No Slide Title
... Making protein from mRNA Most genes encode for proteins -some make RNA as end product ...
... Making protein from mRNA Most genes encode for proteins -some make RNA as end product ...
De niet-covalente interacties
... • Association of apolar groups/molecules in water results in the release of water molecules that surround the apolar surface in a stiff, ice-like structure. • The released water molecules have more possibilities to interact with other water molecules in solution. • This results in an increase of the ...
... • Association of apolar groups/molecules in water results in the release of water molecules that surround the apolar surface in a stiff, ice-like structure. • The released water molecules have more possibilities to interact with other water molecules in solution. • This results in an increase of the ...
Crustacean Physiology in Ribeirão Preto
... blood causes ketoacidosis. Some of them are smelly, so they can be detected by a practitioner without the use of chemical analysis. ...
... blood causes ketoacidosis. Some of them are smelly, so they can be detected by a practitioner without the use of chemical analysis. ...
Nucleic Acids and Protein Synthesis
... • In the first step of protein synthesis, the code of DNA is transcribed onto a molecule of RNA – this step of the process is known as Transcription • DNA is too big and too sensitive to leave the nucleus. However, proteins are made in the ribosomes, so the information in DNA must be transferred. • ...
... • In the first step of protein synthesis, the code of DNA is transcribed onto a molecule of RNA – this step of the process is known as Transcription • DNA is too big and too sensitive to leave the nucleus. However, proteins are made in the ribosomes, so the information in DNA must be transferred. • ...
Slide 1
... be synthesized in our body; The body lacks enzymes that can synthesize these amino acids from any precursor molecules. Non-Essential Amino acids : Need not be present in diet because the body can take care of their synthesis when required or when these are deficient in the diet. Semi-essential: ...
... be synthesized in our body; The body lacks enzymes that can synthesize these amino acids from any precursor molecules. Non-Essential Amino acids : Need not be present in diet because the body can take care of their synthesis when required or when these are deficient in the diet. Semi-essential: ...
4.2.1 Amino acids booklet 2013
... This term means the splitting of water by reaction with another compound. It means the other compound also gets split. If you split a protein you produce amino acids. The hydrolysis of a protein requires 6 moldm-3 HCl, refluxed for 24 hours. The products are amino acids. This process can also be car ...
... This term means the splitting of water by reaction with another compound. It means the other compound also gets split. If you split a protein you produce amino acids. The hydrolysis of a protein requires 6 moldm-3 HCl, refluxed for 24 hours. The products are amino acids. This process can also be car ...
Chapter 4 Cellular Respiration
... NADH and FADH2 from Krebs Cycle are pumped by electron energy across the inner membrane (cristae) and creates a concentration difference. ...
... NADH and FADH2 from Krebs Cycle are pumped by electron energy across the inner membrane (cristae) and creates a concentration difference. ...
(1) Identify the secondary structure described in each of the
... (16) Ingestion of methanol is a medical emergency. It is often treated by the administration of ethanol, which prevents the dangerous effects of methanol metabolism. In the body, methanol is oxidized to formaldehyde (methanal), a toxic molecule that cannot be further oxidized and that damages protei ...
... (16) Ingestion of methanol is a medical emergency. It is often treated by the administration of ethanol, which prevents the dangerous effects of methanol metabolism. In the body, methanol is oxidized to formaldehyde (methanal), a toxic molecule that cannot be further oxidized and that damages protei ...
Peptides and Proteins
... of peptide bond and the carbonyl oxygen of another peptide bond. According to Hbonding there are two main forms of secondary structure: α-helix: It is a spiral structure resulting from hydrogen bonding between one peptide bond and the fourth one β-sheets: is another form of secondary structure in wh ...
... of peptide bond and the carbonyl oxygen of another peptide bond. According to Hbonding there are two main forms of secondary structure: α-helix: It is a spiral structure resulting from hydrogen bonding between one peptide bond and the fourth one β-sheets: is another form of secondary structure in wh ...
Exam 1 454 Study Guide
... Describe general aspects of free energy changes for glycolysis. Explain the energetics under standard and "real" conditions Calculate energy yields for glucose and glycogen synthesis and breakdown (ATP and NADH). Detail the additional steps required to synthesize glucose from pyruvate Citric A ...
... Describe general aspects of free energy changes for glycolysis. Explain the energetics under standard and "real" conditions Calculate energy yields for glucose and glycogen synthesis and breakdown (ATP and NADH). Detail the additional steps required to synthesize glucose from pyruvate Citric A ...
