Prediction of Hydrolysis Pathways and Kinetics for Antibiotics under
... OH− attacks on the C8 site in the initial step. Subsequently, H of OH− transfers directly to the N5 site in the direct process; while in the indirect process, H first transfers to the carboxyl group and then further to the N5 site. According to Figure 3(c,d), the addition of OH− to the C8 site is a r ...
... OH− attacks on the C8 site in the initial step. Subsequently, H of OH− transfers directly to the N5 site in the direct process; while in the indirect process, H first transfers to the carboxyl group and then further to the N5 site. According to Figure 3(c,d), the addition of OH− to the C8 site is a r ...
4Chemical Quantities and Aqueous Reactions
... chemical reaction, the tomato sauce would be the limiting reactant, the reactant that limits the amount of product in a chemical reaction. Notice that the limiting reactant is simply the reactant that makes the least amount of product. Reactants that do not limit the amount of product—such as the cr ...
... chemical reaction, the tomato sauce would be the limiting reactant, the reactant that limits the amount of product in a chemical reaction. Notice that the limiting reactant is simply the reactant that makes the least amount of product. Reactants that do not limit the amount of product—such as the cr ...
Power Point for Equilibrium
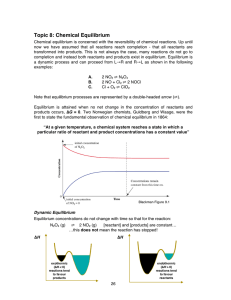
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
... • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO2 builds up, there is a chance that two NO2 molecules will coll ...
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... with the eye, the most effective first aid is the immediate flushing of the eye with copious amounts of tap water. You are seldom more than a few seconds from a faucet. Continue flushing for at least fifteen minutes and then consult a physician at once. If your laboratory is equipped with eye founta ...
... with the eye, the most effective first aid is the immediate flushing of the eye with copious amounts of tap water. You are seldom more than a few seconds from a faucet. Continue flushing for at least fifteen minutes and then consult a physician at once. If your laboratory is equipped with eye founta ...
chemistry-subject test5 w. solutions
... The ideal gas law assumes (among other things) that there are no intermolecular forces among the gas particles, which hold the molecules tighter together, leading to a smaller volume than that predicted by the ideal gas law. Intermolecular forces among neutral particles are due to hydrogen bonding, ...
... The ideal gas law assumes (among other things) that there are no intermolecular forces among the gas particles, which hold the molecules tighter together, leading to a smaller volume than that predicted by the ideal gas law. Intermolecular forces among neutral particles are due to hydrogen bonding, ...
SQA Advanced Higher Chemistry Unit 2 Principles of Chemical
... Which of the following statements applies to this equation? 1. Calcium carbonate reacts with hydrochloric acid to produce calcium chloride solution, water and carbon dioxide. 2. One formula unit of calcium carbonate reacts with two formula units of hydrochloric acid to produce one formula unit each ...
... Which of the following statements applies to this equation? 1. Calcium carbonate reacts with hydrochloric acid to produce calcium chloride solution, water and carbon dioxide. 2. One formula unit of calcium carbonate reacts with two formula units of hydrochloric acid to produce one formula unit each ...
Review Unit 8 Test (Chp 15,17)
... Greater pressure of reactant initially (Q = 0/1.00 = 0) so forward rate is faster due to greater collision frequency of reactant particles. The forward rate slows over time as reactant is consumed and there is a lower collision frequency of reactant particles, but it does not reach zero b/c more rea ...
... Greater pressure of reactant initially (Q = 0/1.00 = 0) so forward rate is faster due to greater collision frequency of reactant particles. The forward rate slows over time as reactant is consumed and there is a lower collision frequency of reactant particles, but it does not reach zero b/c more rea ...
Chem13-14PrecipABNeut
... two if possible. When one becomes broken or lost, you will have a familiar backup if the bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calculator with a 1/x or x-1 , yx or ^ , log or 10x , and ln functions will be sufficient for most ...
