SYLLABUS 5070 Cambridge O Level Chemistry
... (e) deduce from the given melting point and boiling point the identities of substances and their purity (f) explain that the measurement of purity in substances used in everyday life, e.g. foodstuffs and drugs, is important 1.3 Identification of ions and gases (a) describe the use of aqueous sodium ...
... (e) deduce from the given melting point and boiling point the identities of substances and their purity (f) explain that the measurement of purity in substances used in everyday life, e.g. foodstuffs and drugs, is important 1.3 Identification of ions and gases (a) describe the use of aqueous sodium ...
PowerPoint Presentation - Chemical Equilibrium
... The point where the two curves cross shows the concentrations of reactants and products at equilibrium. The slopes of tangent lines to each curve at a specific time prior to reaching equilibrium are equal, but opposite, because the stoichiometry between the products and reactants is all 1:1. Equilib ...
... The point where the two curves cross shows the concentrations of reactants and products at equilibrium. The slopes of tangent lines to each curve at a specific time prior to reaching equilibrium are equal, but opposite, because the stoichiometry between the products and reactants is all 1:1. Equilib ...
chapter 16
... It must be less than 5% of the original concentration to be valid. 2. If "x" is necessary, then see if the problem may be a perfect square and thus, ease the steps of solving. (Sometimes you must use the quadratic formula!) 3. If none of the initial concentrations are zero, then Q must be calculated ...
... It must be less than 5% of the original concentration to be valid. 2. If "x" is necessary, then see if the problem may be a perfect square and thus, ease the steps of solving. (Sometimes you must use the quadratic formula!) 3. If none of the initial concentrations are zero, then Q must be calculated ...
REACTIONS IN AQUEOUS SOLUTION
... Water is a very effective solvent for ionic compounds. Although H2O is an electrically neutral molecule, the O atom is rich in electrons and has a partial negative charge, denoted by d- . Each H atom has a partial positive charge, denoted by d+ . Cations are attracted by the negative end of H2O, and ...
... Water is a very effective solvent for ionic compounds. Although H2O is an electrically neutral molecule, the O atom is rich in electrons and has a partial negative charge, denoted by d- . Each H atom has a partial positive charge, denoted by d+ . Cations are attracted by the negative end of H2O, and ...
advanced placement chemistry workbook and note set
... Atoms that contain equal numbers of electrons and protons are electrically neutral. However, atoms can easily lose or gain electrons and form ions, which are electrically-charged chemical species. The identity of the element does not change – only the electrical charge of the species changes. Recall ...
... Atoms that contain equal numbers of electrons and protons are electrically neutral. However, atoms can easily lose or gain electrons and form ions, which are electrically-charged chemical species. The identity of the element does not change – only the electrical charge of the species changes. Recall ...
LABORATORY MANUAL FOR CHEMISTRY 102
... the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum v ...
... the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum v ...
Version A
... 5. If the temperature increases from 10°C to 60°C at a constant pressure of 0.4 atmosphere, which of the processes occurs? ...
... 5. If the temperature increases from 10°C to 60°C at a constant pressure of 0.4 atmosphere, which of the processes occurs? ...
Periodic table, elements and physical chemistry
... identify and contact all copyright holders whose work is used in this paper. To avoid the issue of disclosure of answer-related information to candidates, all copyright acknowledgements are reproduced in the OCR Copyright Acknowledgements booklet. This is produced for each series of examinations and ...
... identify and contact all copyright holders whose work is used in this paper. To avoid the issue of disclosure of answer-related information to candidates, all copyright acknowledgements are reproduced in the OCR Copyright Acknowledgements booklet. This is produced for each series of examinations and ...
IUPAC System of Nomenclature
... two differences. The parent chain must include the double bond even if it makes it shorter than the others. And the parent alkene chain must be numbered from whichever end gives the first carbon of the double bond the lower of two possible numbers. Also, the location number should be given as to whe ...
... two differences. The parent chain must include the double bond even if it makes it shorter than the others. And the parent alkene chain must be numbered from whichever end gives the first carbon of the double bond the lower of two possible numbers. Also, the location number should be given as to whe ...
Derivatization reactions for the determination of amines by gas
... sponses, but pentafluorobenzoyl (PFB) derivatives possess still higher ECD responses. The ECD responses for chloro and fluoro derivatives of anilines have been compared by Lee [73]. However, chloroacetyl and PFB derivatives tend to be less volatile than the smaller perfluoroacyl derivatives, and the ...
... sponses, but pentafluorobenzoyl (PFB) derivatives possess still higher ECD responses. The ECD responses for chloro and fluoro derivatives of anilines have been compared by Lee [73]. However, chloroacetyl and PFB derivatives tend to be less volatile than the smaller perfluoroacyl derivatives, and the ...
KHARKOV STATE MEDICAL UNIVERSITY
... These elements have not very large atomic radii and intermediate values of electronegativity. These facts are favorable for strong covalent bonds formation. Remaining five macrominerals or minor biogenic elements (elements found in living systems in relatively small quantities), are Ca, Cl, Mg, K, N ...
... These elements have not very large atomic radii and intermediate values of electronegativity. These facts are favorable for strong covalent bonds formation. Remaining five macrominerals or minor biogenic elements (elements found in living systems in relatively small quantities), are Ca, Cl, Mg, K, N ...