Protein Synthesis
... DNA codes for proteins, many of which are enzymes. • Proteins (enzymes) can be used to make all the other molecules a cell needs: carbohydrates, lipids and nucleic acids. • A segment of DNA that carries the instructions to make (codes for) a protein is called a gene. ...
... DNA codes for proteins, many of which are enzymes. • Proteins (enzymes) can be used to make all the other molecules a cell needs: carbohydrates, lipids and nucleic acids. • A segment of DNA that carries the instructions to make (codes for) a protein is called a gene. ...
STUDY GUIDE
... I can: o Analyze the hierarchical organization of life from molecules to organisms. o Explain the structure and biological function of the following: a. Subatomic particles: b. Elements (CHNOPS): c. Organic molecules: i. Carbohydrates ii. Nucleic acids iii. Proteins (enzymes) d. Cellular organelles ...
... I can: o Analyze the hierarchical organization of life from molecules to organisms. o Explain the structure and biological function of the following: a. Subatomic particles: b. Elements (CHNOPS): c. Organic molecules: i. Carbohydrates ii. Nucleic acids iii. Proteins (enzymes) d. Cellular organelles ...
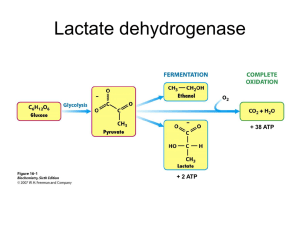
FERMENTATION: an anaerobic biological reaction process in which
... • In bacteria, the trp repressor protein inhibits the transcription of a suite of genes coding for enyzmes required for the synthesis of the amino acid tryptophan • In the absence of tryptophan, the recognition helices are not in the proper orientation to contact the promoter DNA; no repressor binds ...
... • In bacteria, the trp repressor protein inhibits the transcription of a suite of genes coding for enyzmes required for the synthesis of the amino acid tryptophan • In the absence of tryptophan, the recognition helices are not in the proper orientation to contact the promoter DNA; no repressor binds ...
Cell Respiration and Fermentation PPT
... The Cellular isms Metabolism: is the set of chemical reactions that occur in living organisms in order to maintain life. – These processes allow organisms to grow and reproduce, maintain their structures, and respond to their environments. – Usually divided into two categories. Catabolism and Anabol ...
... The Cellular isms Metabolism: is the set of chemical reactions that occur in living organisms in order to maintain life. – These processes allow organisms to grow and reproduce, maintain their structures, and respond to their environments. – Usually divided into two categories. Catabolism and Anabol ...
cell respiration wilk hl ibdp
... electrons to Oxygen (terminal e acceptor) and water is formed as an end product. ...
... electrons to Oxygen (terminal e acceptor) and water is formed as an end product. ...
Document
... Decreasing the amount of energy needed to produce an Activated complex or specifically reducing the activation Energy of the reaction. Catalyst make reaction proceed more Quickly. Enzymes are called biological catalysts and work the same Way as chemical catalysts within living organisms. Inhibitors ...
... Decreasing the amount of energy needed to produce an Activated complex or specifically reducing the activation Energy of the reaction. Catalyst make reaction proceed more Quickly. Enzymes are called biological catalysts and work the same Way as chemical catalysts within living organisms. Inhibitors ...
Key Terms:
... How are all the parts of aerobic respiration connected together? What is the key problem that must be solved for a cell to survive anaerobically? How is respiration commonly regulated? Why might a cell want to slow down respiration? Lecture Outline: Anaerobic Metabolism recall that in glycolys ...
... How are all the parts of aerobic respiration connected together? What is the key problem that must be solved for a cell to survive anaerobically? How is respiration commonly regulated? Why might a cell want to slow down respiration? Lecture Outline: Anaerobic Metabolism recall that in glycolys ...
Chapter 1 The Framework of Biology
... Proteins and lipids can feed into the glucose metabolism pathway to provide cellular energy. Other organic molecules, i.e., lipids and proteins, can also enter cellular respiration and provide energy. 6.4 Metabolic diversity reflects the evolution of energy pathways. Many metabolic pathways are simi ...
... Proteins and lipids can feed into the glucose metabolism pathway to provide cellular energy. Other organic molecules, i.e., lipids and proteins, can also enter cellular respiration and provide energy. 6.4 Metabolic diversity reflects the evolution of energy pathways. Many metabolic pathways are simi ...
Chapter 2 review key
... solute can be observed. A cell would be a good example of a suspension. If we add salt to a pot of boiling water, is a solution. ...
... solute can be observed. A cell would be a good example of a suspension. If we add salt to a pot of boiling water, is a solution. ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.