
Microsoft Word
... Enantioselective Synthesis of Tamiflu In this chapter a brief account of the synthesis of tamiflu by selected research groups is described followed by and an elaborate account of the present work. Oseltamivir phosphate (Tamiflu) 85, is an approved orally effective neuraminidase inhibitor used for th ...
... Enantioselective Synthesis of Tamiflu In this chapter a brief account of the synthesis of tamiflu by selected research groups is described followed by and an elaborate account of the present work. Oseltamivir phosphate (Tamiflu) 85, is an approved orally effective neuraminidase inhibitor used for th ...
Converting Glycerol to Higher Value Products
... This time, a higher percentage of alcohols was produced within minutes of starting the pressure and temperature treatment. The alcohols included propylene glycol, ethanol, acetol, and propanol. Raney nickel catalyst method Of all the catalysts tried, the Raney nickel catalyst worked the best. It con ...
... This time, a higher percentage of alcohols was produced within minutes of starting the pressure and temperature treatment. The alcohols included propylene glycol, ethanol, acetol, and propanol. Raney nickel catalyst method Of all the catalysts tried, the Raney nickel catalyst worked the best. It con ...
Naming Aldehydes & Ketones
... Cu2+ ions in an alkaline medium. • In these tests, the aldehyde group is oxidized to an acid by Cu2+ ions. O C R ...
... Cu2+ ions in an alkaline medium. • In these tests, the aldehyde group is oxidized to an acid by Cu2+ ions. O C R ...
Document
... The carbonyl group is strongly polar but does not produce hydrogen bonding. As a result, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols. Formaldehyde is a gas at room temperature (b.p. = -21 C) but heavier ...
... The carbonyl group is strongly polar but does not produce hydrogen bonding. As a result, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols. Formaldehyde is a gas at room temperature (b.p. = -21 C) but heavier ...
98 pts
... • (T) All E1 reactions involve formation of carbocations; • (T) More stable carbocations are generated faster; • (T) Carbocations are electrophiles; • (T) Carbocations are electron deficient; • (T) Free radicals are electron deficient; • (T) Alcohols are Brønsted bases; • (F) The rate-determining st ...
... • (T) All E1 reactions involve formation of carbocations; • (T) More stable carbocations are generated faster; • (T) Carbocations are electrophiles; • (T) Carbocations are electron deficient; • (T) Free radicals are electron deficient; • (T) Alcohols are Brønsted bases; • (F) The rate-determining st ...
Chem 240 - Napa Valley College
... between phenyl magnesium bromide and benzaldehyde. To make sure his yield was good he added twice as much benzaldehyde as Grignard reagent and got a lot of white crystalline product. When he analyzed his product he found that he had not made diphenyl methanol, but diphenyl methanal (also called benz ...
... between phenyl magnesium bromide and benzaldehyde. To make sure his yield was good he added twice as much benzaldehyde as Grignard reagent and got a lot of white crystalline product. When he analyzed his product he found that he had not made diphenyl methanol, but diphenyl methanal (also called benz ...
Catalytic Nucleophilic Fluorination of Secondary and Tertiary
... CuCl instead of [(IPr)CuCl] [(IMes)CuCl] instead of [(IPr)CuCl] [(IPent)CuCl] instead of [(IPr)CuCl] [(SIPr)CuCl] instead of [(IPr)CuCl] CuOTf·0.5 PhH, L1 (20 mol %) CuOTf·0.5 PhH, L2 (20 mol %) 5 mol % instead of 10 mol % [(IPr)CuCl] 60 8C instead of 30 8C [(IPr)CuF] instead of [(IPr)CuCl] [(IPr)Cu ...
... CuCl instead of [(IPr)CuCl] [(IMes)CuCl] instead of [(IPr)CuCl] [(IPent)CuCl] instead of [(IPr)CuCl] [(SIPr)CuCl] instead of [(IPr)CuCl] CuOTf·0.5 PhH, L1 (20 mol %) CuOTf·0.5 PhH, L2 (20 mol %) 5 mol % instead of 10 mol % [(IPr)CuCl] 60 8C instead of 30 8C [(IPr)CuF] instead of [(IPr)CuCl] [(IPr)Cu ...
