HOTS Worksheet
... Ans. The (— CO — NH —) amide bond in nylon gets hydrolysed. Q. 2. Fibres are of crystalline structure. Why ? Ans. Fibres have strong intermolecular forces of attraction which leads to close packing of their chains and impart crystalline structure. Q. 3. Which artificial polymer is present in bubble ...
... Ans. The (— CO — NH —) amide bond in nylon gets hydrolysed. Q. 2. Fibres are of crystalline structure. Why ? Ans. Fibres have strong intermolecular forces of attraction which leads to close packing of their chains and impart crystalline structure. Q. 3. Which artificial polymer is present in bubble ...
Acrobat () verson
... Thus, ΔS = (– 9.69 × 103 J)/298 K = – 32.5 J K–1. (iii) The initial state of the gas is: 1 mole, Ti = 298K, Pi = 1 atm, Vi. After the reversible, constant pressure process is complete, the final state of the gas is 1 mole, Tf = 323 K, Vf = unknown, Pf = 1.00 atm. Following an analogous approach to t ...
... Thus, ΔS = (– 9.69 × 103 J)/298 K = – 32.5 J K–1. (iii) The initial state of the gas is: 1 mole, Ti = 298K, Pi = 1 atm, Vi. After the reversible, constant pressure process is complete, the final state of the gas is 1 mole, Tf = 323 K, Vf = unknown, Pf = 1.00 atm. Following an analogous approach to t ...
Post Lab Questions
... chemicals, or materials in the laboratory until you are instructed to do so. Do not eat, drink or chew gum. Do not use laboratory glassware as containers for food or beverage. Perform only those experiments authorized by your instructor. Never do anything in the laboratory that is not called for in ...
... chemicals, or materials in the laboratory until you are instructed to do so. Do not eat, drink or chew gum. Do not use laboratory glassware as containers for food or beverage. Perform only those experiments authorized by your instructor. Never do anything in the laboratory that is not called for in ...
Kinetic modelling of the Maillard reaction between proteins and sugars
... 1.3 Sugar degradation ...
... 1.3 Sugar degradation ...
Chemical Equilibrium is reached when
... However, in 1 L of water we have 55.5 M of water which is very large compared with the concentrations of other species in solution, and we assume that it doesn’t change during the course of a reaction. Kc = [CH3COO-][H3O+]/[CH3COOH] Kc = Kc`[H2O] Note that it is general practice not to include units ...
... However, in 1 L of water we have 55.5 M of water which is very large compared with the concentrations of other species in solution, and we assume that it doesn’t change during the course of a reaction. Kc = [CH3COO-][H3O+]/[CH3COOH] Kc = Kc`[H2O] Note that it is general practice not to include units ...
Gas-Phase Reactions of Fe (CH2O)+ and Fe (CH2S)+ with Small
... Abstract: The gas-phase reactions of Fe(CH2O)+ and Fe(CH2S)+ with a series of aliphatic alkanes were studied by Fourier transform ion cyclotron resonance (FTICR) mass spectrometry. Like bare Fe+, C-C insertion, particularly terminal C-C insertion, is predominant for the reactions of Fe(CH2O)+, while ...
... Abstract: The gas-phase reactions of Fe(CH2O)+ and Fe(CH2S)+ with a series of aliphatic alkanes were studied by Fourier transform ion cyclotron resonance (FTICR) mass spectrometry. Like bare Fe+, C-C insertion, particularly terminal C-C insertion, is predominant for the reactions of Fe(CH2O)+, while ...
ExamView - 1984 AP Chemistry Exam.tst
... If the equilibrium constant for the reaction above is 3.7 × 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, ΔG°, for this reaction? A) E° is positive and ΔG° is negative. B) E° is negative and ΔG° is positive. C) E° and ΔG° are both pos ...
... If the equilibrium constant for the reaction above is 3.7 × 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, ΔG°, for this reaction? A) E° is positive and ΔG° is negative. B) E° is negative and ΔG° is positive. C) E° and ΔG° are both pos ...
Chapter 5 Geochemical Weathering
... Finally, the process of biodegradation in soils is accompanied by the production of organic acids. Low molecular weight fatty acids such as acetate [CH3COOH] are important intermediary compounds. These, however, can be rapidly consumed by aerobic bacteria and converted to CO2. The final organic prod ...
