Lecture 3-edited
... This reagent is obtained by adding pyridine to a solution of CrO3 in hydrochloric acid. PCC oxidizes primary and secondary alcohols to aldehydes and ketones, respectively (Scheme 3). As PCC is slightly acidic so it may affect the acid sensitive groups. The powdered NaOAc is used along with PCC for t ...
... This reagent is obtained by adding pyridine to a solution of CrO3 in hydrochloric acid. PCC oxidizes primary and secondary alcohols to aldehydes and ketones, respectively (Scheme 3). As PCC is slightly acidic so it may affect the acid sensitive groups. The powdered NaOAc is used along with PCC for t ...
Discovery and Exploitation of AZADO: The Highly Active Catalyst for
... [(1); Chart 1], has extensively been used as a catalyst for oxidation of alcohols, because TEMPO is readily available from chemical suppliers at a reasonable price, and because the method allows the use of various safe bulk oxidants, thereby offering safe and extremely efficient oxidation of alcohols ...
... [(1); Chart 1], has extensively been used as a catalyst for oxidation of alcohols, because TEMPO is readily available from chemical suppliers at a reasonable price, and because the method allows the use of various safe bulk oxidants, thereby offering safe and extremely efficient oxidation of alcohols ...
Alcohols
... Substituted Phenols Can be more or less acidic than phenol itself. Remember, the acidity of any alcohol is determined by the stability of the alkoxide or phenoxide anion produced. The more stable the anion produced the more acidic the alcohol ...
... Substituted Phenols Can be more or less acidic than phenol itself. Remember, the acidity of any alcohol is determined by the stability of the alkoxide or phenoxide anion produced. The more stable the anion produced the more acidic the alcohol ...
Chapter 18: Ethers and Epoxides
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
Ethers and Epoxides
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
... halide to give dialkyl sulfides For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
Common aldehydes and ketones
... be split easily to regenerate the aldehyde or ketone by treating it with either dilute acid or dilute alkali. ...
... be split easily to regenerate the aldehyde or ketone by treating it with either dilute acid or dilute alkali. ...
Neutral ionic liquid [BMIm]BF4 promoted highly selective
... revealed that the reactivity of tert-butanol could be greatly improved by the use of tetrafluoroborate ionic liquids as media [26,27]. Therefore, we consequently investigated the esterification reaction of tert-butanol in [BMIm]BF4 , which is considered as a neutral polar solvent. To our surprising, ...
... revealed that the reactivity of tert-butanol could be greatly improved by the use of tetrafluoroborate ionic liquids as media [26,27]. Therefore, we consequently investigated the esterification reaction of tert-butanol in [BMIm]BF4 , which is considered as a neutral polar solvent. To our surprising, ...
Final Exam Review Sheet Chemistry 110a/1998
... a. Explain why 3° alcohols do not react. b. Explain why 2° alcohols are converted to ketones. i. Be able to explain how a kinetic isotope effect can be used to determine the rate-determining step of this oxidation process. c. Explain why 1° alcohols are converted to carboxylic acids. d. Explain how ...
... a. Explain why 3° alcohols do not react. b. Explain why 2° alcohols are converted to ketones. i. Be able to explain how a kinetic isotope effect can be used to determine the rate-determining step of this oxidation process. c. Explain why 1° alcohols are converted to carboxylic acids. d. Explain how ...
Carbohydrates
... right- or left-), its mirror image cannot be superimposed upon it (the mirror image and object are non-superimposable) • Non-superimposable means that the image of the original object will not be able to be made to overlap with the object, point-for-point as an exact ...
... right- or left-), its mirror image cannot be superimposed upon it (the mirror image and object are non-superimposable) • Non-superimposable means that the image of the original object will not be able to be made to overlap with the object, point-for-point as an exact ...
Full-Text PDF
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
Organic molecules with functional groups containing oxygen
... • The arrows are double headed in this case, indicating the movement of a pair of electrons • The arrows begin at a definite pair of electrons - a bond or a lone pair – and move towards a positive charge • If they move into the space between two atoms, a bond is formed • If they move out of the spac ...
... • The arrows are double headed in this case, indicating the movement of a pair of electrons • The arrows begin at a definite pair of electrons - a bond or a lone pair – and move towards a positive charge • If they move into the space between two atoms, a bond is formed • If they move out of the spac ...
Production of NMOCs and Trace Organics During the
... Ultimate NMOC yields vary substantially among MSW components Lab-scale NMOC and HAP yields are considerably lower than regulatory estimates NMOC production is characterized by an initial “burst”, followed by much more gradual release ...
... Ultimate NMOC yields vary substantially among MSW components Lab-scale NMOC and HAP yields are considerably lower than regulatory estimates NMOC production is characterized by an initial “burst”, followed by much more gradual release ...
Microsoft Word - Open Access Repository of Indian Theses
... Emulating the basic principles followed by nature to build its vast repertoire of biomolecules, organic chemists are developing many novel multifunctional building blocks to construct peptides that mimic the ordered secondary structures displayed by the biopolymers and their functions. Rationally ch ...
... Emulating the basic principles followed by nature to build its vast repertoire of biomolecules, organic chemists are developing many novel multifunctional building blocks to construct peptides that mimic the ordered secondary structures displayed by the biopolymers and their functions. Rationally ch ...
cape chemistry unit ii module i
... For example, benzoyl chloride has the formula C6H5COCl. The -COCl group is attached directly to a benzene ring. It is much less reactive than simple acyl chlorides like ethanoyl chloride. In order to get a reasonably quick reaction with benzoyl chloride, the phenol is first converted into sodium phe ...
