![(K c ) [A] - Knockhardy](http://s1.studyres.com/store/data/011755527_1-914ea907d1ff7656ef398ad87316c94c-300x300.png)
(K c ) [A] - Knockhardy
... • construct the balanced equation, including state symbols (aq), (g) etc. • determine the number of moles of each species at equilibrium • divide moles by volume (in dm3) to get the equilibrium concentrations in mol dm-3 (If no volume is quoted, use a V; it will probably cancel out) • from the equat ...
... • construct the balanced equation, including state symbols (aq), (g) etc. • determine the number of moles of each species at equilibrium • divide moles by volume (in dm3) to get the equilibrium concentrations in mol dm-3 (If no volume is quoted, use a V; it will probably cancel out) • from the equat ...
Grade XII Unit 1 - Ethiopian Ministry of Education
... obtain a homogeneous mixture, or solution. Air is a gaseous solution, principally of two elementary substances, nitrogen and oxygen, which are physically mixed but not chemically combined. There are various practical reasons for preparing solutions. For instance, most chemical reactions are run in s ...
... obtain a homogeneous mixture, or solution. Air is a gaseous solution, principally of two elementary substances, nitrogen and oxygen, which are physically mixed but not chemically combined. There are various practical reasons for preparing solutions. For instance, most chemical reactions are run in s ...
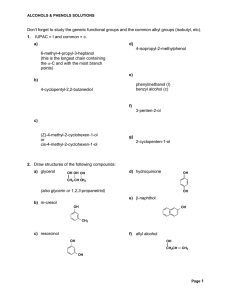
Don`t forget to study the generic functional groups and the common
... 11. This one is tougher. Note the addition of the ethyl group. The only methods we’ve learned for adding Carbon groups are 1) chain lengthening terminal alkynes, 2) FC alkylations/acylations to aromatics, 3) Grignards, organosodium and Gilman reactions. Obviously the first two don’t apply here, so l ...
... 11. This one is tougher. Note the addition of the ethyl group. The only methods we’ve learned for adding Carbon groups are 1) chain lengthening terminal alkynes, 2) FC alkylations/acylations to aromatics, 3) Grignards, organosodium and Gilman reactions. Obviously the first two don’t apply here, so l ...
Exam Review
... At various levels in the tower, trays collect mixtures of substances as they condense, each mixture containing compounds with similar boiling points. These mixtures are called petroleum fractions. The fractions with the lowest boiling points contain the smallest molecules. The low boiling points are ...
... At various levels in the tower, trays collect mixtures of substances as they condense, each mixture containing compounds with similar boiling points. These mixtures are called petroleum fractions. The fractions with the lowest boiling points contain the smallest molecules. The low boiling points are ...
1 Structure, Properties, and Preparation of Boronic Acid - Wiley-VCH
... status of boronic acids as an important class of compounds in chemistry and medicine. This chapter describes the structural and physicochemical properties of boronic acids and their many derivatives, as well as modern methods for their preparation. A brief overview of their synthetic and biological ...
... status of boronic acids as an important class of compounds in chemistry and medicine. This chapter describes the structural and physicochemical properties of boronic acids and their many derivatives, as well as modern methods for their preparation. A brief overview of their synthetic and biological ...
M.Sc. Chemistry - Periyar University
... Chirality due to helical shape, trans cyclooctene. E – Z isomerism of olefins containing one double bond and more than one double bond. Stereospecific and stereoselective synthesis with suitable examples, asymmetric synthesis – Crams rule. UNIT – IV Aliphatic Nucleophilic Substitution Reactions ...
... Chirality due to helical shape, trans cyclooctene. E – Z isomerism of olefins containing one double bond and more than one double bond. Stereospecific and stereoselective synthesis with suitable examples, asymmetric synthesis – Crams rule. UNIT – IV Aliphatic Nucleophilic Substitution Reactions ...
CHAPTER 21 ORGANIC CHEMISTRY
... A hydrocarbon is a compound composed of only carbon and hydrogen. A saturated hydrocarbon has only carbon-carbon single bonds in the molecule. An unsaturated hydrocarbon has one or more carbon-carbon multiple bonds but may also contain carbon-carbon single bonds. A normal hydrocarbon has one chain o ...
... A hydrocarbon is a compound composed of only carbon and hydrogen. A saturated hydrocarbon has only carbon-carbon single bonds in the molecule. An unsaturated hydrocarbon has one or more carbon-carbon multiple bonds but may also contain carbon-carbon single bonds. A normal hydrocarbon has one chain o ...
National German Competition
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
2014 Syllabus - Cambridge International Examinations
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
Chemistry
... Today’s students use textbooks differently than their predecessors. Chemistry, Sixth Edition is designed to map to the way students seek and process information. McMurry/Fay’s text helps students and professors get to the heart of chemistry more effectively, and helps students see the connections to ...
... Today’s students use textbooks differently than their predecessors. Chemistry, Sixth Edition is designed to map to the way students seek and process information. McMurry/Fay’s text helps students and professors get to the heart of chemistry more effectively, and helps students see the connections to ...
EDTA Titrations
... Metal-Chelate Complexes Lewis Acid/Base Chemistry Monodentate Multidentate and Chelates ...
... Metal-Chelate Complexes Lewis Acid/Base Chemistry Monodentate Multidentate and Chelates ...
sol-gel chemistry of transition metal oxides
... two stable oxidation states, namely Cr(Vl) and Cr(lll). ...
