Chapter 10 Structure and Synthesis of Alcohols
... ¾ Diols = alcohols with two –OH groups. ¾ Two numbers are needed to locate the two -OH groups. ¾ Use -diol as suffix instead of -ol. ...
... ¾ Diols = alcohols with two –OH groups. ¾ Two numbers are needed to locate the two -OH groups. ¾ Use -diol as suffix instead of -ol. ...
Green synthesis of 2-amino-7-hydroxy-4-aryl-4H
... 1. Curini, M.; Cravotto, G.; Epofano, F.; Giannone, G. Curr. Med. Chem. 2006, 13, 199–222. 2. Litvinov, Y. M.; Shestopalov, A. M. Adv. Heterocycl. Chem. 2011, 103, 175–260. 3. Desimone, R. W.; Currie, K. S.; Mitchell, S. A.; Darrow, J. W.; Pippin, D. A. Comb. Chem. High T. Scr. 2004, ...
... 1. Curini, M.; Cravotto, G.; Epofano, F.; Giannone, G. Curr. Med. Chem. 2006, 13, 199–222. 2. Litvinov, Y. M.; Shestopalov, A. M. Adv. Heterocycl. Chem. 2011, 103, 175–260. 3. Desimone, R. W.; Currie, K. S.; Mitchell, S. A.; Darrow, J. W.; Pippin, D. A. Comb. Chem. High T. Scr. 2004, ...
Convenient Methods for the Reduction of Amides, Nitriles
... system is useful in the hydrobor_ation of olefins and the corresponding ...
... system is useful in the hydrobor_ation of olefins and the corresponding ...
Factors influencing ring closure through olefin metathesis – A
... main group alkylating agents (such as WCl6/Bu4Sn, MoO3/SiO2, Re2O7/N2O3Cl2) were inefficient for RCM. An efficient RCM catalyst3 should be a good initiator of metathesis, should react exclusively or at least preferentially with olefins over the other polar groups present in the olefinic substrate an ...
... main group alkylating agents (such as WCl6/Bu4Sn, MoO3/SiO2, Re2O7/N2O3Cl2) were inefficient for RCM. An efficient RCM catalyst3 should be a good initiator of metathesis, should react exclusively or at least preferentially with olefins over the other polar groups present in the olefinic substrate an ...
Organic Chemistry
... CH3 - C-H CH2 = C- H • Step 2: Proton transfer from HA to the carbonyl group of a second molecule of aldehyde or ketone. O CH3 -C-H + H A ...
... CH3 - C-H CH2 = C- H • Step 2: Proton transfer from HA to the carbonyl group of a second molecule of aldehyde or ketone. O CH3 -C-H + H A ...
enzymatic And Limited Industrial Use
... A stereospecific reaction is one in which a single starting material yields only a single stereoisomer ...
... A stereospecific reaction is one in which a single starting material yields only a single stereoisomer ...
Kinetic isotope effects of 12CH3D+OH and 13CH3D+OH from 278 to
... greenhouse gas and plays a central role in the chemistry of the Earth’s atmosphere. Nonetheless there are significant uncertainties in its source budget. Analysis of the isotopic composition of atmospheric methane, including the doubly substituted species 13 CH3 D, offers new insight into the methan ...
... greenhouse gas and plays a central role in the chemistry of the Earth’s atmosphere. Nonetheless there are significant uncertainties in its source budget. Analysis of the isotopic composition of atmospheric methane, including the doubly substituted species 13 CH3 D, offers new insight into the methan ...
Alcohols, Phenols, and Ethers
... (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 c - FTF Statements: (1) Glycerin, a three-carbon diol, has a gre ...
... (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 c - FTF Statements: (1) Glycerin, a three-carbon diol, has a gre ...
Oxidation involving CO System ( O
... Metabolizes 1° and 2° alcohols and aldehydes containing at least one “H” attached to a-C; 1° alcohols typically go to the aldehyde then acid; 2° alcohols are converted to ketone, which cannot be further converted to the acid. The aldehyde is converted back to an alcohol by alcohol (keto) reductases ...
... Metabolizes 1° and 2° alcohols and aldehydes containing at least one “H” attached to a-C; 1° alcohols typically go to the aldehyde then acid; 2° alcohols are converted to ketone, which cannot be further converted to the acid. The aldehyde is converted back to an alcohol by alcohol (keto) reductases ...
