Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... The sequence converts C=O to C=C A phosphorus ylide adds to an aldehyde or ketone to yield a dipolar intermediate called a betaine The intermediate spontaneously decomposes through a four-membered ring to yield alkene and triphenylphosphine oxide, (Ph)3P=O Formation of the ylide is shown below ...
... The sequence converts C=O to C=C A phosphorus ylide adds to an aldehyde or ketone to yield a dipolar intermediate called a betaine The intermediate spontaneously decomposes through a four-membered ring to yield alkene and triphenylphosphine oxide, (Ph)3P=O Formation of the ylide is shown below ...
Alcohols Phenols and Ethers
... ALCOHOLS, PHENOLS and ETHERS Nomenclature of Alcohols 1. Alcohols are named by finding the longest continues carbon chain and changing the ending to –ol (e.g., methanol, ethanol, propanol, etc.). 2. With unsaturated alcohols, two endings are needed, one for the double or triple bond and one for the ...
... ALCOHOLS, PHENOLS and ETHERS Nomenclature of Alcohols 1. Alcohols are named by finding the longest continues carbon chain and changing the ending to –ol (e.g., methanol, ethanol, propanol, etc.). 2. With unsaturated alcohols, two endings are needed, one for the double or triple bond and one for the ...
Tin-Catalyzed Esterification and Transesterification Reactions: A
... the fatty acid, favoring his attack by the hydroxyl group of ethanol, thereby generating an increased formation of ethyl oleate. The carbon chain length of alcohol can affect both the rate and also the conversion of fatty acid in esterification reactions (Figure 4) [8]. A different behavior was observ ...
... the fatty acid, favoring his attack by the hydroxyl group of ethanol, thereby generating an increased formation of ethyl oleate. The carbon chain length of alcohol can affect both the rate and also the conversion of fatty acid in esterification reactions (Figure 4) [8]. A different behavior was observ ...
Chapter 4 Stereochemistry and Chirality Flow chart for determining
... If you have a molecule with two chiral centers with different sets of things attached (1 and 2) either which can be R or S, there are four possible molecules that can result: (1R, 2S), (1R, 2R), (1S, 2S), (1S, 2R). • Since for a single chiral center R and S are related by mirror symmetry then if in ...
... If you have a molecule with two chiral centers with different sets of things attached (1 and 2) either which can be R or S, there are four possible molecules that can result: (1R, 2S), (1R, 2R), (1S, 2S), (1S, 2R). • Since for a single chiral center R and S are related by mirror symmetry then if in ...
carbonyl chemistry 1
... Thus, the carbonyl group of an aldehyde or a ketone can be protected in the form of an acetal or ketal. Deprotection following reaction on other regions of the molecule then yields the carbonyl group again – this then is the first protection/deprotection protocol we have encountered. In general, sim ...
... Thus, the carbonyl group of an aldehyde or a ketone can be protected in the form of an acetal or ketal. Deprotection following reaction on other regions of the molecule then yields the carbonyl group again – this then is the first protection/deprotection protocol we have encountered. In general, sim ...
Asymmetric Synthesis: Substrate and Auxiliary Control
... Virtually every chiral molecule ever made by chemists in an enantio-enriched (non-racemic) form ultimately derives its chirality from chiral pool molecules. ...
... Virtually every chiral molecule ever made by chemists in an enantio-enriched (non-racemic) form ultimately derives its chirality from chiral pool molecules. ...
Derivatization of polar compounds for GC - Sigma
... •If there is high humidity in the room, it may be helpful to store syringes, vials, etc. in a dry box •Silyl reagents are used in excess and can tolerate very small amounts of moisture – but still try to keep things dry! •Chemical removal of water by 2,2-dimethoxypropane ...
... •If there is high humidity in the room, it may be helpful to store syringes, vials, etc. in a dry box •Silyl reagents are used in excess and can tolerate very small amounts of moisture – but still try to keep things dry! •Chemical removal of water by 2,2-dimethoxypropane ...
Hein and Arena - faculty at Chemeketa
... magnesium are shifted away from the electropositive Mg to form a strongly polar covalent bond. As a result the charge distribution in the Grignard reagent is such that the organic group (R) is partially negative and the –MgX group is partially positive. This charge distribution directs the manner in ...
