* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download EDTA Titrations
Nanofluidic circuitry wikipedia , lookup
Work (thermodynamics) wikipedia , lookup
History of electrochemistry wikipedia , lookup
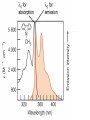
Ultraviolet–visible spectroscopy wikipedia , lookup
Transition state theory wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Enzyme catalysis wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Acid–base reaction wikipedia , lookup
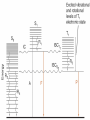
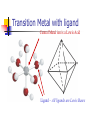
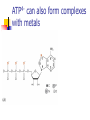
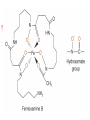
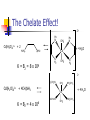
What happens to the absorbed energy? s1 t1 Energy so EDTA Titrations Outline What is EDTA? Metal-Chelate Complexes Chelate Effect EDTA ATP4- with Mg2+ Fe(NTA)23Fe(DTPA)2- Acid Base Properties aY nomenclature Conditional Formation Constants EDTA Titration Metal-Chelate Complexes Lewis Acid/Base Chemistry Monodentate Multidentate and Chelates Review: What is a Lewis Acid? Examples? And a Lewis Base? Examples? Transition Metal with ligand Central Metal ion is a Lewis Acid Ligand – All ligands are Lewis Bases Multidentate Multidentate or chelating ligand attaches to a metal ion through more than one atom is said to be multidentate, or a chelating ligand. Examples? ATP4- can also form complexes with metals Complex of Iron and NTA Fe3+ + 2 Fe(NTA)23- Medical Applications The Thalassemia Story The Chelate Effect Question: Describe in your own words, the “chelate effect”. The Chelate Effect! 2+ H2 N Cd(H2O)62+ + 2 H2N NH2 K = B2 = 8 x 109 OH2 H2 N + 4 H2O Cd N H2 OH2 N H2 2+ H3CH2N Cd(H2O)62+ + 4CH3NH2 K = B2 = 4 x 106 OH2 NH2CH3 + 4 H2O Cd H3CH2N OH2 NH2CH3 13-2 EDTA “EDTA is by far, the most widely used chelator in analytical chemistry. By direct titration or through indirect series of reactions, virtually every element of the periodic table can be measured with EDTA.” - Daniel Harris Acid/Base Properties H H (H6Y2+) Acid/Base Properties pKa = 0.0 H (H5Y+) Acid/Base Properties pKa = 0.0 pKa = 1.5 (H4Y) Acid/Base Properties pKa = 0.0 pKa = 2.0 - pKa = 1.5 (H3Y-) Acid/Base Properties pKa = 0.0 pKa = 2.0 - - pK = 2.7 a pKa = 1.5 (H2Y-2) Acid/Base Properties pKa = 2.0 - pKa = 6.16 pKa = 0.0 - pK = 2.7 a pKa = 1.5 (HY-3) Acid/Base Properties pKa = 2.0 - pKa = 10.24 pKa = 6.16 pKa = 0.0 - pK = 2.7 a pKa = 1.5 (Y-4) Fraction as Y4The fraction of EDTA in form Y4- is given as a4Fraction of EDTA ion the form Y4- [Y ] aY 4 [ EDTA] 4 Concentration in the form Y4- (13-3) Total Concentration of EDTA Fraction as Y4Equation 13-4 in text aY 4 K1 K 2 K 3 K 4 K 5 K 6 [ H ]6 [ H ]5 K1 [ H ]4 K1 K 2 [ H ]3 K1 K 2 K 3 [ H ]2 K1 K 2 K 3 K 4 [ H ]1 K1 K 2 K 3 K 4 K 5 K1 K 2 K 3 K 4 K 5 K 6 Example You make a solution of 0.10 M EDTA and you buffer the pH to (a) 10.0. What is aY4- ? (b) What is aY4- if the pH of the solution is buffered to 11.0? [Y ] aY 4 [ EDTA] 4 4 4 [Y ] 0.36(0.10M ) [Y ] 0.85(0.10M ) 4 [Y ] pH 10.0 0.036M 4 [Y ] pH 11.0 0.085M EDTA reactions with Metals Silver – Ag+ Mercury - Hg2+ Iron (III) – Fe3+ EDTA ethylenediaminetetraacetate anion => EDTA-4 => Y-4 +1 cation Ag+ + Y-4 D AgY-3 EDTA ethylenediaminetetraacetate anion => EDTA-4 => Y-4 +2 cation Hg+2 + Y-4 D HgY-2 EDTA ethylenediaminetetraacetate anion => EDTA-4 => Y-4 +3 cation Fe+3 + Y-4 D FeY-1 EDTA ethylenediaminetetraacetate anion => EDTA-4 => Y-4 +n ion M+n + Y-4 D MY(n-4)+ EDTA KMY [MY(n-4)+] = -------------[M][Y-4] KMY [MY(n-4)+] = ------------------[M+n] * a4 * [EDTA] [Y ] aY 4 [ EDTA] 4 Conditional formation constant! K'MY = KMY [MY(n-4)+] x a4 = ------------------[M+n] [EDTA] Example Calculate the concentration of Ni2+ in a solution that was prepared by mixing 50.0 mL of 0.0300 M Ni2+ with 50.0 mL of 0.0500 M EDTA. The solution was buffered to pH of 3.00. Two Parts 1. Reaction 2. Then equilibrium is established EDTA Titrations Figure 1310 Theoretical titration curves EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. Calculate the conditional constant: Equivalence Volume pCa at Initial Point pCa at Equivalence pCa at Pre-Equivalence Point pCa at Post-Equivalence Point Example Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. [CaY-2] K'CaY = KCaY * a4 = ---------------[Ca+2] * [EDTA] where aY4- = 0.36 K'CaY = at pH = 10.0 KCaY * a4 = 4.9 x 1010 * 0.36 = 1.8 KCaY = 4.9 x 1010 x 1010 EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. Equivalence Volume 1 Mole of EDTA = 1 Mole of Metal M1V1 = M2V2 (Careful of Stoichiometry) 50.0 mL (0.0500 M) = 0.1000 M (V2) V2 = 25.0 mL EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. K'CaY = 1.8 x 1010 0.00 mL EDTA added Initial Point pCa = - log[Ca+2] = - log(0.00500 M) = 2.301 EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. At 25.0 mL (Equivalence Point) Ca2+ Before After 0.0025 moles - + Y4- 0.0025 moles - -> CaY20.0025 moles What can contribute to Ca2+ “after” reaction? EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. Ca2+ + Y4- D I - C +x E CaY2- K 'CaY [CaY 2 ] [ EDTA][Ca 2 ] 0.0025moles/0.075 0.0025 moles/V L - +x -x K 'CaY 0.0333 x x2 X = [Ca2+] = 1.4 x10-6 +x +x 0.0333 –x pX = p[Ca2+] = 5.866 Pre-Equivalence Point Let’s try 15 mL EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. At 15.0 mL Ca2+ Before 0.0025 moles After 0.0010 moles + Y4- 0.0015 moles - -> CaY20.0015 moles What can contribute to Ca2+ after reaction? K’CaY = 1.8 x 1010 negligible EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. At 15.0 mL [Ca2+] = 0.0010 moles/0.065 L [Ca2+] = 0.015384 M p [Ca2+] = 1.812 Post Equivalence Point Let’s Try 28 ml EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. At 28.0 mL Ca2+ Before After 0.0025 moles - + Y4- 0.0028 moles 0.0003 moles -> CaY20.0025 moles What can contribute to Ca2+ after titration? EXAMPLE: Derive a curve (pCa as a function of volume of EDTA) for the titration of 50.0 mL of 0.0500 M Ca+2 with 0.1000 M EDTA in a solution buffered to a constant pH of 10.0. Ca2+ + Y4- CaY2- K 'CaY 0.078 L I C E - 0.0003 moles/V 0.0025 moles/V ' +x [CaY 2 ] [ EDTA][Ca 2 ] +x -x K CaY 0.03205 x (0.003846 x)( x) X = [Ca2+] = 4.6 x10-10 +x 0.003846 + x 0.03205 –x pX = p[Ca2+] = 9.334 Experimental Considerations EDTA Titration Techniques Erichrome Black T MgIn + EDTA (red) a MgEDTA + In (colorless) (blue) Figure 13-13 Guide to EDTA titrations, light color, pH range for quantitative analysis, dark area where ammonia must be present
























































![Synthesis of iron(III) EDTA complex, Na[Fe(EDTA].3H2O](http://s1.studyres.com/store/data/001239502_1-00b41f6a712e5b7594e856146fc86c1e-150x150.png)




