An Introduction to Chemical Science
... any school or college in the country. During the present year the author personally supervises the work of more than 180 different pupils in chemistry. This enables him not only to assure himself that the experiments of the book are practical, but that the directions for performing them are ample. I ...
... any school or college in the country. During the present year the author personally supervises the work of more than 180 different pupils in chemistry. This enables him not only to assure himself that the experiments of the book are practical, but that the directions for performing them are ample. I ...
Review Packet Answers - Bremerton School District
... Salts of Weak Acids and Weak Bases What is the pH of a 1 M NaC2H3O2 solution? ...
... Salts of Weak Acids and Weak Bases What is the pH of a 1 M NaC2H3O2 solution? ...
CSEC Chemistry Revision Guide Answers.indd
... atoms. As it splits, two or three neutrons and a large amount of heat energy are released. The neutrons can then strike other atoms, causing them to split and release more neutrons and energy. This causes a chain reaction which releases very large amounts of heat energy that can be used to generate ...
... atoms. As it splits, two or three neutrons and a large amount of heat energy are released. The neutrons can then strike other atoms, causing them to split and release more neutrons and energy. This causes a chain reaction which releases very large amounts of heat energy that can be used to generate ...
Ciprofloxacin Hcl (Cas No 86393-32-0)
... Ciprofloxacin is 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinolinecarboxylic acid. Its empirical formula is C17H18FN3O3 and its molecular weight is 331.4. It is a faintly yellowish to light yellow crystalline substance and its chemical structure is as follows: ...
... Ciprofloxacin is 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinolinecarboxylic acid. Its empirical formula is C17H18FN3O3 and its molecular weight is 331.4. It is a faintly yellowish to light yellow crystalline substance and its chemical structure is as follows: ...
Unfamiliar Oxidation States and Tkeir Stabilization
... magnetic susceptibility measurements; and (4) X-ray studies. In some instances a combination of two or more of these has been necessary for the complete characterization of the oxidation state in question. Analytical data, in conjunction with a study of the chemical properties of the substance, freq ...
... magnetic susceptibility measurements; and (4) X-ray studies. In some instances a combination of two or more of these has been necessary for the complete characterization of the oxidation state in question. Analytical data, in conjunction with a study of the chemical properties of the substance, freq ...
The Application of Hydrolytic Enzymes for Biotransformations of
... anti-cancer properties. Lipases have been used effectively in the production of flavonoid ester derivatives that have shown both increased antioxidant and antimicrobial activity. Enzymatic esterifications of flavonoids are performed in organic solvents that increase substrate solubility of complex o ...
... anti-cancer properties. Lipases have been used effectively in the production of flavonoid ester derivatives that have shown both increased antioxidant and antimicrobial activity. Enzymatic esterifications of flavonoids are performed in organic solvents that increase substrate solubility of complex o ...
Equilibrium - Tenafly High School
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
The Equilibrium Constant
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
Equilibrium - AP Chemistry
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
Magnetic properties of nanocrystalline iron group thin film alloys
... current efficiencies in chloride baths were higher than in sulfate baths during the electrodeposition of NiFe thin film alloys. Therefore, higher current efficiency for CoFe electrodeposition in chloride baths compared to sulfate baths in our experiments may result from a similar catalytic mechanism ...
... current efficiencies in chloride baths were higher than in sulfate baths during the electrodeposition of NiFe thin film alloys. Therefore, higher current efficiency for CoFe electrodeposition in chloride baths compared to sulfate baths in our experiments may result from a similar catalytic mechanism ...
Chap15 - Bakersfield College
... –Much like water in a U-shaped tube, there is constant mixing back and forth through the lower portion of the tube. “reactants” ...
... –Much like water in a U-shaped tube, there is constant mixing back and forth through the lower portion of the tube. “reactants” ...
Practice Problems in Biomedical Organic Chemistry
... This problem set was developed to assist undergraduate students taking a one semester or two semester, nonmajors course in organic chemistry. Students in these courses often come to organic chemistry from diverse backgrounds including biology, microbiology, and a variety of medical-related fields (e ...
... This problem set was developed to assist undergraduate students taking a one semester or two semester, nonmajors course in organic chemistry. Students in these courses often come to organic chemistry from diverse backgrounds including biology, microbiology, and a variety of medical-related fields (e ...
Analytical Chemistry - University of Delhi
... Enantiomerism, Diastereomerism and Meso compounds) . Threo and erythro; D and L; cis - trans nomenclature; CIP Rules: R/ S (for upto 2 chiral carbon atoms) and E / Z Nomenclature (for upto two C=C systems). Unit 5 Aliphatic Hydrocarbons Functional group approach for the following reactions (preparat ...
