/number substituents ...
... I. Nomenclature (Review) 1. Parent chain is longest containing C bonded to –N 2. Change suffix “-e” to “-amine” 3. Number from end closest to –N. Show location of –N. 4. Name/number substituents ...
R - Evans - Harvard University
... demonstrated good levels of diastereoselection in asymmetric enolate a l k y l a t i ~ nacylation,6 ...
... demonstrated good levels of diastereoselection in asymmetric enolate a l k y l a t i ~ nacylation,6 ...
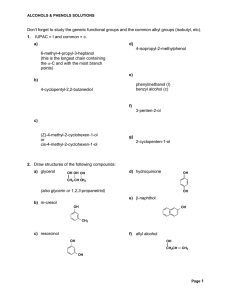
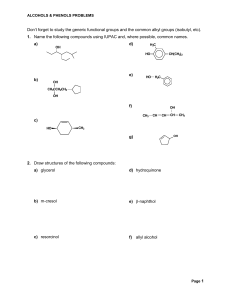
Don`t forget to study the generic functional groups and the common
... 3. Give IUPAC names and, where possible, common names for the following compounds. a) ...
... 3. Give IUPAC names and, where possible, common names for the following compounds. a) ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.