Chapter 15
... added and one of the oxygens is lost as HO- or CH3O- to give a primary alcohol. Since aldehydes are more easily reduced (we will learn why in a later chapter) than esters or carboxylic acids, we cannot isolate the aldehyde intermediate. ...
... added and one of the oxygens is lost as HO- or CH3O- to give a primary alcohol. Since aldehydes are more easily reduced (we will learn why in a later chapter) than esters or carboxylic acids, we cannot isolate the aldehyde intermediate. ...
Exam 3 - Napa Valley College
... mean that you would get a lot of by-products but you would end up getting more product also (SN1 major, E1 minor). 4) There are a number of ways of substituting a halogen for an alcohol group, but some ways are better than others. What advantage is there in using PCl3 rather than HCl in the chloride ...
... mean that you would get a lot of by-products but you would end up getting more product also (SN1 major, E1 minor). 4) There are a number of ways of substituting a halogen for an alcohol group, but some ways are better than others. What advantage is there in using PCl3 rather than HCl in the chloride ...
Chapter 3 Alcohols, Phenols, and Ethers
... Where Does the Word “Alcohol” Come From? • The word “alcohol” comes from the Arabic term al kohl meaning “the fine powder.” Originally, this referred to an antimony sulfide (Sb2S3)compound used for eye shadow, which was ground up to form a fine powder, but then later came to refer to any finely divi ...
... Where Does the Word “Alcohol” Come From? • The word “alcohol” comes from the Arabic term al kohl meaning “the fine powder.” Originally, this referred to an antimony sulfide (Sb2S3)compound used for eye shadow, which was ground up to form a fine powder, but then later came to refer to any finely divi ...
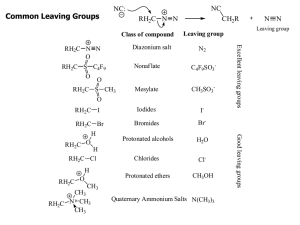
Common Leaving Groups
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
Get PDF - Wiley Online Library
... of the TMS-enol ether of 2 (TMSOTf, Et3N), cleanly provided the C10 tertiary alcohol (18, 99 %). This was then utilized to set the adjacent C11 alcohol stereocenter through hydroxydirected reduction with Me4NBH(OAc)3 (87 %, > 19:1 d.r.).[22] TES protection proved necessary to facilitate the subseque ...
... of the TMS-enol ether of 2 (TMSOTf, Et3N), cleanly provided the C10 tertiary alcohol (18, 99 %). This was then utilized to set the adjacent C11 alcohol stereocenter through hydroxydirected reduction with Me4NBH(OAc)3 (87 %, > 19:1 d.r.).[22] TES protection proved necessary to facilitate the subseque ...
The First Chiral Organometallic Triangle for Asymmetric Catalysis
... two minor bands due to the other two lower energy π f π* transitions (Figure 1). CD spectra of 1-4 exhibited these three bands similar to L1-4, but with red-shifts in energy and higher intensities. Interestingly, a new intense CD band appeared at ∼202 nm, assignable to the transitions associated wit ...
... two minor bands due to the other two lower energy π f π* transitions (Figure 1). CD spectra of 1-4 exhibited these three bands similar to L1-4, but with red-shifts in energy and higher intensities. Interestingly, a new intense CD band appeared at ∼202 nm, assignable to the transitions associated wit ...
13.1 Alcohols and Phenols 13.1 Alcohols and Phenols 13.1
... With an ester substrate, excess Grignard reagent is required. WHY? Propose a mechanism. List some functional groups that are NOT compatible with the Grignard. Practice with SKILLBUILDER 13.5. ...
... With an ester substrate, excess Grignard reagent is required. WHY? Propose a mechanism. List some functional groups that are NOT compatible with the Grignard. Practice with SKILLBUILDER 13.5. ...
ether - HCC Southeast Commons
... Practice Problem: Why do you suppose only symmetrical ethers are prepared by the sulfuric acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal rea ...
... Practice Problem: Why do you suppose only symmetrical ethers are prepared by the sulfuric acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal rea ...
