Unit 10 complete 2016-2017
... 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water? 2. Ethane, C2H6, can undergo combustion. a. If 4.50 moles of ethane are avai ...
... 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water? 2. Ethane, C2H6, can undergo combustion. a. If 4.50 moles of ethane are avai ...
Honors Chemistry
... 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water? 2. Ethane, C2H6, can undergo combustion. a. If 4.50 moles of ethane are avai ...
... 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water? 2. Ethane, C2H6, can undergo combustion. a. If 4.50 moles of ethane are avai ...
Chemistry_Stoichiome..
... 80. 100 mL of 10 % NaOH (w/V) is added to 100 mL of 10 % HCl (w/V). The resultant solution becomes: a) alkaline b) strongly alkaline c) acidic d) neutral 81. Calculate the molality of 1 L solution of 80 % H2SO4 (w/V), given that the density of the solution is 1.80 g mL−1 . a) 9.18 b) 8.6 c) 1.02 d) ...
... 80. 100 mL of 10 % NaOH (w/V) is added to 100 mL of 10 % HCl (w/V). The resultant solution becomes: a) alkaline b) strongly alkaline c) acidic d) neutral 81. Calculate the molality of 1 L solution of 80 % H2SO4 (w/V), given that the density of the solution is 1.80 g mL−1 . a) 9.18 b) 8.6 c) 1.02 d) ...
Problem 1-2 - IPN-Kiel
... v) Does the formation of Fe3O4 lead to a higher or to a lower calculated content of iron? Account for your answer. vi) Calculate the mass of the iron(III) chloride sample which was given into the measuring flask. ...
... v) Does the formation of Fe3O4 lead to a higher or to a lower calculated content of iron? Account for your answer. vi) Calculate the mass of the iron(III) chloride sample which was given into the measuring flask. ...
PREPARATION, STRUCTURAL STUDIES AND CHEMICAL
... First of all, I want to thank the entire University of Minnesota Duluth and its Department of Chemistry and Biochemistry for giving me the opportunity of Graduate studies. It will stay in my memories for the rest of my life. I am especially thankful to Dr. Viktor V. Zhdankin for being my advisor, fo ...
... First of all, I want to thank the entire University of Minnesota Duluth and its Department of Chemistry and Biochemistry for giving me the opportunity of Graduate studies. It will stay in my memories for the rest of my life. I am especially thankful to Dr. Viktor V. Zhdankin for being my advisor, fo ...
Class-XII, Summer assignment
... Ans: Cu2+ + 4 NH3 [Cu(NH3)4]+2 and this is deep blue in color. 10. Why does NO2 dimerise ? Ans: Due to presence one odd electron on Nitrogen of NO2 11. What is the covalence of nitrogen in N2O5 ? Ans: Covalency is 4 because N is bonded with 4 bonds ...
... Ans: Cu2+ + 4 NH3 [Cu(NH3)4]+2 and this is deep blue in color. 10. Why does NO2 dimerise ? Ans: Due to presence one odd electron on Nitrogen of NO2 11. What is the covalence of nitrogen in N2O5 ? Ans: Covalency is 4 because N is bonded with 4 bonds ...
Answer
... Under these conditions, calculate the energy per mole that is available from the splitting of ATP. With these concentrations, the reaction quotient, Q, is: ...
... Under these conditions, calculate the energy per mole that is available from the splitting of ATP. With these concentrations, the reaction quotient, Q, is: ...
fulltext
... Summary of the contributions of Karim E. Cassimjee (KC) to Papers I to V Paper I: KC supplied the idea of using a virtually irreversible half transamination reaction for active site quantification, designed and performed the active site quantification method, designed the molecular cloning, and per ...
... Summary of the contributions of Karim E. Cassimjee (KC) to Papers I to V Paper I: KC supplied the idea of using a virtually irreversible half transamination reaction for active site quantification, designed and performed the active site quantification method, designed the molecular cloning, and per ...
chemistry - Ethiopian Ministry of Education
... The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated by distance. Since chemistry is ...
... The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated by distance. Since chemistry is ...
General and Inorganic Chemistry – Laboratory Techniques
... Knowledge of students on Chemistry at the beginning of their graduate studies is rather different. Most of the students do not have proper laboratory expertise. This educational experience prompted the faculty of the institute to compile an educational material that can help students to make themsel ...
... Knowledge of students on Chemistry at the beginning of their graduate studies is rather different. Most of the students do not have proper laboratory expertise. This educational experience prompted the faculty of the institute to compile an educational material that can help students to make themsel ...
Chemistry.of Organic Compounds
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
Organic Chemistry – Who needs it?
... three double bonds distributed evenly over the six carbon atoms. This resonance of the double bond provides the compound in which it is contained with very specific characteristics: A delocalized conjugated π system, most commonly an arrangement of alternating single and double bonds. Coplanar s ...
... three double bonds distributed evenly over the six carbon atoms. This resonance of the double bond provides the compound in which it is contained with very specific characteristics: A delocalized conjugated π system, most commonly an arrangement of alternating single and double bonds. Coplanar s ...
Chemical Equilibrium
... Imagine you are stranded in a rowboat in the middle of the ocean. Suddenly, your boat springs a small leak, and you need to bail out water. You grab a bucket and begin to bail. After a few minutes, your efforts against the leak keep the water to only about half an inch, but any further bailing doesn’ ...
