2009 Nov (9746) Paper 1
... In the purification process, pure water is separated from that contaminated with impurities (other molecules and ions) and so, there is an increase in orderliness; i.e. ∆S < 0. Since ∆G = ∆H – T ∆S and ∆H is zero, ∆G and ∆S, therefore, must have opposite signs. (ans) © Step-by-Step ...
... In the purification process, pure water is separated from that contaminated with impurities (other molecules and ions) and so, there is an increase in orderliness; i.e. ∆S < 0. Since ∆G = ∆H – T ∆S and ∆H is zero, ∆G and ∆S, therefore, must have opposite signs. (ans) © Step-by-Step ...
Chemsheets AS 1027
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
Section – B - About iTutoring
... (67). Which instrument is used to determine accurate pH of solution? pH meter is used to measure pH. (68). What is meant by hydrolysis constant ? Hydrolysis reaction is an equilibrium reaction and so its corresponding equilibrium constant can be calculated which is known as hydrolysis constant. (69) ...
... (67). Which instrument is used to determine accurate pH of solution? pH meter is used to measure pH. (68). What is meant by hydrolysis constant ? Hydrolysis reaction is an equilibrium reaction and so its corresponding equilibrium constant can be calculated which is known as hydrolysis constant. (69) ...
Brønsted Acidity in Metal−Organic Frameworks
... their acidity. A systematic study of mixed-metal MOFs containing several different kinds of metal ions within one SBU83 is highly desirable for developing stronger Brønsted acids based on bridging hydroxyl groups. Water molecules bound to metal sites could also result in Brønsted acidity, as exemplifi ...
... their acidity. A systematic study of mixed-metal MOFs containing several different kinds of metal ions within one SBU83 is highly desirable for developing stronger Brønsted acids based on bridging hydroxyl groups. Water molecules bound to metal sites could also result in Brønsted acidity, as exemplifi ...
1. (a) (i) 2Ca(NO3)2 → 2CaO + 4NO2 + O2 formulae correct (1
... Starting pH of (just above) 11 (1) Graph showing vertical line between pH 4 and 6 With vertical section 3–5 units in length (1) at a volume of HCl of 20 cm3 (1) Final pH of between 1 and 2 (1) Named indicator consequential on vertical part of their graph (1) Because all of its range is within the ve ...
... Starting pH of (just above) 11 (1) Graph showing vertical line between pH 4 and 6 With vertical section 3–5 units in length (1) at a volume of HCl of 20 cm3 (1) Final pH of between 1 and 2 (1) Named indicator consequential on vertical part of their graph (1) Because all of its range is within the ve ...
Writing Equilibrium Cons... and Liquids - Chemwiki
... value for including its units (which vary from case to case). Alternatively you might have to calculate equilibrium concentrations from a given value of and given starting concentrations. 1) What is and for the following reaction at equilibrium: 2) In a chemical reaction, one part chemical A(s) ...
... value for including its units (which vary from case to case). Alternatively you might have to calculate equilibrium concentrations from a given value of and given starting concentrations. 1) What is and for the following reaction at equilibrium: 2) In a chemical reaction, one part chemical A(s) ...
CHAPTER 16 ACID-BASE EQUILIBRIA AND SOLUBILITY
... This problem is greatly simplified because the concentration of the weak acid (acetic acid) is equal to the concentration of its conjugate base (acetate ion). Let’s set up a table of initial concentrations, change in concentrations, and equilibrium concentrations. ...
... This problem is greatly simplified because the concentration of the weak acid (acetic acid) is equal to the concentration of its conjugate base (acetate ion). Let’s set up a table of initial concentrations, change in concentrations, and equilibrium concentrations. ...
LABORATORY MANUAL FOR GENERAL CHEMISTRY I
... through previous experiments provides the basis for today’s chemistry courses. The information now being gathered will form the basis of future courses. There are basically two types of experiments that chemists conduct: 1 Qualitative – to determine the nature of processes, which are often unanticip ...
... through previous experiments provides the basis for today’s chemistry courses. The information now being gathered will form the basis of future courses. There are basically two types of experiments that chemists conduct: 1 Qualitative – to determine the nature of processes, which are often unanticip ...
AP Chemistry Notes and Worksheets 2014
... not tell use the actual masses of reactants and products we will use or expect to produce. For this, we need to relate the reactants and products in terms of their mole ratios. The mole ratio = moles required/moles given. o Ex. What mass of NH3 is formed when 5.38g of Li3N reacts with water accord ...
... not tell use the actual masses of reactants and products we will use or expect to produce. For this, we need to relate the reactants and products in terms of their mole ratios. The mole ratio = moles required/moles given. o Ex. What mass of NH3 is formed when 5.38g of Li3N reacts with water accord ...
Higher Chemistry Resources Guide - Glow Blogs
... The following pages show the SQA Higher Chemistry course and unit support notes with an extra column. Content new to the course from the Higher Still Higher are highlighted in green. Practitioners should refer to the SQA website for the most up to course and unit support notes. The extra column, en ...
... The following pages show the SQA Higher Chemistry course and unit support notes with an extra column. Content new to the course from the Higher Still Higher are highlighted in green. Practitioners should refer to the SQA website for the most up to course and unit support notes. The extra column, en ...