... two if possible. When one becomes broken or lost, you will have a familiar backup if the bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calculator with a 1/x or x-1 , yx or ^ , log or 10x , and ln functions will be sufficient for most ...
ap 2005 chemistry_b scoring guidelines - AP Central
... educational organizations. Each year, the College Board serves over three and a half million students and their parents, 23,000 high schools, and 3,500 colleges through major programs and services in college admissions, guidance, assessment, financial aid, enrollment, and teaching and learning. Amon ...
... educational organizations. Each year, the College Board serves over three and a half million students and their parents, 23,000 high schools, and 3,500 colleges through major programs and services in college admissions, guidance, assessment, financial aid, enrollment, and teaching and learning. Amon ...
Multiple-choice questions : 1. Which of the following solutions
... = 0.0315 mol [1] From the equation, mole ratio of H2SO4 : Na2CO3 = 1 : 1 Number of moles of Na2CO3 in the sample = 0.0315 mol [1] Molar mass of Na2CO3 = 106.0 g mol−1 Mass of Na2CO3 in the sample = 0.0315 × 106.0 g = 3.339 g [1] Percentage purity of the Na2CO3 sample ...
... = 0.0315 mol [1] From the equation, mole ratio of H2SO4 : Na2CO3 = 1 : 1 Number of moles of Na2CO3 in the sample = 0.0315 mol [1] Molar mass of Na2CO3 = 106.0 g mol−1 Mass of Na2CO3 in the sample = 0.0315 × 106.0 g = 3.339 g [1] Percentage purity of the Na2CO3 sample ...
document
... 5. Halogens: The oxidation number of fluorine is −1. Each of the other halogens (Cl, Br, I) has an oxidation number of −1 in binary compounds, except when the other element is another halogen above it in the periodic table or the other element is oxygen. 6. Compounds and ions: The sum of the oxidat ...
... 5. Halogens: The oxidation number of fluorine is −1. Each of the other halogens (Cl, Br, I) has an oxidation number of −1 in binary compounds, except when the other element is another halogen above it in the periodic table or the other element is oxygen. 6. Compounds and ions: The sum of the oxidat ...
Name:
... 3. What are the allowed values of ml for an electron with each orbital-shape quantum number. a) l = 3 b) l = 1 4. What are the allowed values of l for an electron with each principal quantum number. a) n = 4 b) n = 6 5. Explain what is wrong with each set of quantum numbers. a) n = 3, l = 3, ml = 2; ...
... 3. What are the allowed values of ml for an electron with each orbital-shape quantum number. a) l = 3 b) l = 1 4. What are the allowed values of l for an electron with each principal quantum number. a) n = 4 b) n = 6 5. Explain what is wrong with each set of quantum numbers. a) n = 3, l = 3, ml = 2; ...
1 Introduction
... Nearly a whole kilogram of waste for every kilogram of product! Remember, this is for the ideal case of 100% yield and 100% selectivity. In real life, the E-factor is usually much higher, because product yields are less than 100% and the reagents are often used in excess. Furthermore, in many cases ...
... Nearly a whole kilogram of waste for every kilogram of product! Remember, this is for the ideal case of 100% yield and 100% selectivity. In real life, the E-factor is usually much higher, because product yields are less than 100% and the reagents are often used in excess. Furthermore, in many cases ...
Modern inorganic chemistry
... Holliday. This new book, like its predecessor, should also be of value in first-year tertiary level chemistry courses. The new syllabuses have made it possible to go much further in systematising and explaining the facts of inorganic chemistry, and in this book the first four chapters—-the periodic ...
... Holliday. This new book, like its predecessor, should also be of value in first-year tertiary level chemistry courses. The new syllabuses have made it possible to go much further in systematising and explaining the facts of inorganic chemistry, and in this book the first four chapters—-the periodic ...
chapter 21 chemistry of the main-group elements i
... Once we return to Mg(OH)2 from MgSO4, the other substances can be made by the indicated pathways. The return reaction is: MgSO 4 (aq) + 2 NaOH(aq) Mg(OH) 2 (s) + Na 2SO 4 (aq). Then the other reactions are ...
