Postprint
... compounds containing tetra substituted double bonds.7 The latter had displayed very poor rates of reaction with most platinum group catalysts of the time. Replacing two small mono-dentate ligands by a larger chiral bi-dentate one opens the possibility for asymmetric hydrogenation, as the chirality c ...
... compounds containing tetra substituted double bonds.7 The latter had displayed very poor rates of reaction with most platinum group catalysts of the time. Replacing two small mono-dentate ligands by a larger chiral bi-dentate one opens the possibility for asymmetric hydrogenation, as the chirality c ...
electrical energy and capacitance
... Example 1. A compound is discovered with a 58.12 g/mol molar mass. Its empirical formula is C2H5. What is the molecular formula of this compound? 1A. (1) C = 12.01 amu (2) H = 1.01 amu (3) C2 + H5 (4) C2H5 = 2(12.01 amu) + 5(1.01 amu) (5) EF = C2H5 = 29.07 g/mol (6) MF = 58.12 g/mol (7) MF = n(EF) ( ...
... Example 1. A compound is discovered with a 58.12 g/mol molar mass. Its empirical formula is C2H5. What is the molecular formula of this compound? 1A. (1) C = 12.01 amu (2) H = 1.01 amu (3) C2 + H5 (4) C2H5 = 2(12.01 amu) + 5(1.01 amu) (5) EF = C2H5 = 29.07 g/mol (6) MF = 58.12 g/mol (7) MF = n(EF) ( ...
Organic synthesis and methodology related to the malaria drug artemisinin
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
Ethers and Epoxides
... Since ethers are relatively unreactive and are somewhat polar (due to the lone pairs on the oxygen), they are commonly used as solvents for organic reactions. (Diethyl ether and THF, the Grignard reaction). Ethers will often form complexes with molecules that have vacant orbitals, enabling ‘unstable ...
... Since ethers are relatively unreactive and are somewhat polar (due to the lone pairs on the oxygen), they are commonly used as solvents for organic reactions. (Diethyl ether and THF, the Grignard reaction). Ethers will often form complexes with molecules that have vacant orbitals, enabling ‘unstable ...
Protection (and Deprotection) of Functional Groups in Organic
... liquid phase and frequently involving protectiondeprotection steps. The use of blocking functions in organic synthesis, developed for nearly 100 years, makes more complex the entire synthetic plan since it requires at least two additional steps. At the same time, environmental and economic considera ...
... liquid phase and frequently involving protectiondeprotection steps. The use of blocking functions in organic synthesis, developed for nearly 100 years, makes more complex the entire synthetic plan since it requires at least two additional steps. At the same time, environmental and economic considera ...
© John Congleton, Orange Coast College Organic Chemistry 220
... • Why is bromination so much more selective? • Allylic bromination • What is the Hammond Postulate o What does the transition state have to do with this postulate? • Be able to determine chlorination and bromination products taking stereochemistry into consideration Chapter 5 • Definitions: o Chiral ...
... • Why is bromination so much more selective? • Allylic bromination • What is the Hammond Postulate o What does the transition state have to do with this postulate? • Be able to determine chlorination and bromination products taking stereochemistry into consideration Chapter 5 • Definitions: o Chiral ...
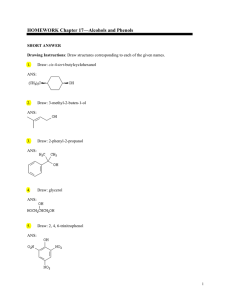
HOMEWORK Chapter 17—Alcohols and Phenols
... Refer to Exhibit 17-6. The electrophile in this reaction is: ...
... Refer to Exhibit 17-6. The electrophile in this reaction is: ...
Sample Midterm 1B
... 2. a. (1 Mark) Butanol, C4H9OH, Diethyl Ether CH3CH2OCH2CH3 and Pentane C5H12 all have similar molecular weights but Butanol has a much higher boiling point. What property of butanol would explain this boiling point difference? ...
