as a PDF
... atoms appear in Table 11. A listing of the observed and calculated structure amplitudes for those data used in the refinements is available.’O The structure consists of tetrameric molecules [CC13C02Pd00-t-Bu]4, presenting crystallographic C2 symmetry; the twofold symmetry axis is normal to the plane ...
... atoms appear in Table 11. A listing of the observed and calculated structure amplitudes for those data used in the refinements is available.’O The structure consists of tetrameric molecules [CC13C02Pd00-t-Bu]4, presenting crystallographic C2 symmetry; the twofold symmetry axis is normal to the plane ...
class notes 4
... Acid-Base Reactions (Acid-Base Reactions Always Go) Acid: Substance that produces H+ ions in aqueous solution is the Arrhenius definition of acid. Base: Substance that produces OH- in aqueous solution is the Arrhenius definition of base. Actually a hydrogen ion is a bare proton and will associate wi ...
... Acid-Base Reactions (Acid-Base Reactions Always Go) Acid: Substance that produces H+ ions in aqueous solution is the Arrhenius definition of acid. Base: Substance that produces OH- in aqueous solution is the Arrhenius definition of base. Actually a hydrogen ion is a bare proton and will associate wi ...
indian association of chemistry teachers
... Of the interhalogen compounds, ClF3 is more reactive than BrF3, but BrF3 has higher conductance in the liquid state. The reason is that (A) BrF3 has higher molecular weight ...
... Of the interhalogen compounds, ClF3 is more reactive than BrF3, but BrF3 has higher conductance in the liquid state. The reason is that (A) BrF3 has higher molecular weight ...
Chap 4 - Bakersfield College
... – You will find it important to be able to identify an acid or base as strong or weak. – When you write an ionic equation, strong acids and bases are represented as separate ions. – Weak acids and bases are represented as undissociated “molecules” in ionic equations. ...
... – You will find it important to be able to identify an acid or base as strong or weak. – When you write an ionic equation, strong acids and bases are represented as separate ions. – Weak acids and bases are represented as undissociated “molecules” in ionic equations. ...
Unit 5 Organic Chemistry
... fuels. They are also the primary sources of hydrocarbons—compounds containing carbon atoms bonded to hydrogen atoms. Hydrocarbons are the starting points in the synthesis of thousands of products, including specific fuels, plastics, and synthetic fibres. Some hydrocarbons are obtained directly by ph ...
... fuels. They are also the primary sources of hydrocarbons—compounds containing carbon atoms bonded to hydrogen atoms. Hydrocarbons are the starting points in the synthesis of thousands of products, including specific fuels, plastics, and synthetic fibres. Some hydrocarbons are obtained directly by ph ...
CHANNELING OF SUBSTRATES AND INTERMEDIATES IN
... β-subunit inserts directly into the channel (35). Strikingly, exchange of potassium or cesium ions for sodium ions results in a movement of this Phe-280 side chain out of the tunnel, which suggests that this residue may indeed play a role as a molecular gate (35). The second site of close interactio ...
... β-subunit inserts directly into the channel (35). Strikingly, exchange of potassium or cesium ions for sodium ions results in a movement of this Phe-280 side chain out of the tunnel, which suggests that this residue may indeed play a role as a molecular gate (35). The second site of close interactio ...
Name: Period:______ Let`s make some sandwiches! Introduction: If
... 4. Notice that the labels changed from molecules to moles. This does not change the mole ratio, as a mole is simply a large number of molecules. How many molecules are in a mole? 5. In Trial #6, what reactant limits you from making more water molecules? Which reactant was present in excess and remai ...
... 4. Notice that the labels changed from molecules to moles. This does not change the mole ratio, as a mole is simply a large number of molecules. How many molecules are in a mole? 5. In Trial #6, what reactant limits you from making more water molecules? Which reactant was present in excess and remai ...
Chapter 15. Chemical Equilibrium
... This reaction is endothermic. For an endothermic reaction (∆H > 0), heat can be considered as a reactant. • Thus, adding heat causes a shift in the forward direction. • The room-temperature equilibrium mixture is placed in a beaker of ice water. • The mixture turns bright pink. • This indicates a sh ...