Wilhelm Ostwald, the Father of Physical Chemistry
... favourable. In order to reduce their surface to volume ratio, and thereby reduce their free energy, the small particles deposit onto large particles with time. At infinite time, we can predict that the solution will be completely devoid of small particles and there will be only one large spherical p ...
... favourable. In order to reduce their surface to volume ratio, and thereby reduce their free energy, the small particles deposit onto large particles with time. At infinite time, we can predict that the solution will be completely devoid of small particles and there will be only one large spherical p ...
Kinetic multi-layer model of aerosol surface and bulk chemistry (KM
... not require simplifying assumptions about steady-state conditions and radial mixing. The temporal evolution and concentration profiles of volatile and non-volatile species at the gas-particle interface and in the particle bulk can be modeled along with surface concentrations and gas uptake coefficie ...
... not require simplifying assumptions about steady-state conditions and radial mixing. The temporal evolution and concentration profiles of volatile and non-volatile species at the gas-particle interface and in the particle bulk can be modeled along with surface concentrations and gas uptake coefficie ...
Higher Chemistry Resources Guide - Glow Blogs
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
Ans:- (i) Gluconic acid - Kendriya Vidyalaya No.2, Kribhco, Surat
... Ans: Negative type of deviation is present. In the negative deviation the solute-solution (A-A) interaction and solvent-solvent (B-B) interaction will be weaker than solute-solvent(A-B ) interaction. Since the new forces are stronger therefore heat is evolved and solution becomes warm. Q13. Which ty ...
... Ans: Negative type of deviation is present. In the negative deviation the solute-solution (A-A) interaction and solvent-solvent (B-B) interaction will be weaker than solute-solvent(A-B ) interaction. Since the new forces are stronger therefore heat is evolved and solution becomes warm. Q13. Which ty ...
Nordonia Hills City Schools Honors Chemistry Course of Study
... In a chemical process, recognize that there is an ideal ratio of reactants. 2. Apply ideal ratio concept to reaction coefficients in a balanced equation. 3. Solve stoichiometric problems involving moles and mass. 4. Identify limiting reactant to determine the quantity of product (s) formed. 5. Calcu ...
... In a chemical process, recognize that there is an ideal ratio of reactants. 2. Apply ideal ratio concept to reaction coefficients in a balanced equation. 3. Solve stoichiometric problems involving moles and mass. 4. Identify limiting reactant to determine the quantity of product (s) formed. 5. Calcu ...
answer ch6 - Mr Khaled Nasr
... (14)A chemical process in which the concentration of a solution can be determined by using standard solution of known concentration. (15)A reaction which is used for the determination of the concentration of acids and bases. (16)A reaction which is used for the determination of the concentration of ...
... (14)A chemical process in which the concentration of a solution can be determined by using standard solution of known concentration. (15)A reaction which is used for the determination of the concentration of acids and bases. (16)A reaction which is used for the determination of the concentration of ...
L A B O
... through previous experiments provides the basis for today’s chemistry courses. The information now being gathered will form the basis of future courses. There are basically two types of experiments that chemists conduct: 1 Qualitative – to determine the nature of processes, which are often unanticip ...
... through previous experiments provides the basis for today’s chemistry courses. The information now being gathered will form the basis of future courses. There are basically two types of experiments that chemists conduct: 1 Qualitative – to determine the nature of processes, which are often unanticip ...
Chemical Reactions
... • The problem with word equations is they do not actually show the number of atoms or molecules of each substance… formulas would have to be written out for this to happen. (Absent? We looked at examples of these in class) ...
... • The problem with word equations is they do not actually show the number of atoms or molecules of each substance… formulas would have to be written out for this to happen. (Absent? We looked at examples of these in class) ...
Chapter 7 - NordoniaHonorsChemistry
... Under appropriate conditions at 1000°C, ammonia gas reacts with oxygen gas to produce gaseous nitrogen monoxide and steam ...
... Under appropriate conditions at 1000°C, ammonia gas reacts with oxygen gas to produce gaseous nitrogen monoxide and steam ...
Holt Modern Chemistry Workbook: ch 11
... unit named after the French mathematician and philosopher Blaise Pascal. One pascal, Pa, is equal to the pressure exerted by a force of 1 N acting on an area of 1 m 2 . In many situations, it is more convenient to use the unit kilopascal, kPa. For example, one atmosphere of pressure, 1 atm, is e ...
... unit named after the French mathematician and philosopher Blaise Pascal. One pascal, Pa, is equal to the pressure exerted by a force of 1 N acting on an area of 1 m 2 . In many situations, it is more convenient to use the unit kilopascal, kPa. For example, one atmosphere of pressure, 1 atm, is e ...
Support Material
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
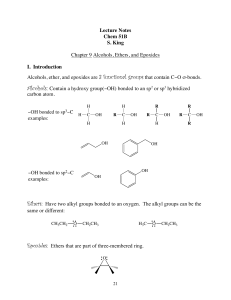
Lecture Notes Chem 51B S. King Chapter 9 Alcohols, Ethers, and
... The mechanism depends on the structure of the alcohol. Under these strongly acidic conditions, both 2° & 3° alcohols undergo an E1 reaction, whereas 1° alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make ...
... The mechanism depends on the structure of the alcohol. Under these strongly acidic conditions, both 2° & 3° alcohols undergo an E1 reaction, whereas 1° alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.