Experiment 7: Determination of the concentration of a solution of an
... for‐vacations, and studying‐for‐tests‐the‐following‐day do not constitute valid reasons. Students who feel their reasons are valid should see the lecturer‐in‐charge beforehand to make arrangements. Please keep in mind that a DP certificate will be refused to any student wh ...
... for‐vacations, and studying‐for‐tests‐the‐following‐day do not constitute valid reasons. Students who feel their reasons are valid should see the lecturer‐in‐charge beforehand to make arrangements. Please keep in mind that a DP certificate will be refused to any student wh ...
Chemistry (Revised)
... FeS(s) + 2HCl(aq) → FeCl2(aq) + H2S(g) (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of ...
... FeS(s) + 2HCl(aq) → FeCl2(aq) + H2S(g) (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of ...
B.Sc. (Hons.) Chemistry
... b. Study the variation of viscosity of sucrose solution with the concentration of solute. 3. Indexing of a given powder diffraction pattern of a cubic crystalline system. 4. pH metry a. Study the effect on pH of addition of HCl/NaOH to solutions of acetic acid, sodium acetate and their mixtures. b. ...
... b. Study the variation of viscosity of sucrose solution with the concentration of solute. 3. Indexing of a given powder diffraction pattern of a cubic crystalline system. 4. pH metry a. Study the effect on pH of addition of HCl/NaOH to solutions of acetic acid, sodium acetate and their mixtures. b. ...
Lab Manual (Eng. Medium)
... small quantities of a liquid. Suction is required to draw the liquid into a pipette. However, suction by mouth is unwise because of the danger of drawing toxic or corrosive liquids into the mouth. An adaptor should be used to pipette out harmful/corrosive liquids. It is used as follows: 1. Insert th ...
... small quantities of a liquid. Suction is required to draw the liquid into a pipette. However, suction by mouth is unwise because of the danger of drawing toxic or corrosive liquids into the mouth. An adaptor should be used to pipette out harmful/corrosive liquids. It is used as follows: 1. Insert th ...
2013 - SQA
... FeS(s) + 2HCl(aq) → FeCl2(aq) + H2S(g) (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of ...
... FeS(s) + 2HCl(aq) → FeCl2(aq) + H2S(g) (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of ...
MEDICAL CHEMISTRY STUDY GUIDE
... ions or molecules. This term is usually used to describe homogeneous mixtures of two or more liquids or of a liquid and one or more solids. Solutions may exist as gases, liquids, or solids. Nonreactive gases can mix in all proportions to give a gaseous solution. Liquid solutions are the most common ...
... ions or molecules. This term is usually used to describe homogeneous mixtures of two or more liquids or of a liquid and one or more solids. Solutions may exist as gases, liquids, or solids. Nonreactive gases can mix in all proportions to give a gaseous solution. Liquid solutions are the most common ...
Basic chemistry help is available here for high school or college
... problem, you need to ASK FOR HELP from a student or instructor. There is some help in math for you in Chemtutor, but if you have live help who can emphasize the points your instructor is requiring, that can be as useful. Many times your instructor will suggest a problem solving technique to you. Lea ...
... problem, you need to ASK FOR HELP from a student or instructor. There is some help in math for you in Chemtutor, but if you have live help who can emphasize the points your instructor is requiring, that can be as useful. Many times your instructor will suggest a problem solving technique to you. Lea ...
Solutions Manual
... Cracking is used to produce hydrocarbons with lower molecular mass which have greater market demand, e.g. petrol, branch-chained alkanes to improve the performance of petrol; and to produce ethene which can be used as a starting ...
... Cracking is used to produce hydrocarbons with lower molecular mass which have greater market demand, e.g. petrol, branch-chained alkanes to improve the performance of petrol; and to produce ethene which can be used as a starting ...
STOICHIOMETRY via ChemLog - Small
... H2O is supplied, there is an excess of H2O. So CaC2 (s) is the limiting reactant and all of it can react. ...
