An Epoxidation Reaction: The Epoxidation of Cholesterol to 5 ,6
... The Experimental Reaction In this experiment, we will prepare an epoxide or oxirane by epoxidizing chlolesterol. Cholesterol is the most common compound in the class of compounds called sterols. As the name implies, sterols contain a hydroxyl (-OH) group. Because sterols are alcohols, they have the ...
... The Experimental Reaction In this experiment, we will prepare an epoxide or oxirane by epoxidizing chlolesterol. Cholesterol is the most common compound in the class of compounds called sterols. As the name implies, sterols contain a hydroxyl (-OH) group. Because sterols are alcohols, they have the ...
PDF w - Erowid
... acids to the corresponding alcohols using sodium borohydride and iodine in THF. We now report that this was found to be an excellent process for the direct reduction of amino acids. The reactions were routinely carried out on a 10-g scale while the reduction of phenylalanine haa been successfdy perf ...
... acids to the corresponding alcohols using sodium borohydride and iodine in THF. We now report that this was found to be an excellent process for the direct reduction of amino acids. The reactions were routinely carried out on a 10-g scale while the reduction of phenylalanine haa been successfdy perf ...
C - Deans Community High School
... Reacting hydrogen with ethene to form ethane would be an example of this type of reaction. ...
... Reacting hydrogen with ethene to form ethane would be an example of this type of reaction. ...
Lecture - Ch 17
... Compounds: Grignard Reagents • Grignard reagents do not give addition products with carboxylic acids – Acidic carboxyl hydrogen reacts with the basic Grignard reagent to yield a hydrocarbon and the magnesium salt of the acid ...
... Compounds: Grignard Reagents • Grignard reagents do not give addition products with carboxylic acids – Acidic carboxyl hydrogen reacts with the basic Grignard reagent to yield a hydrocarbon and the magnesium salt of the acid ...
Faculteit der Natuurwetenschappen, Wiskunde en Informatica
... To obtain aldehyde 3 first compound 11 needs to be synthesized (figure 12). The αiodoenone (9)7 was reacted with optically active (R,R)-hydrobenzoin (10)8 and compound 11 was obtained in 62% yield. ...
... To obtain aldehyde 3 first compound 11 needs to be synthesized (figure 12). The αiodoenone (9)7 was reacted with optically active (R,R)-hydrobenzoin (10)8 and compound 11 was obtained in 62% yield. ...
Ethers and Epoxides - faculty at Chemeketa
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
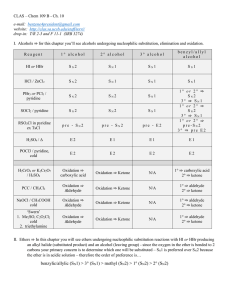
Chapter 10 - UCSB CLAS
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
DEVELOPMENT OF GREEN AND OF POLYMER
... known as Fetizon’s reagent. It is a very versatile and useful oxidizing agent, its chief advantages being its ease of use, selectivity and mild conditions under which it is used. Lead tetraacetate finds major application in the cleavage of 1,2-diols. The reaction is virtually quantitative and leads ...
... known as Fetizon’s reagent. It is a very versatile and useful oxidizing agent, its chief advantages being its ease of use, selectivity and mild conditions under which it is used. Lead tetraacetate finds major application in the cleavage of 1,2-diols. The reaction is virtually quantitative and leads ...
document
... • In order to write correct equations, if you are not asked for the mechanism, just ...
... • In order to write correct equations, if you are not asked for the mechanism, just ...
Exam 2
... Chern 24 2 (w 2016) exam #2B 1. (10 pts) Circle what is true about Substitution and elimination reactions. ...
... Chern 24 2 (w 2016) exam #2B 1. (10 pts) Circle what is true about Substitution and elimination reactions. ...
ALCOHOLS, PHENOLS AND ETHERS
... The reduction is carried out using hydride reagents such as lithium aluminium hydride (LiAIH4) and sodium borohydride (NaBH4). LiAIH4 is stronger and reacts explosively with water while NaBH4 is convenient to handle and reacts slowly. Lithium aluminium hydride reduces all of the above classes of com ...