... magnesium are shifted away from the electropositive Mg to form a strongly polar covalent bond. As a result the charge distribution in the Grignard reagent is such that the organic group (R) is partially negative and the –MgX group is partially positive. This charge distribution directs the manner in ...
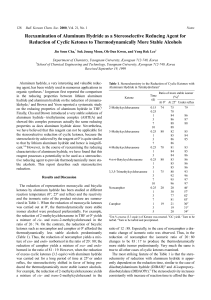
Reexamination of Aluminum Hydride as a Stereoselective Reducing
... ketones such as norcamphor and camphor at 0o afforded the thermodynamically less stable alcohols predominantly (Table 1). Thus, the reduction of norcamphor yields a mixture of exo- and endo- norborneol in the ratio of 20 : 80; the reduction of camphor yields a mixture of exo- and endoborneol in the ...
... ketones such as norcamphor and camphor at 0o afforded the thermodynamically less stable alcohols predominantly (Table 1). Thus, the reduction of norcamphor yields a mixture of exo- and endo- norborneol in the ratio of 20 : 80; the reduction of camphor yields a mixture of exo- and endoborneol in the ...
Montmorillonite: An efficient, heterogeneous and
... An extensive study has been made of a wide range of organic reactions catalyzed by clay minerals (see, e.g., Fripiat and Cruz-Cumplido, 1974; Theng, 1974; Thomas et al., 1977; Bittles et al., 1964a, 1964b, 1964c). Recently, renewed interest has been shown in the use of natural and synthetic smectiti ...
... An extensive study has been made of a wide range of organic reactions catalyzed by clay minerals (see, e.g., Fripiat and Cruz-Cumplido, 1974; Theng, 1974; Thomas et al., 1977; Bittles et al., 1964a, 1964b, 1964c). Recently, renewed interest has been shown in the use of natural and synthetic smectiti ...
Full Text - Verlag der Zeitschrift für Naturforschung
... i. e. drugs, flavourings, fragrant substances and plant protection agents. An advantage of enzymatic reactions over the classic synthesis is that they usually provide a high level of stereoselectivity. Therefore, they may be used for the synthesis of bioactive compounds, as well as chiral synthons, ...
... i. e. drugs, flavourings, fragrant substances and plant protection agents. An advantage of enzymatic reactions over the classic synthesis is that they usually provide a high level of stereoselectivity. Therefore, they may be used for the synthesis of bioactive compounds, as well as chiral synthons, ...
投影片 1
... groups, these generally are related to a particular application as will be discussed in the sections on uses of blocked isocyanates in the subsequent second paper. ...
... groups, these generally are related to a particular application as will be discussed in the sections on uses of blocked isocyanates in the subsequent second paper. ...
molecules
... configuration. In contrast, epoxidation of cis-stilbene is associated with some lose of stereochemistry and affords 80% cis- and 10% trans-stilbene oxides, respectively. This catalytic system exhibits a good regioselectivity for epoxidation of R-(+)-limonene. The ratio among 1,2- and 8,9-epoxides wa ...
... configuration. In contrast, epoxidation of cis-stilbene is associated with some lose of stereochemistry and affords 80% cis- and 10% trans-stilbene oxides, respectively. This catalytic system exhibits a good regioselectivity for epoxidation of R-(+)-limonene. The ratio among 1,2- and 8,9-epoxides wa ...
Synthesis of Cyclobutanes by Lewis Acid-Promoted Ketene
... Triethylamine (>99.5%) was purchased from Sigma Aldrich and used as received. Upon the addition of triethylamine the solution becomes a canary yellow color and a white solid forms within minutes. Cyclopentene (97%) was purchased from Alfa Aesar and used as received. Ethylaluminum dichloride (1 M in ...
... Triethylamine (>99.5%) was purchased from Sigma Aldrich and used as received. Upon the addition of triethylamine the solution becomes a canary yellow color and a white solid forms within minutes. Cyclopentene (97%) was purchased from Alfa Aesar and used as received. Ethylaluminum dichloride (1 M in ...