74 CHAPTER-IV "LEAD (IV) ACETATE OXIDATIONS"
... A survey of the literature revealed that a successful attempt at 1, 2-carbonyl transposition was simultaneously reported by Perkin48 and Bredt49 in 1911. Since then other methods were developed dealing with 1,2-carbonyl transposition in the terpene systems50' 51 and steroids systems. In 1944, Ruzika ...
... A survey of the literature revealed that a successful attempt at 1, 2-carbonyl transposition was simultaneously reported by Perkin48 and Bredt49 in 1911. Since then other methods were developed dealing with 1,2-carbonyl transposition in the terpene systems50' 51 and steroids systems. In 1944, Ruzika ...
Chapter 7 Hydrosilylation of Carbon
... to the participation of π-benzylic palladium intermediates [1, 2]. It is known that bisphosphine-palladium complexes are catalytically much less active than monophosphine-palladium complexes and hence asymmetric synthesis has been attempted by use of chiral monodentate phosphine ligands. In the first ...
... to the participation of π-benzylic palladium intermediates [1, 2]. It is known that bisphosphine-palladium complexes are catalytically much less active than monophosphine-palladium complexes and hence asymmetric synthesis has been attempted by use of chiral monodentate phosphine ligands. In the first ...
synthesis, chemistry and optical resol
... estimated by comparison of the rotation with that of (-)-transcyclooctene. We have found that both I26.101- and [22.10]betweenanene (13c and 13b) undergo asymmetric epoxidation upon treatment with (+)-monoperoxycamphoric acid in chloroform.19 If the reaction is quenched at 50% conversion (30 equiv o ...
... estimated by comparison of the rotation with that of (-)-transcyclooctene. We have found that both I26.101- and [22.10]betweenanene (13c and 13b) undergo asymmetric epoxidation upon treatment with (+)-monoperoxycamphoric acid in chloroform.19 If the reaction is quenched at 50% conversion (30 equiv o ...
Dehydration of ROH
... depends primarily on the degree of stabilization and solvation of the alkoxide ion. • The negatively charged oxygens of methoxide and ethoxide are about as accessible as the oxygen of hydroxide ion for solvation; these alcohol are about as acidic as water. • As the bulk of the alkyl group increases, ...
... depends primarily on the degree of stabilization and solvation of the alkoxide ion. • The negatively charged oxygens of methoxide and ethoxide are about as accessible as the oxygen of hydroxide ion for solvation; these alcohol are about as acidic as water. • As the bulk of the alkyl group increases, ...
Zinc Alkyls in Organic Synthesis
... Asymmetric Additions to Aldehydes and Ketones DEZ is also useful in catalytic asymmetric addition to aldehydes or ketones forming chiral secondary or tertiary alcohols. These reactions typically involve an amine or a sulfonamide ligand in combination with tetraisopropyl titanate (TIPT). The added s ...
... Asymmetric Additions to Aldehydes and Ketones DEZ is also useful in catalytic asymmetric addition to aldehydes or ketones forming chiral secondary or tertiary alcohols. These reactions typically involve an amine or a sulfonamide ligand in combination with tetraisopropyl titanate (TIPT). The added s ...
215-216 HH W12-notes
... historical use of the term: (1) oxide (oxyd/oxyde) – the ‘acid’ form of an element; e.g., S + air → oxide of S (acid of sulfur) (2) oxidation or oxidize – to make such an acid, to make the oxide (3) oxygen – Lavoisier: substance in the air that makes acids; “the bringer of acids” = “oxygen” (4) oxid ...
... historical use of the term: (1) oxide (oxyd/oxyde) – the ‘acid’ form of an element; e.g., S + air → oxide of S (acid of sulfur) (2) oxidation or oxidize – to make such an acid, to make the oxide (3) oxygen – Lavoisier: substance in the air that makes acids; “the bringer of acids” = “oxygen” (4) oxid ...
Chapter 4
... If the organic molecules interacting with the protein are also chiral, then there will be an energy difference depending upon which enantiomer is interacting with the protein ...
... If the organic molecules interacting with the protein are also chiral, then there will be an energy difference depending upon which enantiomer is interacting with the protein ...
interaction of alcohols with alkalies under autogeneous pressure
... electric furnace, the temperature of which can be controlled with a Sunvic regulator correct to ±3° C. The top cover of the bomb is connected with the gas inlet and the pressure gauge, while the gas outlet is situated at the bottom. Connections are made using Amine° stainless steel tubing 1" o.d. an ...