... Once we return to Mg(OH)2 from MgSO4, the other substances can be made by the indicated pathways. The return reaction is: MgSO 4 (aq) + 2 NaOH(aq) Mg(OH) 2 (s) + Na 2SO 4 (aq). Then the other reactions are ...
The Carbonyl Group - Angelo State University
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
Chemistry written examination 1 2008–2011
... Much research is taking place in Australia into the field of Proteomics. Proteomics is the large scale study of the proteins present in a living organism. The DNA of a cell provides the blueprint for the assembly of the primary structure of proteins, the large biomolecules essential to life. In huma ...
... Much research is taking place in Australia into the field of Proteomics. Proteomics is the large scale study of the proteins present in a living organism. The DNA of a cell provides the blueprint for the assembly of the primary structure of proteins, the large biomolecules essential to life. In huma ...
chemical reaction equation - parmod cobra insititution.
... This reaction is a decomposition reaction (B) ZnCO3(s) ZnO(s) + CO2(g) This reaction is a double-displacement reaction. Why should a magnesium ribbon be cleaned before burning in air? Magnesium reacts with the constituent gases of the atmosphere to from various compound which get deposited over ...
... This reaction is a decomposition reaction (B) ZnCO3(s) ZnO(s) + CO2(g) This reaction is a double-displacement reaction. Why should a magnesium ribbon be cleaned before burning in air? Magnesium reacts with the constituent gases of the atmosphere to from various compound which get deposited over ...
4 Expressing and Measuring Chemical Change
... Historical evidence connects Joseph Priestley’s experiments with oxygen with another famous chemist, Antoine Lavoisier. Lavoisier’s experiments were more quantitative than those of Priestley. That is, Lavoisier liked to measure the volumes and masses of the chemicals he studied. Lavoisier is general ...
... Historical evidence connects Joseph Priestley’s experiments with oxygen with another famous chemist, Antoine Lavoisier. Lavoisier’s experiments were more quantitative than those of Priestley. That is, Lavoisier liked to measure the volumes and masses of the chemicals he studied. Lavoisier is general ...
Equilibrium notes (complete)
... Think of the water in a bathtub representing a system at equilibrium with the reactants at one side of the tub and the products at the other. • If you dump some water at one end of the tub the water flows towards the other end of the tub to reestablish equilibrium • If you scoop out some water at on ...
... Think of the water in a bathtub representing a system at equilibrium with the reactants at one side of the tub and the products at the other. • If you dump some water at one end of the tub the water flows towards the other end of the tub to reestablish equilibrium • If you scoop out some water at on ...
Stoichiometry File
... tremendous number of chemical compounds exist in nature and undergo myriad reactions. By building and exploiting a systematic understanding of reactivity, chemists also have produced an impressive array of man-made compounds. Many of the explosives and polymers that we’ve encountered in the last two ...
... tremendous number of chemical compounds exist in nature and undergo myriad reactions. By building and exploiting a systematic understanding of reactivity, chemists also have produced an impressive array of man-made compounds. Many of the explosives and polymers that we’ve encountered in the last two ...
Chapter 9: Non-aqueous media
... NH3 ) encourage acids to ionize in them. Thus, in a basic solvent, solvent all acids are strong. The solvent is said to exhibit a levelling effect on the acid, since the strength of the dissolved acid cannot exceed that of the protonated solvent. For example, in aqueous solution, no acidic species c ...
... NH3 ) encourage acids to ionize in them. Thus, in a basic solvent, solvent all acids are strong. The solvent is said to exhibit a levelling effect on the acid, since the strength of the dissolved acid cannot exceed that of the protonated solvent. For example, in aqueous solution, no acidic species c ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.