ELECTROPHILIC ADDITIONS OF ALKENES AS THE
... addition of water to the p-bond. The result is the transformation of alkenes into alcohols. We now look at the generalities of this type of reaction (electrophilic addition) and then we look at each reaction individually. The C=C p-bond of alkenes is a source of electrons. It is considered a weak ba ...
... addition of water to the p-bond. The result is the transformation of alkenes into alcohols. We now look at the generalities of this type of reaction (electrophilic addition) and then we look at each reaction individually. The C=C p-bond of alkenes is a source of electrons. It is considered a weak ba ...
Alcohols, Phenols and Ethers
... group in alcohols. 42. Explain why nucleophilic substitution reactions are not very common in phenols. 43. Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism. 44. Explain why is O==C==O nonpolar while R—O—R is polar. 45. Why is the rea ...
... group in alcohols. 42. Explain why nucleophilic substitution reactions are not very common in phenols. 43. Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism. 44. Explain why is O==C==O nonpolar while R—O—R is polar. 45. Why is the rea ...
Chapter 12: Aldehydes, Ketones and Carboxylic acids
... Reactions of aldehydes and ketones: Aldehydes are generally more reactive than ketones in nucleophilic addition reactions due to steric and electronic reasons (or inductive effect). Electronic Effect: Relative reactivities of aldehydes and ketones in nucleophilic addition reactions is due the positi ...
... Reactions of aldehydes and ketones: Aldehydes are generally more reactive than ketones in nucleophilic addition reactions due to steric and electronic reasons (or inductive effect). Electronic Effect: Relative reactivities of aldehydes and ketones in nucleophilic addition reactions is due the positi ...
Enantioselective Organocatalytic Aminomethylation of Aldehydes: A
... catalyst F and recrystallizing the HCl salt of the γ-amino alcohol gave a 72% yield of material with >98% ee. The benzyl groups were removed and replaced by Boc in an efficient one-pot operation. Jones oxidation12 then provided desired β2-amino acid product after simple extraction, with >50% overall ...
... catalyst F and recrystallizing the HCl salt of the γ-amino alcohol gave a 72% yield of material with >98% ee. The benzyl groups were removed and replaced by Boc in an efficient one-pot operation. Jones oxidation12 then provided desired β2-amino acid product after simple extraction, with >50% overall ...
Isomers and Isomerism Isomers
... a pair of enantiomers will react different only with chiral reagents. Because living systems usually react with only one of a pair of enanatiomers, the living system is also chiral. The part of the living system that is chiral is the enzyme. An enzyme has the ability to react with only one of a pair ...
... a pair of enantiomers will react different only with chiral reagents. Because living systems usually react with only one of a pair of enanatiomers, the living system is also chiral. The part of the living system that is chiral is the enzyme. An enzyme has the ability to react with only one of a pair ...
Carbonyls
... alcohols by oxidation with a mild oxidizer, eg (CrO3/py), pyridinium dichromate Example: Note: this oxidation removes two hydrogens. It is also an oxidative elimination reaction. ...
... alcohols by oxidation with a mild oxidizer, eg (CrO3/py), pyridinium dichromate Example: Note: this oxidation removes two hydrogens. It is also an oxidative elimination reaction. ...
Orbitals - drjosephryan.com
... ketones with hydride reagents may be represented as proceeding through a nucleophilic addition of a hydride ion (:H–) to the C=O carbon • LiAlH4 and NaBH4 act as if they are donors of hydride ion ...
... ketones with hydride reagents may be represented as proceeding through a nucleophilic addition of a hydride ion (:H–) to the C=O carbon • LiAlH4 and NaBH4 act as if they are donors of hydride ion ...
Aldehydes and ketones
... • Acetals can be isolated and used in subsequent chemical reactions. (Hemiacetals are less stable and generally can’t be isolated.) • If an acetal is treated with acid in the presence of water, a hydrolysis reaction occurs acid catalyst H2O aldehyde ...
... • Acetals can be isolated and used in subsequent chemical reactions. (Hemiacetals are less stable and generally can’t be isolated.) • If an acetal is treated with acid in the presence of water, a hydrolysis reaction occurs acid catalyst H2O aldehyde ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.