... two stable oxidation states, namely Cr(Vl) and Cr(lll). ...
Chemistry - Department of Education and Skills
... Department of Education and Science’s Equality of Opportunity Programme. The project developed out of the Department’s scheme of Intervention Projects in Physics and Chemistry which was implemented from 1985 with a view to increasing the participation of girls in the study of the physical sciences. ...
... Department of Education and Science’s Equality of Opportunity Programme. The project developed out of the Department’s scheme of Intervention Projects in Physics and Chemistry which was implemented from 1985 with a view to increasing the participation of girls in the study of the physical sciences. ...
APPROACHES TO CARBOHYDRATE-BASED CHEMICAL LIBRARIES: THE
... development has been successful, but increasing competition for shrinking profits has dictated an industry-wide search for a faster and more cost-effective approach. One such approach is structure-based rational drug design.* Conceptually, rational drug design is very simple. If one can define the t ...
... development has been successful, but increasing competition for shrinking profits has dictated an industry-wide search for a faster and more cost-effective approach. One such approach is structure-based rational drug design.* Conceptually, rational drug design is very simple. If one can define the t ...
LaBrake, Fundamentals Diagnostic Questions
... 73. Write the following word equation as a balanced chemical equation: Copper reacts with sulfuric acid to yield copper(II)sulfate, water, and sulfur dioxide. a) Cu + 2 H2SO4 CuSO4 + 2 H2O + SO2 (correct) b) 2 Cu + 3 H2S Cu2S + 3 H2O + S2O c) Cu + H2SO4 Cu2SO4 + H2O + SO2 d) 2 Cu + 2 H2SO4 ...
... 73. Write the following word equation as a balanced chemical equation: Copper reacts with sulfuric acid to yield copper(II)sulfate, water, and sulfur dioxide. a) Cu + 2 H2SO4 CuSO4 + 2 H2O + SO2 (correct) b) 2 Cu + 3 H2S Cu2S + 3 H2O + S2O c) Cu + H2SO4 Cu2SO4 + H2O + SO2 d) 2 Cu + 2 H2SO4 ...
questions based on high order thinking skill - Entrance
... Q. 4. Why water cannot be separated completely from ethyl alcohol by fractional distillation ? Ans. Ethyl alcohol and water (95.4% ethyl alcohol and 4.6% water) form constant boiling mixture (azeotrope) boiling at 351.1 °K. Hence, further water cannot be separated completely from ethyl alcohol by fr ...
... Q. 4. Why water cannot be separated completely from ethyl alcohol by fractional distillation ? Ans. Ethyl alcohol and water (95.4% ethyl alcohol and 4.6% water) form constant boiling mixture (azeotrope) boiling at 351.1 °K. Hence, further water cannot be separated completely from ethyl alcohol by fr ...
questions based on high order thinking skill
... Q. 4. Why water cannot be separated completely from ethyl alcohol by fractional distillation ? Ans. Ethyl alcohol and water (95.4% ethyl alcohol and 4.6% water) form constant boiling mixture (azeotrope) boiling at 351.1 °K. Hence, further water cannot be separated completely from ethyl alcohol by fr ...
... Q. 4. Why water cannot be separated completely from ethyl alcohol by fractional distillation ? Ans. Ethyl alcohol and water (95.4% ethyl alcohol and 4.6% water) form constant boiling mixture (azeotrope) boiling at 351.1 °K. Hence, further water cannot be separated completely from ethyl alcohol by fr ...
14.1 Dynamic Equilibrium, Keq , and the Mass Action Expression
... When making assumptions, if a reaction has a relatively small keq and a relatively large initial reactant concentration, then the concentration change (x) can often be neglected without introducing significant error. This does not mean x = 0, because then this would mean there is no reaction. It mea ...
... When making assumptions, if a reaction has a relatively small keq and a relatively large initial reactant concentration, then the concentration change (x) can often be neglected without introducing significant error. This does not mean x = 0, because then this would mean there is no reaction. It mea ...
Chapter 4 - AP Chemistry with dr hart
... Similarly, when a sulfite reacts with an acid, the products are a salt, sulfur dioxide, and water. ...
... Similarly, when a sulfite reacts with an acid, the products are a salt, sulfur dioxide, and water. ...
Contents and Concepts Learning Objectives
... separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another, and so forth. – After most of the Ba2+ ion has precipitated, strontium ...
... separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another, and so forth. – After most of the Ba2+ ion has precipitated, strontium ...
Solubility and Complex-ion Equilibria
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
... • Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another ion, and so forth. • The solubility of an insoluble salt can be manipulated by adding a species that reacts with e ...
Equilibrium
... The value of the equilibrium constant for any reaction can be determined by experiment. As detailed in the above section, the equilibrium position for a given reaction does not depend on the starting concentrations, so the equilibrium constant has the same value regardless of the initial amounts of ...
... The value of the equilibrium constant for any reaction can be determined by experiment. As detailed in the above section, the equilibrium position for a given reaction does not depend on the starting concentrations, so the equilibrium constant has the same value regardless of the initial amounts of ...
Aldehydes and ketones
... 1. Oxidation – Tollens Test - Benedicts Test 2. Reduction – Hydrogen addition – NaBH4 reagent 3. Addition of Alcohols – hemiacetal/acetal and tautomerism ...
... 1. Oxidation – Tollens Test - Benedicts Test 2. Reduction – Hydrogen addition – NaBH4 reagent 3. Addition of Alcohols – hemiacetal/acetal and tautomerism ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.