... The reduction is carried out using hydride reagents such as lithium aluminium hydride (LiAIH4) and sodium borohydride (NaBH4). LiAIH4 is stronger and reacts explosively with water while NaBH4 is convenient to handle and reacts slowly. Lithium aluminium hydride reduces all of the above classes of com ...
alcohols03
... hydrogen bonds must be overcome when molecules in the liquid separate into the gaseous state during boiling. The bp of ethers are similar to those of alkanes of equivalent MW. For example, diethyl ether (‘Quick Start’) with MW = 44, has a bp of 35 C and n-pentane (MW = 44) is also 35 C. Simila ...
... hydrogen bonds must be overcome when molecules in the liquid separate into the gaseous state during boiling. The bp of ethers are similar to those of alkanes of equivalent MW. For example, diethyl ether (‘Quick Start’) with MW = 44, has a bp of 35 C and n-pentane (MW = 44) is also 35 C. Simila ...
4-Dimethylaminopyridine - Sigma
... DMAP has been used in natural-product chemistry for the acetylation of a tertiary hydroxyl group of an amino sugar,9 and for acylations of terpenes4,10 and acetylenes11,12 containing secondary and tertiary hydroxyl groups. Connors and Albert recommend using acetic anhydride/DMAP as a reagent for the ...
... DMAP has been used in natural-product chemistry for the acetylation of a tertiary hydroxyl group of an amino sugar,9 and for acylations of terpenes4,10 and acetylenes11,12 containing secondary and tertiary hydroxyl groups. Connors and Albert recommend using acetic anhydride/DMAP as a reagent for the ...
Nuggets of Knowledge for Chapter 14 – Ethers
... When an epoxide reacts with a nucleophile, an alkoxide is formed, and needs to be neutralized to complete the reaction. The proton source depends on the nucleophile being used – it might be the solvent, or an acid added after the nucleophilic attack is finished. ...
... When an epoxide reacts with a nucleophile, an alkoxide is formed, and needs to be neutralized to complete the reaction. The proton source depends on the nucleophile being used – it might be the solvent, or an acid added after the nucleophilic attack is finished. ...
Unit-8-Alcohols-Aldehydes
... Unit 8 - Organic Molecules III Alcohols, Thiols, Ethers, Aldehydes and Ketones In this unit we continue surveying some of the families of organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, ty ...
... Unit 8 - Organic Molecules III Alcohols, Thiols, Ethers, Aldehydes and Ketones In this unit we continue surveying some of the families of organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, ty ...
Alcohols - La Salle University
... • In men, ethanol lowers levels of testosterone (and sperm count) due to lack of enzymes needed for the steroid biosynthesis. • The enzyme CYP2E1, which is responsible for converting acetaminophen into liver toxins, is activated by ethanol. • Ethanol has a caloric value of 7.1Cal/g (fat has a value ...
... • In men, ethanol lowers levels of testosterone (and sperm count) due to lack of enzymes needed for the steroid biosynthesis. • The enzyme CYP2E1, which is responsible for converting acetaminophen into liver toxins, is activated by ethanol. • Ethanol has a caloric value of 7.1Cal/g (fat has a value ...
Links - American Chemical Society
... and aryl fluorides is especially noteworthy, as several of these transformations were previously6c carried out in the presence of PMHS only with difficulty and concomitant hydrodehalogenation. The selective reduction of methyl esters in the presence of terminal alkynes and tert-butyl esters should b ...
... and aryl fluorides is especially noteworthy, as several of these transformations were previously6c carried out in the presence of PMHS only with difficulty and concomitant hydrodehalogenation. The selective reduction of methyl esters in the presence of terminal alkynes and tert-butyl esters should b ...
heterogeneous chiral catalyst derived from hydrolyzed
... instigated by stricter regulations from health authorities, a growing number of new drugs are now marketed as single enantiomers [21-23]. The thalidomide tragedy of the 1960’s is commonly used as an example. It is an anti-emetic drug that was prescribed to pregnant women to help combat morning sickn ...