... H2O is supplied, there is an excess of H2O. So CaC2 (s) is the limiting reactant and all of it can react. ...
engineering chemistry
... An Atom is the smallest invisible particle of element, having all the characteristics of the parent element, which can neither be created nor destroyed by any chemical change. It cannot exist freely. It is the ultimate particle of an element, which may or may not have independent existence. The atom ...
... An Atom is the smallest invisible particle of element, having all the characteristics of the parent element, which can neither be created nor destroyed by any chemical change. It cannot exist freely. It is the ultimate particle of an element, which may or may not have independent existence. The atom ...
(omit), and Epoxides
... Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ...
... Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ...
Document
... [3] This defect occurs if cation and anion having similar size with high coordination number. [4] It is found in NaCl (there is one schottky defect for 1016 ions. One c.c of sodium chloride contains 1022 ) ions. Therefore, one cubic centimetre (c.c.) of NaCl possesses 106 Schottky pair of ions. # Fr ...
... [3] This defect occurs if cation and anion having similar size with high coordination number. [4] It is found in NaCl (there is one schottky defect for 1016 ions. One c.c of sodium chloride contains 1022 ) ions. Therefore, one cubic centimetre (c.c.) of NaCl possesses 106 Schottky pair of ions. # Fr ...
Soln Chem 2008Nov(9746)
... Boiling point of Group VII elements increases from Cl2 to I2 due to stronger intermolecular van der Waals' forces as the number of electrons increases from Cl2 to I2. From Cl to I, electron affinity becomes less negative due to the increase in atomic size and hence, weaker attraction for the additio ...
... Boiling point of Group VII elements increases from Cl2 to I2 due to stronger intermolecular van der Waals' forces as the number of electrons increases from Cl2 to I2. From Cl to I, electron affinity becomes less negative due to the increase in atomic size and hence, weaker attraction for the additio ...
content - Thesis Scientist
... Phenol, 0.2% solution of phenol acts as antiseptic where as 1% solution acts as disinfectant. In haloarenes C—X bond acquires a partial double bond character due to resonance. As a result the bond cleavage in haloarenes is difficult than haloalkanes and therefore, they are less reactive towards nucl ...
... Phenol, 0.2% solution of phenol acts as antiseptic where as 1% solution acts as disinfectant. In haloarenes C—X bond acquires a partial double bond character due to resonance. As a result the bond cleavage in haloarenes is difficult than haloalkanes and therefore, they are less reactive towards nucl ...
The Mole
... What if we had 3 moles of oxygen, how much hydrogen would we need to react and how much water would we get? 6 mol H2, 6 mol H2O What if we had 50 moles of hydrogen, how much oxygen would we need and how much water produced? 25 mol O2, 50 mol H2O ...
... What if we had 3 moles of oxygen, how much hydrogen would we need to react and how much water would we get? 6 mol H2, 6 mol H2O What if we had 50 moles of hydrogen, how much oxygen would we need and how much water produced? 25 mol O2, 50 mol H2O ...
Ternary nucleation of inorganic acids, ammonia, and water
... rest of the paper, the H2 SO4 concentration refers to the total concentration of H2 SO4 共including H2 O–H2 SO4 hydrates兲. Vapors containing nitric acid or HCl are most affected by ammonia. The nucleation rate increases over ten orders of magnitude if the ammonia concentration increases just one orde ...
... rest of the paper, the H2 SO4 concentration refers to the total concentration of H2 SO4 共including H2 O–H2 SO4 hydrates兲. Vapors containing nitric acid or HCl are most affected by ammonia. The nucleation rate increases over ten orders of magnitude if the ammonia concentration increases just one orde ...
COMPARATIVE EVALUATION OF TCF BLEACHED
... On the other hand alkaline cooking conditions promote rather uniform pulping reactions due to the high swelling properties. Hamilton and Thompson have studied the main differentes in the carbohydrate constituents of wood celluloses prepared by the sulfite and kraft pulping processes [4]. According t ...
... On the other hand alkaline cooking conditions promote rather uniform pulping reactions due to the high swelling properties. Hamilton and Thompson have studied the main differentes in the carbohydrate constituents of wood celluloses prepared by the sulfite and kraft pulping processes [4]. According t ...