... This reaction is endothermic. For an endothermic reaction (∆H > 0), heat can be considered as a reactant. • Thus, adding heat causes a shift in the forward direction. • The room-temperature equilibrium mixture is placed in a beaker of ice water. • The mixture turns bright pink. • This indicates a sh ...
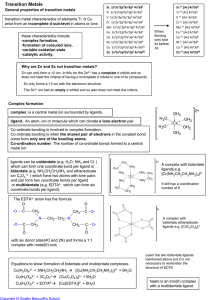
Transition Metals
... Reducing Chromium Cr3+ (green) and Cr2+ (blue) are formed by reduction of Cr2O72(orange) by zinc in acid solution, but Fe2+ will only reduce it to Cr3+ The Fe2+ and Cr2O7 2- in acid solution reaction can be used as a quantitative redox titration. This does not need an indicator Manganate redox titr ...
... Reducing Chromium Cr3+ (green) and Cr2+ (blue) are formed by reduction of Cr2O72(orange) by zinc in acid solution, but Fe2+ will only reduce it to Cr3+ The Fe2+ and Cr2O7 2- in acid solution reaction can be used as a quantitative redox titration. This does not need an indicator Manganate redox titr ...
Document
... Predicting a precipitation reaction and writing the chemical equation 1 Write the reactants for the reaction 2 Show the reactants in their ionic form, i.e., if they are soluble, write their dissociated form 3 Determine which cation-anion combination yields an insoluble compound ...
... Predicting a precipitation reaction and writing the chemical equation 1 Write the reactants for the reaction 2 Show the reactants in their ionic form, i.e., if they are soluble, write their dissociated form 3 Determine which cation-anion combination yields an insoluble compound ...
chemistry - Textbooks Online
... Alchemy was a mixture of scientific investigation and mystical quest, with strands of philosophy from Greece, China, Egypt and Arabia mixed in. The main aims of alchemy that emerged with time were the quest for the elixir of life (the drinking of which would endue the alchemist with immortality), an ...
... Alchemy was a mixture of scientific investigation and mystical quest, with strands of philosophy from Greece, China, Egypt and Arabia mixed in. The main aims of alchemy that emerged with time were the quest for the elixir of life (the drinking of which would endue the alchemist with immortality), an ...
Follow Along Notes - Jackson County School System
... Calculations involving equilibrium How to solve Equilibrium Problems: 1. Start with a balanced Chemical Equation 2. Write down the amounts (either concentration or pressure units) in an ICE table. 3. Shift the equilibrium by subtracting and adding x to either side to the equation. 4. Solve for x us ...
... Calculations involving equilibrium How to solve Equilibrium Problems: 1. Start with a balanced Chemical Equation 2. Write down the amounts (either concentration or pressure units) in an ICE table. 3. Shift the equilibrium by subtracting and adding x to either side to the equation. 4. Solve for x us ...
Syllabus Advanced Level and Advanced Subsidiary Level
... The syllabus has been constructed with a compulsory Advanced Subsidiary core. That part of the core syllabus which will be examined only in the full Advanced Level qualification is indicated in bold type. A full Advanced Level qualification requires the study of further core material together with s ...
... The syllabus has been constructed with a compulsory Advanced Subsidiary core. That part of the core syllabus which will be examined only in the full Advanced Level qualification is indicated in bold type. A full Advanced Level qualification requires the study of further core material together with s ...
Principles of Reactivity: Chemical Equilibria
... Practice Problem Consider the effect of temperature changes on the following equilibria. Does the concentration of NOCl increase or decrease at equilibrium as the temperature of the system is increased? ...
... Practice Problem Consider the effect of temperature changes on the following equilibria. Does the concentration of NOCl increase or decrease at equilibrium as the temperature of the system is increased? ...
Tro Chemistry a Molecular Approach, 3E
... chemical reactions. In Chapter 3, you learned how to write balanced chemical equations for chemical reactions. Here we will examine more closely the meaning of those balanced equations. In the second half of this chapter, we turn to describing chemical reactions that occur in water. You have probabl ...
... chemical reactions. In Chapter 3, you learned how to write balanced chemical equations for chemical reactions. Here we will examine more closely the meaning of those balanced equations. In the second half of this chapter, we turn to describing chemical reactions that occur in water. You have probabl ...