... instigated by stricter regulations from health authorities, a growing number of new drugs are now marketed as single enantiomers [21-23]. The thalidomide tragedy of the 1960’s is commonly used as an example. It is an anti-emetic drug that was prescribed to pregnant women to help combat morning sickn ...
Hydrogenation, Transfer Hydrogenat- ion and Hydrogen Transfer Reactions
... Catalysis is the process that facilitates a chemical reaction, and was first introduced by Jöns Jacob Berzelius in 1835.2 A catalyst is an additive that triggers and participates in the reaction to make the reaction go faster by decreasing the activation energy, without itself being consumed. It doe ...
... Catalysis is the process that facilitates a chemical reaction, and was first introduced by Jöns Jacob Berzelius in 1835.2 A catalyst is an additive that triggers and participates in the reaction to make the reaction go faster by decreasing the activation energy, without itself being consumed. It doe ...
Titania-catalysed oxidative dehydrogenation of ethyl lactate
... (eqn (1)). In all cases no solvent was used and the catalyst : substrate ratio was kept constant at 100 : 1 w/w. Four materials, Fe2O3, ZrO2, CeO2 and ZnO, showed <5% conversion after 24 h, and were therefore discarded. Another, V2O5/MgO–Al2O3 showed good catalytic activity in the first run. However ...
... (eqn (1)). In all cases no solvent was used and the catalyst : substrate ratio was kept constant at 100 : 1 w/w. Four materials, Fe2O3, ZrO2, CeO2 and ZnO, showed <5% conversion after 24 h, and were therefore discarded. Another, V2O5/MgO–Al2O3 showed good catalytic activity in the first run. However ...
Ultrasound Assisted Synthesis of 5,9
... Recent research has shown that several hydrocarbons with a 1,5-dimethyl skeleton are insect pheromones [1]. 5,9-Dimethylpentadecane (1) and 5,9-dimethylhexadecane (2) are known as the major and minor constituents, respectively, of the sex pheromone of Leucoptera coffeella, a pest of coffee trees [2] ...
... Recent research has shown that several hydrocarbons with a 1,5-dimethyl skeleton are insect pheromones [1]. 5,9-Dimethylpentadecane (1) and 5,9-dimethylhexadecane (2) are known as the major and minor constituents, respectively, of the sex pheromone of Leucoptera coffeella, a pest of coffee trees [2] ...
Organic Chemistry Notes by Jim Maxka jim.maxka
... Reiew of oxidation/reduction First some stuff to memorize: Oxidation means gain of bonds Æ O; Loss of bonds Æ H. Reduction means gain of bonds Æ H; Loss of bonds Æ O. CH4 is the most oxidized or reduced organic molecule? What is the oxidation state of C? What is the most oxidized? What is the oxidat ...
... Reiew of oxidation/reduction First some stuff to memorize: Oxidation means gain of bonds Æ O; Loss of bonds Æ H. Reduction means gain of bonds Æ H; Loss of bonds Æ O. CH4 is the most oxidized or reduced organic molecule? What is the oxidation state of C? What is the most oxidized? What is the oxidat ...
Grignard-syn-12-ques
... Direct formation of a Grignard reagent, involves an alkyl halide reacted with Mg metal. ...
... Direct formation of a Grignard reagent, involves an alkyl halide reacted with Mg metal. ...
Preparation of d, l-Phenylalanine by Amidocarbonylation of Benzyl
... in this patent.10 In view of the fact that benzyl chloride is a cheap and readily available commodity we decided to further develop this reaction to make it amenable to large scale production. The process as described in the patent has several drawbacks that need to be overcome before scale-up can b ...
... in this patent.10 In view of the fact that benzyl chloride is a cheap and readily available commodity we decided to further develop this reaction to make it amenable to large scale production. The process as described in the patent has several drawbacks that need to be overcome before scale-up can b ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.