KCET – CHEMISTRY – 2016 - Medicine.careers360.com
... 12. Replacement of Cl of Chlorobenzene to give phenol requires drastic conditions, but Cl of 2, 4dinitro chlorobenene is readily replaced. This is because 1) –NO2 group makes the ring electron rich at ortho and para positions ...
... 12. Replacement of Cl of Chlorobenzene to give phenol requires drastic conditions, but Cl of 2, 4dinitro chlorobenene is readily replaced. This is because 1) –NO2 group makes the ring electron rich at ortho and para positions ...
Chapter 16 Controlling the yield of reactions
... By referring to the equilibrium: H2O(g) H2O(l) explain what is meant by the ‘dynamic nature’ of chemical equilibrium and why wet clothes in a closed laundry bag do not dry. When the bag in part a is opened the clothes begin to dry. Is this due to an equilibrium process? Explain your answer. ...
... By referring to the equilibrium: H2O(g) H2O(l) explain what is meant by the ‘dynamic nature’ of chemical equilibrium and why wet clothes in a closed laundry bag do not dry. When the bag in part a is opened the clothes begin to dry. Is this due to an equilibrium process? Explain your answer. ...
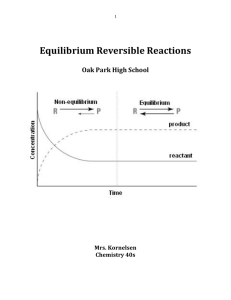
Equilibrium Reversible Reactions
... If we combine the two reactants A and B, the forward reaction starts immediately; then, as the products C and D begin to build up, the reverse process gets underway. As the reaction proceeds, the rate of the forward reaction diminishes while that of the reverse reaction increases. Eventually the two ...
... If we combine the two reactants A and B, the forward reaction starts immediately; then, as the products C and D begin to build up, the reverse process gets underway. As the reaction proceeds, the rate of the forward reaction diminishes while that of the reverse reaction increases. Eventually the two ...
File
... 18 Total removal of the pollutant sulfur dioxide, SO2, is difficult, both for economic and technical reasons. The quantities emitted from furnace chimneys can be lowered by using desulfurisation plants. The gases are scrubbed (washed) with calcium hydroxide to remove the SO2. What is the main produc ...
... 18 Total removal of the pollutant sulfur dioxide, SO2, is difficult, both for economic and technical reasons. The quantities emitted from furnace chimneys can be lowered by using desulfurisation plants. The gases are scrubbed (washed) with calcium hydroxide to remove the SO2. What is the main produc ...
2008 Equilibrium -- without math (PowerPoint 13 MB)
... Adding helium to the equilibrium mixture at constant volume increases the total gas pressure and decreases the mole fractions of both NO2 and N2O4; but the partial pressure of each gas, given by the product of its mole fraction and total pressure, does not change. Thus the presence of an inert gas d ...
... Adding helium to the equilibrium mixture at constant volume increases the total gas pressure and decreases the mole fractions of both NO2 and N2O4; but the partial pressure of each gas, given by the product of its mole fraction and total pressure, does not change. Thus the presence of an inert gas d ...
Rubidium
... It occurs naturally in the minerals leucite, pollucite, and zinnwaldite, which contains traces of up to 1% of its oxide. Lepidolite contains 1.5% rubidium and this is the commercial source of the element. Some potassium minerals and potassium chlorides also contain the element in commercially signif ...
... It occurs naturally in the minerals leucite, pollucite, and zinnwaldite, which contains traces of up to 1% of its oxide. Lepidolite contains 1.5% rubidium and this is the commercial source of the element. Some potassium minerals and potassium chlorides also contain the element in commercially signif ...
AS Chemistry Teacher Handbook
... The guide is not intended as a comprehensive reference, but as support for professional teachers to develop stimulating and exciting courses tailored to the needs and skills of their own learners in their particular institutions. The guide offers assistance to teachers with regard to the depth of co ...
... The guide is not intended as a comprehensive reference, but as support for professional teachers to develop stimulating and exciting courses tailored to the needs and skills of their own learners in their particular institutions. The guide offers assistance to teachers with regard to the depth of co ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.