... For example, benzoyl chloride has the formula C6H5COCl. The -COCl group is attached directly to a benzene ring. It is much less reactive than simple acyl chlorides like ethanoyl chloride. In order to get a reasonably quick reaction with benzoyl chloride, the phenol is first converted into sodium phe ...
Chapter 20. Aldehydes and Ketones
... A naïve graduate student (who for our purposes will remain nameless) set out to prepare the compound (C6H5)2CHOH by adding C6H5MgBr to benzaldehyde. To insure that the chemical yield would be high, our dedicated student prepared one mole of the Grignard reagent, added two moles of benzaldehyde, and, ...
... A naïve graduate student (who for our purposes will remain nameless) set out to prepare the compound (C6H5)2CHOH by adding C6H5MgBr to benzaldehyde. To insure that the chemical yield would be high, our dedicated student prepared one mole of the Grignard reagent, added two moles of benzaldehyde, and, ...
CARBONYL COMPOUNDS - Aldehydes and Ketones C=O C C C
... • contains copper(II) ions complexed with tartrate ions giving a blue solution • on warming, it will oxidise aliphatic (but not aromatic) aldehydes • copper(II) is reduced and a red precipitate of copper(I) oxide, Cu2O, is formed The silver mirror test is the better alternative as it works with all ...
... • contains copper(II) ions complexed with tartrate ions giving a blue solution • on warming, it will oxidise aliphatic (but not aromatic) aldehydes • copper(II) is reduced and a red precipitate of copper(I) oxide, Cu2O, is formed The silver mirror test is the better alternative as it works with all ...
Porphyrin Complex - Center for Biomimetic Systems
... as the key oxidants in the catalytic cycle of heme-containing enzymes.1 Especially, oxoiron(IV) porphyrin π-cation radicals, referred to as compound I, are believed to carry out oxygen atom transfer reactions in the catalytic oxidation of organic substrates by cytochromes P450 and iron porphyrin mod ...
... as the key oxidants in the catalytic cycle of heme-containing enzymes.1 Especially, oxoiron(IV) porphyrin π-cation radicals, referred to as compound I, are believed to carry out oxygen atom transfer reactions in the catalytic oxidation of organic substrates by cytochromes P450 and iron porphyrin mod ...
Grignard Reactions - faculty at Chemeketa
... magnesium are shifted away from the electropositive Mg to form a strongly polar covalent bond. As a result the charge distribution in the Grignard reagent is such that the organic group (R) is partially negative and the –MgX group is partially positive. This charge distribution directs the manner in ...
... magnesium are shifted away from the electropositive Mg to form a strongly polar covalent bond. As a result the charge distribution in the Grignard reagent is such that the organic group (R) is partially negative and the –MgX group is partially positive. This charge distribution directs the manner in ...
Microsoft Word
... Vicinal diols are present in many naturally occurring compounds. The ring opening of epoxides with water in the presence of acid catalysts generates synthetically useful vicdiols. However, most of these epoxide ring opening reactions involve the use of strongly acidic conditions, stoichiometric amou ...
... Vicinal diols are present in many naturally occurring compounds. The ring opening of epoxides with water in the presence of acid catalysts generates synthetically useful vicdiols. However, most of these epoxide ring opening reactions involve the use of strongly acidic conditions, stoichiometric amou ...
Mechanism
... There are two different transformations referred as the Ullmann Reaction. The "classic" Ullmann Reaction is the synthesis of symmetric biaryls via copper-catalyzed coupling. The "Ullmanntype" Reactions include copper-catalyzed Nucleophilic Aromatic Substitution between various nucleophiles (e.g. sub ...
... There are two different transformations referred as the Ullmann Reaction. The "classic" Ullmann Reaction is the synthesis of symmetric biaryls via copper-catalyzed coupling. The "Ullmanntype" Reactions include copper-catalyzed Nucleophilic Aromatic Substitution between various nucleophiles (e.g. sub ...
Synthesis of p-hydroxy alkyl benzoates
... using Toluene as azeotropic agent in presence of minimum amount of concentrated sulphuric acid with corresponding alcohol. This method helps to avoid the etherification of free hydroxyl group and polycondensation of phenol containing carboxylic acid as an impurity. The product produced by this techn ...
... using Toluene as azeotropic agent in presence of minimum amount of concentrated sulphuric acid with corresponding alcohol. This method helps to avoid the etherification of free hydroxyl group and polycondensation of phenol containing carboxylic acid as an impurity. The product produced by this techn ...
Advanced Organic Chemistry (Chapter 7)
... presence of a measurable concentration of the carbanion at any time. The relative ease of carbanion formation is judged from the rate at which exchange occurs. This ...
... presence of a measurable concentration of the carbanion at any time. The relative ease of carbanion formation is judged from the rate at which exchange occurs. This ...
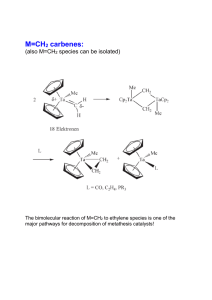
Steric protection of alkylidene is not needed:
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
Aldehid dan Keton
... Boiling Points • More polar, so higher boiling point than comparable alkane or ether. • Cannot H-bond to each other, so lower boiling point than comparable alcohol. ...
... Boiling Points • More polar, so higher boiling point than comparable alkane or ether. • Cannot H-bond to each other, so lower boiling point than comparable alcohol. ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.





![Neutral ionic liquid [BMIm]BF4 promoted highly selective](http://s1.studyres.com/store/data/017897985_1-047f9869d5604c115b21339541ccfffe-300x300.png)