B.Sc. Industrial Chemistry
... B.Sc. Industrial Chemistry The course on B.Sc. Industrial Chemistry was introduced in the University of Delhi in 1984 and since then this course has undergone many changes and has become more comprehensive and relevant. The importance of industrial chemistry hardly needs any emphasis. It basically ...
... B.Sc. Industrial Chemistry The course on B.Sc. Industrial Chemistry was introduced in the University of Delhi in 1984 and since then this course has undergone many changes and has become more comprehensive and relevant. The importance of industrial chemistry hardly needs any emphasis. It basically ...
CHEMICAL AND PROCESS DESIGN HANDBOOK
... Thus, aniline, with a considerable excess of methyl alcohol and a catalytic amount of sulfuric acid, is heated in an autoclave at about 200o C for 5 or 6 hours at a high reaction pressure of 540 psi (3.7 MPa). Vacuum distillation is used for purification. In the alkylation of aniline to diethylanili ...
... Thus, aniline, with a considerable excess of methyl alcohol and a catalytic amount of sulfuric acid, is heated in an autoclave at about 200o C for 5 or 6 hours at a high reaction pressure of 540 psi (3.7 MPa). Vacuum distillation is used for purification. In the alkylation of aniline to diethylanili ...
ORGANIC CHEMISTRY
... CH3-CH2-CH2-COOH and CH3-CH2-CO-CH2-CH3 have roughly the same London forces and both contain a carbonyl group (C=O) so they both have a polar area. However, they differ because butanoic acid has an –OH bond so it has hydrogen bonding as an IMF. ...
... CH3-CH2-CH2-COOH and CH3-CH2-CO-CH2-CH3 have roughly the same London forces and both contain a carbonyl group (C=O) so they both have a polar area. However, they differ because butanoic acid has an –OH bond so it has hydrogen bonding as an IMF. ...
Reaction Rates/Chemical Kinetics
... Plan: We can determine the starting concentration of each species in the reaction mixture. We can then substitute the starting concentrations into the equilibrium-constant expression to calculate the reaction quotient, Qc. Comparing the magnitudes of the equilibrium constant, which is given, and the ...
... Plan: We can determine the starting concentration of each species in the reaction mixture. We can then substitute the starting concentrations into the equilibrium-constant expression to calculate the reaction quotient, Qc. Comparing the magnitudes of the equilibrium constant, which is given, and the ...
TOPIC 11 Further equilibrium 11.1 Chemical equilibrium
... When making solution A, 25 cm3 of the NaOH solution reacts with 25 cm3 of the CH3COOH solution. This forms some ethanoate ions, CH3COO-(aq), and leaves some unreacted ethanoic acid molecules, CH3COOH. CH3COOH(aq) + OH−(aq) → CH3COO−(aq) + H2O(l) So, solution A contains a mixture of a weak acid, CH3C ...
... When making solution A, 25 cm3 of the NaOH solution reacts with 25 cm3 of the CH3COOH solution. This forms some ethanoate ions, CH3COO-(aq), and leaves some unreacted ethanoic acid molecules, CH3COOH. CH3COOH(aq) + OH−(aq) → CH3COO−(aq) + H2O(l) So, solution A contains a mixture of a weak acid, CH3C ...
kcse chemistry questions
... What is the purpose of hot compressed air in this process? (1mk) Study the diagram below and answer the questions that follow. ...
... What is the purpose of hot compressed air in this process? (1mk) Study the diagram below and answer the questions that follow. ...
Question Bank - Edudel.nic.in
... A metal compound X reacts with dilute Hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle and turns lime water milky. Identify the gas evolved. Write a balanced chemical equation for the reaction if one of the compound formed is CaCl2. ...
... A metal compound X reacts with dilute Hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle and turns lime water milky. Identify the gas evolved. Write a balanced chemical equation for the reaction if one of the compound formed is CaCl2. ...
- Wiley Online Library
... nucleation are briefly reviewed in the companion paper Duplissy et al. [2016] (hereafter referred to as Paper 2). There is evidence from statistical mechanics that the failure of CNT results directly from its incorrect description of the smallest prenucleated clusters [Merikanto et al., 2007]. One of ...
... nucleation are briefly reviewed in the companion paper Duplissy et al. [2016] (hereafter referred to as Paper 2). There is evidence from statistical mechanics that the failure of CNT results directly from its incorrect description of the smallest prenucleated clusters [Merikanto et al., 2007]. One of ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.