... 2. a. (1 Mark) Butanol, C4H9OH, Diethyl Ether CH3CH2OCH2CH3 and Pentane C5H12 all have similar molecular weights but Butanol has a much higher boiling point. What property of butanol would explain this boiling point difference? ...
Development of New Synthetic Routes to Organoboronates by Catalytic Allylic Substitution and
... To access simple boronates such as allyl (R1=H, R2=H, Scheme 9) and crotyl (R1=Me, R2=H, Scheme 9) species, this method has the advantage of using readily available and inexpensive starting materials. However, these reactive species are known to undergo facile metallotropic rearrangements,109-111 wh ...
... To access simple boronates such as allyl (R1=H, R2=H, Scheme 9) and crotyl (R1=Me, R2=H, Scheme 9) species, this method has the advantage of using readily available and inexpensive starting materials. However, these reactive species are known to undergo facile metallotropic rearrangements,109-111 wh ...
Reduction of Aldehydes and Ketones to Corresponding
... functional group is of great importance in organic chemistry and there are numerous applications in fine chemical industry and laboratory1. A large number of methods including catalytic reduction2, Meerwin-ponndorff-verley reduction2, hydride transfer reagents3, homogeneous catalysis4-6 and heteroge ...
... functional group is of great importance in organic chemistry and there are numerous applications in fine chemical industry and laboratory1. A large number of methods including catalytic reduction2, Meerwin-ponndorff-verley reduction2, hydride transfer reagents3, homogeneous catalysis4-6 and heteroge ...
Experimental and Computational Evidence of Metal‑O2 Activation
... oxidative transformations, oxygen isotope fractionation from natural abundance levels provides a potential solution. Competitive oxygen-18 kinetic isotope effects (18O KIEs) have been determined on a number of stoichiometric and catalytic reactions.36−47 These probes are particularly useful when comb ...
... oxidative transformations, oxygen isotope fractionation from natural abundance levels provides a potential solution. Competitive oxygen-18 kinetic isotope effects (18O KIEs) have been determined on a number of stoichiometric and catalytic reactions.36−47 These probes are particularly useful when comb ...
Conjugate addition_Clayden
... Conjugate addition of alcohols can be catalysed by acid or base Alcohols undergo conjugate addition only very slowly in the absence of a catalyst: they are not such good nucleophiles as amines for the very reason we have just mentioned in connection with the reactivity of hydroxylamine—oxygen is mor ...
... Conjugate addition of alcohols can be catalysed by acid or base Alcohols undergo conjugate addition only very slowly in the absence of a catalyst: they are not such good nucleophiles as amines for the very reason we have just mentioned in connection with the reactivity of hydroxylamine—oxygen is mor ...
Chapter 7. Alcohols, Thiols, Phenols, Ethers
... CH3CH2CH2COOH, CH3CH2CH2CH2OH H2SO4 Secondary alcohols are easily converted to ketones. Further oxidation of the ketone occurs only under very harsh conditions. The mechanism of the oxidation involves formation of a chromate (VI) ester of the alcohol followed by loss of chromous (IV) acid. Tertiary ...
... CH3CH2CH2COOH, CH3CH2CH2CH2OH H2SO4 Secondary alcohols are easily converted to ketones. Further oxidation of the ketone occurs only under very harsh conditions. The mechanism of the oxidation involves formation of a chromate (VI) ester of the alcohol followed by loss of chromous (IV) acid. Tertiary ...
Ring-Opening Metathesis Polymerization of Norbornene by Cp
... J. J. J. Am. Chem. Soc. 1987, 109, 899-901. (35) Benedicto, A. D.; Novak, B. M.; Grubbs, R. H. Macromolecules 1992, 25, 5893-5900, and references therein. (36) France, M. B.; Grubbs, R. H.; McGrath, D. V.; Paciello, R. A. ...
... J. J. J. Am. Chem. Soc. 1987, 109, 899-901. (35) Benedicto, A. D.; Novak, B. M.; Grubbs, R. H. Macromolecules 1992, 25, 5893-5900, and references therein. (36) France, M. B.; Grubbs, R. H.; McGrath, D. V.; Paciello, R. A. ...