... electric furnace, the temperature of which can be controlled with a Sunvic regulator correct to ±3° C. The top cover of the bomb is connected with the gas inlet and the pressure gauge, while the gas outlet is situated at the bottom. Connections are made using Amine° stainless steel tubing 1" o.d. an ...
Studies toward the Stereoselective Synthesis of the
... agents of various diseases in man and his domestic animals. Human beings and animals get the diseases, commonly called mycotoxicoses through the ingestion of foods or feeds contaminated by these toxic fungal metabolites. The outbreak of a mycotoxicosis that caused the death of 100 000 turkeys, 14 00 ...
... agents of various diseases in man and his domestic animals. Human beings and animals get the diseases, commonly called mycotoxicoses through the ingestion of foods or feeds contaminated by these toxic fungal metabolites. The outbreak of a mycotoxicosis that caused the death of 100 000 turkeys, 14 00 ...
Aldehydes and Ketones - Belle Vernon Area School District
... Reduction of aldes and kets to alcohols O R-C-H + H2 –Pt/Pd R-C-OH RCHO + H2 –Pt/Pd RCH2OH O O R-C-R + H2 –Pt/Pd R-C-R RCOR + H2 –Pt/Pd RCHOHR ...
... Reduction of aldes and kets to alcohols O R-C-H + H2 –Pt/Pd R-C-OH RCHO + H2 –Pt/Pd RCH2OH O O R-C-R + H2 –Pt/Pd R-C-R RCOR + H2 –Pt/Pd RCHOHR ...
RheniumCatalyzed Deoxydehydration of Diols and Polyols
... While the majority of oil, coal, and gas is used for energy production, the realization of an economy completely independent of fossil resources also requires biomass-based substitutes for polymers, medicine, pesticides, and so forth.[1] The evergrowing world population makes it questionable to use ...
... While the majority of oil, coal, and gas is used for energy production, the realization of an economy completely independent of fossil resources also requires biomass-based substitutes for polymers, medicine, pesticides, and so forth.[1] The evergrowing world population makes it questionable to use ...
Asymmetric Catalytic Aldol
... • Over 30 have been identified to date • Type I aldolases are primarily found in animals and plants and activate the donor by forming a schiff base as an intermediate. • Type II aldolases are found in bacteria and fungi and contain a Zn2+ cofactor in the active site. • In both types of aldolases the ...
... • Over 30 have been identified to date • Type I aldolases are primarily found in animals and plants and activate the donor by forming a schiff base as an intermediate. • Type II aldolases are found in bacteria and fungi and contain a Zn2+ cofactor in the active site. • In both types of aldolases the ...
Chapter 13. Alcohols, Diols, and Ethers
... historical use of the term: (1) oxide (oxyd/oxyde) – the ‘acid’ form of an element; e.g., S + air → oxide of S (acid of sulfur) (2) oxidation or oxidize – to make such an acid, to make the oxide (3) oxygen – Lavoisier: substance in the air that makes acids; “the bringer of acids” = “oxygen” (4) oxid ...
... historical use of the term: (1) oxide (oxyd/oxyde) – the ‘acid’ form of an element; e.g., S + air → oxide of S (acid of sulfur) (2) oxidation or oxidize – to make such an acid, to make the oxide (3) oxygen – Lavoisier: substance in the air that makes acids; “the bringer of acids” = “oxygen” (4) oxid ...
Kinetics of Oxidation of Benzyl Alcohol with Dilute Nitric Acid
... oxidation of benzyl alcohol using molecular oxygen requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited ...
... oxidation of benzyl alcohol using molecular oxygen requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited ...
Heterogeneous Catalysts for Biodiesel Production
... in one step.9,11–13 (b) use of acid catalysts. Acid catalyst can also promote esterification and transesterification.5,10,15 (c) pre-esterification method. FFAs are first esterified to FAMEs using an acid catalyst, and then, transesterification is performed, as usual, by using an alkaline catalyst.5 ...
... in one step.9,11–13 (b) use of acid catalysts. Acid catalyst can also promote esterification and transesterification.5,10,15 (c) pre-esterification method. FFAs are first esterified to FAMEs using an acid catalyst, and then, transesterification is performed, as usual, by using an alkaline catalyst.5 ...
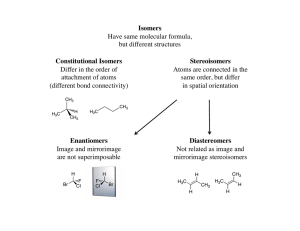
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.