... Finally, the process of biodegradation in soils is accompanied by the production of organic acids. Low molecular weight fatty acids such as acetate [CH3COOH] are important intermediary compounds. These, however, can be rapidly consumed by aerobic bacteria and converted to CO2. The final organic prod ...
chemistry - Brilliant Public School Sitamarhi
... An element of atomic mass 98.5 g mol–1 occurs in fcc structure. If its unit cell edge length is 500 pm and its density is 5.22 g cm–3. Calculate the value of Avogadro constant. [Ans. : 6.03 × 1023 mol–1] ...
... An element of atomic mass 98.5 g mol–1 occurs in fcc structure. If its unit cell edge length is 500 pm and its density is 5.22 g cm–3. Calculate the value of Avogadro constant. [Ans. : 6.03 × 1023 mol–1] ...
2014 International Practice Exam: Chemistry
... when you have finished. . . . Sign your name, and write today’s date. Look up when you have finished. . . . Now print your full legal name where indicated. Are there any questions? . . . Turn to the back cover and read it completely. Look up when you have finished. . . . Are there any questions? . . ...
... when you have finished. . . . Sign your name, and write today’s date. Look up when you have finished. . . . Now print your full legal name where indicated. Are there any questions? . . . Turn to the back cover and read it completely. Look up when you have finished. . . . Are there any questions? . . ...
visual problems - Western Oregon University
... a. Use the appropriate ∆G°f value(s) from Appendix 4 to calculate ∆G°rxn for the reaction known as steam–methane reforming: CH4(g) + H2O(g) → CO(g) + 3 H2(g) b. To drive this nonspontaneous reaction the CO that is produced can be oxidized to CO 2 using more steam: CO(g) + H2O(g) → CO2(g) + H2(g) Us ...
... a. Use the appropriate ∆G°f value(s) from Appendix 4 to calculate ∆G°rxn for the reaction known as steam–methane reforming: CH4(g) + H2O(g) → CO(g) + 3 H2(g) b. To drive this nonspontaneous reaction the CO that is produced can be oxidized to CO 2 using more steam: CO(g) + H2O(g) → CO2(g) + H2(g) Us ...
Exam Review Packet Table of Contents
... (i) electron-‐ electron repulsion increases (or shielding increases or the electron-‐proton ratio increases or the effective nuclear charge decreases) (ii) an extra electron generally increases the size c) t ...
... (i) electron-‐ electron repulsion increases (or shielding increases or the electron-‐proton ratio increases or the effective nuclear charge decreases) (ii) an extra electron generally increases the size c) t ...
Conceptual Integrated Science—Chapter 13
... Conceptual Integrated Science—Chapter 13 The warm air from a lit birthday candle does not rise within an orbiting space station because there is no up or down. As a result, what happens to the burning candle and why? A. The warm air surrounding the candle speeds up the rate of reaction so that the ...
... Conceptual Integrated Science—Chapter 13 The warm air from a lit birthday candle does not rise within an orbiting space station because there is no up or down. As a result, what happens to the burning candle and why? A. The warm air surrounding the candle speeds up the rate of reaction so that the ...
Stoichiometry worksheet KEY
... H2 + Cl2 → 2 HCl a) If you have 3.56 g of hydrogen, and 8.94 g chlorine, how much hydrogen chloride can you make? (Hint: One of them will be used completely, the other will have leftovers, find which is which, then do the stoichiometry.) 9.19 g HCl ...
... H2 + Cl2 → 2 HCl a) If you have 3.56 g of hydrogen, and 8.94 g chlorine, how much hydrogen chloride can you make? (Hint: One of them will be used completely, the other will have leftovers, find which is which, then do the stoichiometry.) 9.19 g HCl ...
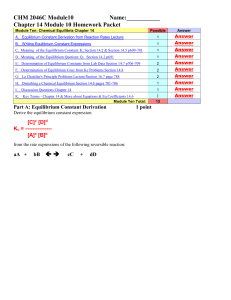
CHEM 1212 Module Ten-Chapter 16 Name
... gases), then the we say the system is in _______________equilibrium. __________________ 2. If at least one of the reactants or one of the products is not in the same phase, then we say the system is in __________________ equilibrium. __________________ 3. The state where the concentrations of the re ...
... gases), then the we say the system is in _______________equilibrium. __________________ 2. If at least one of the reactants or one of the products is not in the same phase, then we say the system is in __________________ equilibrium. __________________ 3. The state where the concentrations of the re ...
AS/A level
... The activation energy of the reaction can be calculated by finding the value of the rate constant, k, at a series of different temperatures, T. The following graph shows a plot of ln k against 1/T. The gradient, m, of the line is related to the activation energy, Ea. ...