13_lecture_ppt
... Formation • The aldehyde and alcohol functional groups can both be parts of the same molecule in an intramolecular reaction ...
... Formation • The aldehyde and alcohol functional groups can both be parts of the same molecule in an intramolecular reaction ...
CHEM 121 Chapter 14. Name: Date: ______ 1. The simplest alcohol
... C) absolute alcohol is pure methanol D) absolute ethanol has all water removed from it 5. In which of the following pairs of alcohols do both members of the pair contain two or more hydroxyl groups? A) s-butanol and ethylene glycol B) propylene glycol and glycerol C) t-butyl alcohol and ethylene gly ...
... C) absolute alcohol is pure methanol D) absolute ethanol has all water removed from it 5. In which of the following pairs of alcohols do both members of the pair contain two or more hydroxyl groups? A) s-butanol and ethylene glycol B) propylene glycol and glycerol C) t-butyl alcohol and ethylene gly ...
Stereoselective Construction of a β
... (8) (a) Zhu, L.; Wehmeyer, R. M.; Rieke, R. D. J. Org. Chem. 1991, 56, 1445-1453. (b) Biller, S. A.; Abt, J. W.; Pudzianowski, A. T.; Rich, L. C.; Slusarchyk, D. A.; Ciosek, C. P., Jr. Bioorg. Med. Chem. Lett. 1993, 3, 595-600. (c) Tamaru, Y.; Ochiai, H.; Nakamura, T.; Yoshida, Z. Angew. Chem., Int. ...
... (8) (a) Zhu, L.; Wehmeyer, R. M.; Rieke, R. D. J. Org. Chem. 1991, 56, 1445-1453. (b) Biller, S. A.; Abt, J. W.; Pudzianowski, A. T.; Rich, L. C.; Slusarchyk, D. A.; Ciosek, C. P., Jr. Bioorg. Med. Chem. Lett. 1993, 3, 595-600. (c) Tamaru, Y.; Ochiai, H.; Nakamura, T.; Yoshida, Z. Angew. Chem., Int. ...
View/Open - AURA - Alfred University
... benzyl alcohol (35) and diphenylmethanol with a variety of nitriles. As previously mentioned, understanding the mechanism of PEDOT-mediated reactions is not only synthetically important, but may also lead to the production of novel polymer reagents at lower costs than PEDOT. The reactions of triphen ...
... benzyl alcohol (35) and diphenylmethanol with a variety of nitriles. As previously mentioned, understanding the mechanism of PEDOT-mediated reactions is not only synthetically important, but may also lead to the production of novel polymer reagents at lower costs than PEDOT. The reactions of triphen ...
A Practical RuCl3-Catalyzed Oxidation Using Trichloroisocyanuric
... toxic chromium(VI) species. Corey’s pyridinium dichromate (PDC) in DMF4b could have been used more frequently to oxidize primary alcohols 1 to carboxylic acids 3 because of the neutral nature of the reaction (Scheme 1). However, besides the hazardous nature of the chromium species in PDC, use of the ...
... toxic chromium(VI) species. Corey’s pyridinium dichromate (PDC) in DMF4b could have been used more frequently to oxidize primary alcohols 1 to carboxylic acids 3 because of the neutral nature of the reaction (Scheme 1). However, besides the hazardous nature of the chromium species in PDC, use of the ...
Scope and Limitations - Organic Reactions Wiki
... Osmium tetroxide has since established itself as the reagent of choice for the syndihydroxylation of olefins, primarily because of its inertness toward other functional groups and lack of over-oxidation products.4 Researchers from the UpJohn company reported a convenient and reliable procedure for ...
... Osmium tetroxide has since established itself as the reagent of choice for the syndihydroxylation of olefins, primarily because of its inertness toward other functional groups and lack of over-oxidation products.4 Researchers from the UpJohn company reported a convenient and reliable procedure for ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.