Contents and Concepts Learning Objectives
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with ei ...
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with ei ...
Topic 6 Section C
... Option A —The reaction between sulphur dioxide and oxygen is reversible. The percentage yield of sulphur trioxide is about 98%. Option B —The conversion of sulphur dioxide into sulphur trioxide is carried out at 450 oC. Option C —Platinum or vanadium(V) oxide is used as a catalyst. Option D —The con ...
... Option A —The reaction between sulphur dioxide and oxygen is reversible. The percentage yield of sulphur trioxide is about 98%. Option B —The conversion of sulphur dioxide into sulphur trioxide is carried out at 450 oC. Option C —Platinum or vanadium(V) oxide is used as a catalyst. Option D —The con ...
Chap18 - Bakersfield College
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with ei ...
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with ei ...
Perfumery_Synthetics_and_Isolates_pg_283
... 4 8 Dupont and Labaune, Rome-Bertrand fils, Sci. Ind. Bull. [2], 10, 19 (1909) ; [3], 1, 42 (1910) ; [3], 3 ...
... 4 8 Dupont and Labaune, Rome-Bertrand fils, Sci. Ind. Bull. [2], 10, 19 (1909) ; [3], 1, 42 (1910) ; [3], 3 ...
Brilliant Preparatory Section, Sitamarhi
... While carrying out reaction we are often interested in knowing the number of atoms and molecules. Some times, we have to take the atoms or molecules of different reactants in a definite ratio. ...
... While carrying out reaction we are often interested in knowing the number of atoms and molecules. Some times, we have to take the atoms or molecules of different reactants in a definite ratio. ...
1412_lecture_ch16 Fall_2014
... Because the oxalate ion is conjugate to a weak acid (HC2O4-), it will react with H3O+. ...
... Because the oxalate ion is conjugate to a weak acid (HC2O4-), it will react with H3O+. ...
Contents and Concepts Learning Objectives
... two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with either the cation or the anion. • Effect of pH on Solubility • When a salt contains the ...
... two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with either the cation or the anion. • Effect of pH on Solubility • When a salt contains the ...
Solubility and Complex-ion Equilibria
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
PDF File
... (see also ref 37): The same K1/2 values were observed in concentration dependences in which the maximal rate constant for reaction varied by more than 10-fold, which was accomplished by a 2′-H substitution at position -1 and by varying the pH (38). The affinity of S or P for the ribozyme is very hig ...
... (see also ref 37): The same K1/2 values were observed in concentration dependences in which the maximal rate constant for reaction varied by more than 10-fold, which was accomplished by a 2′-H substitution at position -1 and by varying the pH (38). The affinity of S or P for the ribozyme is very hig ...
Solubility and Complex-ion Equilibria
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
PART 3-ICHO 11-15
... Bromination in the dark in the presence of iron also gives two monobromo derivatives. If the reaction is carried out to a higher degree, the formation of four dibromo derivatives may occur. Suggest the structure for compound X and for the bromination products. Write schemes for the reactions. ______ ...
... Bromination in the dark in the presence of iron also gives two monobromo derivatives. If the reaction is carried out to a higher degree, the formation of four dibromo derivatives may occur. Suggest the structure for compound X and for the bromination products. Write schemes for the reactions. ______ ...
Modification of Polycaprolactone by Titania Through a Sol
... PCL-g-AA/TiO2 hybrid also produced a broad O−H bond stretching at about 3000-3800cm−1 (Fig. 1D). This is due to the formation of hetero-associated hydrogen bonds between carboxylic acid groups of PCL-g-AA and the titanium bonded isopropyl group. In addition, acetate ligands of the PCL-g-AA/TiO2 hybr ...
... PCL-g-AA/TiO2 hybrid also produced a broad O−H bond stretching at about 3000-3800cm−1 (Fig. 1D). This is due to the formation of hetero-associated hydrogen bonds between carboxylic acid groups of PCL-g-AA and the titanium bonded isopropyl group. In addition, acetate ligands of the PCL-g-AA/TiO2 hybr ...
Introduction to Inorganic Chemistry
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
EQUILIBRIUM
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
EQUILIBRIUM
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
Revised (12 Sept 2009) Topic: Chemical Equilibrium
... and NCS−(aq) ions (making the denominator bigger). Thus, although both forward and reverse reactions occur simultaneously, the reverse reaction will predominate to decrease the Fe(NCS)2+(aq) concentration until equilibrium is established. When the reverse reaction occurs to a greater extent than the ...
... and NCS−(aq) ions (making the denominator bigger). Thus, although both forward and reverse reactions occur simultaneously, the reverse reaction will predominate to decrease the Fe(NCS)2+(aq) concentration until equilibrium is established. When the reverse reaction occurs to a greater extent than the ...
Introduction to Inorganic Chemistry
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
Specification – AS/A Level Chemistry A
... rejection) of different theories for the structure (d) deduce the numbers of protons, neutrons and of the atom from the Greeks, Dalton, electrons in: Thompson and Rutherford, Moseley, et al. (i) an atom given its atomic and mass number, (ii) an ion given its atomic number, mass number and ionic cha ...
... rejection) of different theories for the structure (d) deduce the numbers of protons, neutrons and of the atom from the Greeks, Dalton, electrons in: Thompson and Rutherford, Moseley, et al. (i) an atom given its atomic and mass number, (ii) an ion given its atomic number, mass number and ionic cha ...
Document
... A) There will be a net gain in product. B) There will be a net gain in reactant. C) There will be a net gain in both product and ...
... A) There will be a net gain in product. B) There will be a net gain in reactant. C) There will be a net gain in both product and ...
Modulated 2:1 layer silicates: Review, systematics, and
... To complete the octahedral sheet in 2: I layer structures, a secondtetrahedral sheetis inverted to opposethe first so that a similar configuration of apical oxygensand OH groups surround the octahedralcation. In l:1 layer structures, the octahedral cations complete their sixfold coordination with OH ...
... To complete the octahedral sheet in 2: I layer structures, a secondtetrahedral sheetis inverted to opposethe first so that a similar configuration of apical oxygensand OH groups surround the octahedralcation. In l:1 layer structures, the octahedral cations complete their sixfold coordination with OH ...
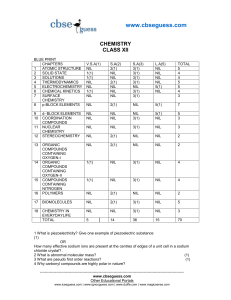
guess paper class xii
... (2) It is difficult to prepare pure amines by ammonolysis of alkyl halides. (3) Aniline is a weaker base than cyclohexyl amine. OR Explain giving reasons: 1) Alkyl amines are stronger bases than aryl amines. 2) Toluene is more easily nitrated than benzene. 3) Amino group is o,p-directing for aromati ...
... (2) It is difficult to prepare pure amines by ammonolysis of alkyl halides. (3) Aniline is a weaker base than cyclohexyl amine. OR Explain giving reasons: 1) Alkyl amines are stronger bases than aryl amines. 2) Toluene is more easily nitrated than benzene. 3) Amino group is o,p-directing for aromati ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.