... The activation energy of the reaction can be calculated by finding the value of the rate constant, k, at a series of different temperatures, T. The following graph shows a plot of ln k against 1/T. The gradient, m, of the line is related to the activation energy, Ea. ...
grafted chitosan - Repositorio Académico
... rapidly cooling down the reactor, then the product was precipitated by pouring the polymerization mixture into a large amount of acetone. The precipitate was filtered, washed thoroughly with acetone, and dried under vacuum at 60 8C to constant weight. The dry sample was extracted with water in a Sox ...
... rapidly cooling down the reactor, then the product was precipitated by pouring the polymerization mixture into a large amount of acetone. The precipitate was filtered, washed thoroughly with acetone, and dried under vacuum at 60 8C to constant weight. The dry sample was extracted with water in a Sox ...
Quiz contsts questions chemistry
... 56 cm3 of oxygen combine with 112 cm3 of hydrogen to form water : When 56 cm3 of H2 is passed over heated capric oxide, the latter loses 0.04 g of its weight. All measurements are done under similar conditions of temperature and pressure (at. wt., H=1, O=16). Which of the following law is obeyed by ...
... 56 cm3 of oxygen combine with 112 cm3 of hydrogen to form water : When 56 cm3 of H2 is passed over heated capric oxide, the latter loses 0.04 g of its weight. All measurements are done under similar conditions of temperature and pressure (at. wt., H=1, O=16). Which of the following law is obeyed by ...
Mole-Volume Conversion Assignment
... Yesterday’s calculations we found out that when we use 50mL of 5% acetic acid solutions, we require 3.5g of sodium bicarbonate to completely react. Trial 1: use 1.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left over: NaHCO3 all used Trial 2: use 2.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left ...
... Yesterday’s calculations we found out that when we use 50mL of 5% acetic acid solutions, we require 3.5g of sodium bicarbonate to completely react. Trial 1: use 1.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left over: NaHCO3 all used Trial 2: use 2.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left ...
caribbeanexaminations council report on candidates` work in
... various energy levels, atomic number, group number and period number. However, there were some candidates who were unable to determine group number and period number based on the electronic configuration. Also, there was some apparent confusion of mass number with proton number. Part (b) Most candid ...
... various energy levels, atomic number, group number and period number. However, there were some candidates who were unable to determine group number and period number based on the electronic configuration. Also, there was some apparent confusion of mass number with proton number. Part (b) Most candid ...
www.iitvidya.com salt analysis assignment 1. A compound on
... (iii) Its aqueous solution turns blue litmus to red. (iv) Addition of NH4OH and NaOH separately to a solution of (A) gives white precipitate which is however soluble in excess of NaOH. An inorganic compound (A), transparent like glass is a strong reducing agent. Its hydrolysis in water gives a white ...
... (iii) Its aqueous solution turns blue litmus to red. (iv) Addition of NH4OH and NaOH separately to a solution of (A) gives white precipitate which is however soluble in excess of NaOH. An inorganic compound (A), transparent like glass is a strong reducing agent. Its hydrolysis in water gives a white ...
Test bank questions
... 50.0 g of N2O4 is introduced into an evacuated 2.00 L vessel and allowed to come to equilibrium with its decomposition product, N2O4(g) 2NO2(g). For this reaction Kc = 0.133. Once the system has reached equilibrium, 5.00 g of NO 2 is injected into the vessel, and the system is allowed to equilibrate ...
... 50.0 g of N2O4 is introduced into an evacuated 2.00 L vessel and allowed to come to equilibrium with its decomposition product, N2O4(g) 2NO2(g). For this reaction Kc = 0.133. Once the system has reached equilibrium, 5.00 g of NO 2 is injected into the vessel, and the system is allowed to equilibrate ...
The role of formic acid pretreatment in improving the carboxyl
... cellulose fiber along their principal axis, are high aspect ratio fibers with diameters ranging from 3 to 100 nm, depending on their origin. Cellulose nanofibers or NFC have gained much attention for their potential uses as coatings, coating layers, and films. The most relevant and important propert ...
... cellulose fiber along their principal axis, are high aspect ratio fibers with diameters ranging from 3 to 100 nm, depending on their origin. Cellulose nanofibers or NFC have gained much attention for their potential uses as coatings, coating layers, and films. The most relevant and important propert ...
CHEMICAL EQUILIBRIUM
... Reactions with a very large equilibrium constant go essentially to completion. ...
... Reactions with a very large equilibrium constant go essentially to completion. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.