... Imagine you are stranded in a rowboat in the middle of the ocean. Suddenly, your boat springs a small leak, and you need to bail out water. You grab a bucket and begin to bail. After a few minutes, your efforts against the leak keep the water to only about half an inch, but any further bailing doesn’ ...
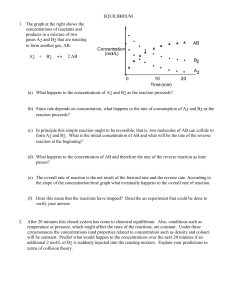
EQUILIBRIUM - SCH4U1-CCVI
... thiocyanate solution to each of these five test tubes. To test tube (1) add 5.0 mL of 0.2 mol/L iron (III) nitrate solution. This tube will be used as the standard. (b) Measure 10.0 mL of 0.2 mol/L iron (III) nitrate solution in a graduated cylinder, and fill to the 25.0 mL mark with distilled water ...
... thiocyanate solution to each of these five test tubes. To test tube (1) add 5.0 mL of 0.2 mol/L iron (III) nitrate solution. This tube will be used as the standard. (b) Measure 10.0 mL of 0.2 mol/L iron (III) nitrate solution in a graduated cylinder, and fill to the 25.0 mL mark with distilled water ...
Synthetic Polymers
... always bonded to the $-carbon of the next, as shown in the following partial structure for polystyrene. The polymer is said to be formed by head-to-tail bonding of the monomer units. ...
... always bonded to the $-carbon of the next, as shown in the following partial structure for polystyrene. The polymer is said to be formed by head-to-tail bonding of the monomer units. ...
Mole Concept - Shailendra Kumar Chemistry
... One of the earliest method for determining the molecular weight of protein was based on chemical analysis. A haemoglobin preparation was found to contain 0.335% iron. (a) If the haemoglobin molecule contain one atom of iron. What is its molecular weight ? (b) If the haemoglobin molecule contains fou ...
... One of the earliest method for determining the molecular weight of protein was based on chemical analysis. A haemoglobin preparation was found to contain 0.335% iron. (a) If the haemoglobin molecule contain one atom of iron. What is its molecular weight ? (b) If the haemoglobin molecule contains fou ...
Thermodynamics - Shailendra Kumar Chemistry
... Ba(OH)2 .8 H2O(s) + 2 NH4NO3(s) –––→ Ba(NO3)2(aq) + 2 NH3(g) + 10 H2O(l) For this reaction , ∆H° = 170.44 kJ and ∆S° = 657.4 J/K. Which of the following statements must be TRUE about the process? a. Enthalpy is favorable and entropy is unfavorable. b. Neither entropy nor entropy are favorable. c. Bo ...
... Ba(OH)2 .8 H2O(s) + 2 NH4NO3(s) –––→ Ba(NO3)2(aq) + 2 NH3(g) + 10 H2O(l) For this reaction , ∆H° = 170.44 kJ and ∆S° = 657.4 J/K. Which of the following statements must be TRUE about the process? a. Enthalpy is favorable and entropy is unfavorable. b. Neither entropy nor entropy are favorable. c. Bo ...
Reactions in Aqueous Solution
... When weak electrolytes9 dissolve, they produce relatively few ions in solution. This does not mean that the compounds do not dissolve readily in water; many weak electrolytes contain polar bonds and are therefore very soluble in a polar solvent such as water. They do not completely dissociate to for ...
... When weak electrolytes9 dissolve, they produce relatively few ions in solution. This does not mean that the compounds do not dissolve readily in water; many weak electrolytes contain polar bonds and are therefore very soluble in a polar solvent such as water. They do not completely dissociate to for ...
Chapter 4 "Reactions in Aqueous Solution"
... When weak electrolytes9 dissolve, they produce relatively few ions in solution. This does not mean that the compounds do not dissolve readily in water; many weak electrolytes contain polar bonds and are therefore very soluble in a polar solvent such as water. They do not completely dissociate to for ...
... When weak electrolytes9 dissolve, they produce relatively few ions in solution. This does not mean that the compounds do not dissolve readily in water; many weak electrolytes contain polar bonds and are therefore very soluble in a polar solvent such as water. They do not completely dissociate to for ...
Topic 7.2 Equilibrium The Position of Equilibrium
... It shortens the time needed to attain equilibrium concentrations It has no effect on the position of equilibrium, however equilibrium will be attained more quickly. ...
... It shortens the time needed to attain equilibrium concentrations It has no effect on the position of equilibrium, however equilibrium will be attained more quickly. ...
Post Lab Questions
... chemicals, or materials in the laboratory until you are instructed to do so. Do not eat, drink or chew gum. Do not use laboratory glassware as containers for food or beverage. Perform only those experiments authorized by your instructor. Never do anything in the laboratory that is not called for in ...
... chemicals, or materials in the laboratory until you are instructed to do so. Do not eat, drink or chew gum. Do not use laboratory glassware as containers for food or beverage. Perform only those experiments authorized by your instructor. Never do anything in the laboratory that is not called for in ...
An Introduction to Chemical Science
... any school or college in the country. During the present year the author personally supervises the work of more than 180 different pupils in chemistry. This enables him not only to assure himself that the experiments of the book are practical, but that the directions for performing them are ample. I ...
... any school or college in the country. During the present year the author personally supervises the work of more than 180 different pupils in chemistry. This enables him not only to assure himself that the experiments of the book are practical, but that the directions for performing them are ample. I ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.