CHEMICAL EQUILIBRIUM (Part II)II)
... Constants Knowing the equilibrium constant (Kc or ) and/or the initial concentrations of KP) and/or the initial concentrations of reactants and products for a given reaction allows you to predict several features of that reaction, such as: o whether the reaction tends to occur or not. or not ...
... Constants Knowing the equilibrium constant (Kc or ) and/or the initial concentrations of KP) and/or the initial concentrations of reactants and products for a given reaction allows you to predict several features of that reaction, such as: o whether the reaction tends to occur or not. or not ...
Chapter 2 Matter and Components F11 110pt
... alphabetically by metal name; the ions in boldface are most common. Chap 2: Matter and Nomenclature ...
... alphabetically by metal name; the ions in boldface are most common. Chap 2: Matter and Nomenclature ...
Chapter 2 Matter and Components F11 110
... older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root name for that element plus -ous and the HIGHER charged cation wil ...
... older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root name for that element plus -ous and the HIGHER charged cation wil ...
PURPOSE: To determine the value of the equilibrium constant for a
... with the iron (III) ion. What part of the water molecule is involved and what part of the iron (III) ion is involved? Identify the Lewis Acid and Lewis Base. Fe3+ ion has an empty orbital; with that empty orbital the iron can overlap its orbital to form a covalent bond. The water molecule has two lo ...
... with the iron (III) ion. What part of the water molecule is involved and what part of the iron (III) ion is involved? Identify the Lewis Acid and Lewis Base. Fe3+ ion has an empty orbital; with that empty orbital the iron can overlap its orbital to form a covalent bond. The water molecule has two lo ...
2007 Nov Paper 1 - A Level Tuition
... solutions with E o value less positive than +1.23 V will be oxidised. Hence, Cr2+(aq) will be oxidised to Cr3+(aq), while Fe(OH)2 which is formed when FeSO4 reacts with NaOH, will be oxidised to Fe(OH)3. However, [Fe(CN)6]3– remains unchanged (chemically stable) because it is already in its highest ...
... solutions with E o value less positive than +1.23 V will be oxidised. Hence, Cr2+(aq) will be oxidised to Cr3+(aq), while Fe(OH)2 which is formed when FeSO4 reacts with NaOH, will be oxidised to Fe(OH)3. However, [Fe(CN)6]3– remains unchanged (chemically stable) because it is already in its highest ...
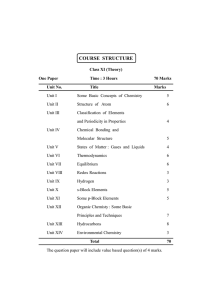
COURSE STRUCTURE
... In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ratio by mass. Law of multiple proportion : Two elements combined together ...
... In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ratio by mass. Law of multiple proportion : Two elements combined together ...
ch17
... 960°C. At this temperature, Kc = 0.036. (a) In which direction will the reaction proceed to reach equilibrium? (b) If [CH4] = 5.56 M at equilibrium, what are the equilibrium concentrations of the other substances? PLAN: (a) To find the direction of reaction we determine the initial concentrations fr ...
... 960°C. At this temperature, Kc = 0.036. (a) In which direction will the reaction proceed to reach equilibrium? (b) If [CH4] = 5.56 M at equilibrium, what are the equilibrium concentrations of the other substances? PLAN: (a) To find the direction of reaction we determine the initial concentrations fr ...
Chemical Equilibrium - Department of Chemistry
... Water can exist simultaneously in the gas and liquid phase. The vapor pressure of H2O at a given temperature is a property associated with an equilibrium condition. H2O(g) H2O(l) I2 originally dissolved in water (left) will partition between the CCl4 and H2O liquids such that [I2]CCl4/ [I2]H2O = 86. ...
... Water can exist simultaneously in the gas and liquid phase. The vapor pressure of H2O at a given temperature is a property associated with an equilibrium condition. H2O(g) H2O(l) I2 originally dissolved in water (left) will partition between the CCl4 and H2O liquids such that [I2]CCl4/ [I2]H2O = 86. ...
chromatographic study of photolysis of aqueous cyanocobalamin
... Hydroxocobalamin (solvent systems S1 and S2) was found to be the only photoproduct of cyanocobalamin at pH 1-7 alone, or in the presence of individual B/C vitamins. The relative intensity of the TLC spots appears to depend upon the rate of reaction and the added vitamin and decreases with an increas ...
... Hydroxocobalamin (solvent systems S1 and S2) was found to be the only photoproduct of cyanocobalamin at pH 1-7 alone, or in the presence of individual B/C vitamins. The relative intensity of the TLC spots appears to depend upon the rate of reaction and the added vitamin and decreases with an increas ...
CHEM 1212 Principles of Chemistry II Course Study Guide
... Study Skills and Suggestions There are lots of ideas out there and some are organized here. Most important: 1) You must study and you must practice to be successful. 2) This is college and now you are responsible to do this regardless of whether or not specifics are assigned by the professor. If th ...
... Study Skills and Suggestions There are lots of ideas out there and some are organized here. Most important: 1) You must study and you must practice to be successful. 2) This is college and now you are responsible to do this regardless of whether or not specifics are assigned by the professor. If th ...
2 - equations
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
Glycosyl amines
... to a primary, secondary or a tertiary amino group. If two glycosyl moieties are linked to a secondary amino group, the derivatives are named as bisglycosyl amines. ...
... to a primary, secondary or a tertiary amino group. If two glycosyl moieties are linked to a secondary amino group, the derivatives are named as bisglycosyl amines. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.