Sample Exam #2 Answer Key
... Alcohols are very polar compared to thiols due to the greater electronegativity of the oxygen in these molecules. Alcohols also exhibit good hydrogen bonding properties while thiols are incapable of hydrogen bonding. Consequently, alcohols have comparatively higher melting points, boiling points and ...
... Alcohols are very polar compared to thiols due to the greater electronegativity of the oxygen in these molecules. Alcohols also exhibit good hydrogen bonding properties while thiols are incapable of hydrogen bonding. Consequently, alcohols have comparatively higher melting points, boiling points and ...
A Simple and Advantageous Protocol for the Oxidation of Alcohols
... carbonyl compound in >90% yield and substantially free of impurities by 1H NMR and TLC analysis (Table 2) with two exceptions.10 The first exception was benzyl alcohol (Table 2, entry 2) which, for reasons still under investigation, was quantitatively oxidized to benzoic acid under our reaction cond ...
... carbonyl compound in >90% yield and substantially free of impurities by 1H NMR and TLC analysis (Table 2) with two exceptions.10 The first exception was benzyl alcohol (Table 2, entry 2) which, for reasons still under investigation, was quantitatively oxidized to benzoic acid under our reaction cond ...
Mock Exam One
... a.) LiAlH4 and a Ketone b.) CH3CH2MgBr and an Aldehyde c.) 2-butene and Hg(OAc)2, H2O followed by NaBH4 d.) All of these. 3.) Which of the following terms best describes the reactive nature of the Grignard ...
... a.) LiAlH4 and a Ketone b.) CH3CH2MgBr and an Aldehyde c.) 2-butene and Hg(OAc)2, H2O followed by NaBH4 d.) All of these. 3.) Which of the following terms best describes the reactive nature of the Grignard ...
06. Alcohols. Phenols. Ethers
... Тhe alkyl alcohol system. In this system of common nomenclature, the name of an alcohol is derived by combining the name of the alkyl group with the word alcohol. The names are mitten as two words. ...
... Тhe alkyl alcohol system. In this system of common nomenclature, the name of an alcohol is derived by combining the name of the alkyl group with the word alcohol. The names are mitten as two words. ...
T_AllylCF3paperBM[5]
... molecules of 1a to form two new C-C bonds. Among the Lewis acids (Entries 6-22) the highest yield of the compound 2a was achieved for anhydrous FeCl3. In this case alcohol 1a gave E-2a as the only reaction product with excess of benzene (Entry 6). Reaction of equimolar amounts of 1a and benzene affo ...
... molecules of 1a to form two new C-C bonds. Among the Lewis acids (Entries 6-22) the highest yield of the compound 2a was achieved for anhydrous FeCl3. In this case alcohol 1a gave E-2a as the only reaction product with excess of benzene (Entry 6). Reaction of equimolar amounts of 1a and benzene affo ...
Q4) How the following conversions can be carried out?
... are soluble in aqueous solution of sodium carbonate? ANS 2, 4-Dinitrophenol and 2, 4, 6-trinitrophenol are stronger acids then carbonic acid (H2CO3) due to the presence of electron withdrawing – NO2 groups. Hence, they react with Na2CO3 to form their corresponding salts and dissolve in aq. Na2CO3 so ...
... are soluble in aqueous solution of sodium carbonate? ANS 2, 4-Dinitrophenol and 2, 4, 6-trinitrophenol are stronger acids then carbonic acid (H2CO3) due to the presence of electron withdrawing – NO2 groups. Hence, they react with Na2CO3 to form their corresponding salts and dissolve in aq. Na2CO3 so ...
Alcohols, Phenols, and Thiols
... Alcohols are comparable in acidity to water, but phenols are much more acidic. This increased acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxid ...
... Alcohols are comparable in acidity to water, but phenols are much more acidic. This increased acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxid ...
$doc.title
... Ether Complexes • Grignard reagents: ComplexaHon of an ether with a Grignard reagent stabilizes the reagent and helps keep it in soluHon. ...
... Ether Complexes • Grignard reagents: ComplexaHon of an ether with a Grignard reagent stabilizes the reagent and helps keep it in soluHon. ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.

















![T_AllylCF3paperBM[5]](http://s1.studyres.com/store/data/003584459_1-3decab572f7fca68901a941affab18ea-300x300.png)