... Enantiomerism, Diastereomerism and Meso compounds) . Threo and erythro; D and L; cis - trans nomenclature; CIP Rules: R/ S (for upto 2 chiral carbon atoms) and E / Z Nomenclature (for upto two C=C systems). Unit 5 Aliphatic Hydrocarbons Functional group approach for the following reactions (preparat ...
LABORATORY MANUAL CHEMISTRY 121 2013
... problems we have had in recent years, the product is sometimes yellow or orange. To determine the rate law and rate constants for this reaction, we shall measure the half-life for each experiment. When 50% of the reactant has been converted to product, the mixture (50% green and 50% red) has a chara ...
... problems we have had in recent years, the product is sometimes yellow or orange. To determine the rate law and rate constants for this reaction, we shall measure the half-life for each experiment. When 50% of the reactant has been converted to product, the mixture (50% green and 50% red) has a chara ...
General Concepts of Chemical Equilibrium
... Increasing the concentration of a reactant or removing a product will result in a shift of reaction towards more products and vice versa. d. Some reactions can be facilitated by addition of a catalyst (a substance that is not a part of reactants or products but its presence makes the reaction faster ...
... Increasing the concentration of a reactant or removing a product will result in a shift of reaction towards more products and vice versa. d. Some reactions can be facilitated by addition of a catalyst (a substance that is not a part of reactants or products but its presence makes the reaction faster ...
Supporting Information - Royal Society of Chemistry
... Figure S2. Changes in the absorption maxima are plotted against the ratio of enzyme to complex 3. The dotted line is connecting the data points; it is not a fitted curve. Inhibition data for the complexes: For determining the Ki values of complexes 2-4, the concentrations of different compounds requ ...
... Figure S2. Changes in the absorption maxima are plotted against the ratio of enzyme to complex 3. The dotted line is connecting the data points; it is not a fitted curve. Inhibition data for the complexes: For determining the Ki values of complexes 2-4, the concentrations of different compounds requ ...
General and Inorganic Chemistry
... 2. VI.2 Experimental task: Investigation of temperature- and pH- dependence of the rate of hydrolysis of acetylsalicylic acid (ASA) ................................................................................. 77 3. VI.3 Calculations .............................................................. ...
... 2. VI.2 Experimental task: Investigation of temperature- and pH- dependence of the rate of hydrolysis of acetylsalicylic acid (ASA) ................................................................................. 77 3. VI.3 Calculations .............................................................. ...
Practical Assessment of Sanitizers Steve Gray November
... • Soil can react with sanitizers and neutralize them ...
... • Soil can react with sanitizers and neutralize them ...
silbchp4
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
June 2000 Practice Diploma
... Use the following information to answer the next two questions. In order to “hide” gold during the Second World War, Nobel Prize winner Neils Bohr “dissolved” the gold, stored it in a solution, and recovered it at the end of the war. One way to “dissolve” gold is to react it with Aqua-Regia, a mixt ...
... Use the following information to answer the next two questions. In order to “hide” gold during the Second World War, Nobel Prize winner Neils Bohr “dissolved” the gold, stored it in a solution, and recovered it at the end of the war. One way to “dissolve” gold is to react it with Aqua-Regia, a mixt ...
Problem 1-2
... Henry’s law should only be used for pressures up to 5·105 Pa. It’s valid for diluted solutions and small partial pressures. ...
... Henry’s law should only be used for pressures up to 5·105 Pa. It’s valid for diluted solutions and small partial pressures. ...
Course Notes
... H + strength of the magnetic field r = radius of the path V = accelerating potential placing a detector at some point along the flight path of the ion allows its mass to charge ratio to be calculated. Since almost all the ions will have a charge of +1, the mass to charge ratio is also the mass. A ma ...
... H + strength of the magnetic field r = radius of the path V = accelerating potential placing a detector at some point along the flight path of the ion allows its mass to charge ratio to be calculated. Since almost all the ions will have a charge of +1, the mass to charge ratio is also the mass. A ma ...
Worked solutions to the problems
... materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these to their attention. For our junior colleagues who will spend many hours over the ...
... materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these to their attention. For our junior colleagues who will spend many hours over the ...
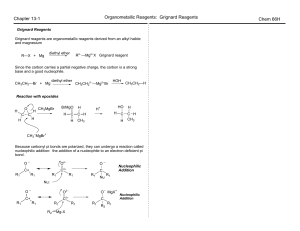
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.