Classification of Amino Acids
... Sequencing Large Proteins Breaking disulfide bonds Oxidation by performic acid Reduction and carboxymethylation Cleaving the polypeptide chain Using proteases Cleavage of peptide bond next to particular amino acid residues ...
... Sequencing Large Proteins Breaking disulfide bonds Oxidation by performic acid Reduction and carboxymethylation Cleaving the polypeptide chain Using proteases Cleavage of peptide bond next to particular amino acid residues ...
Crash Course in Biochemistry
... • Some proteins bind (stick) to each other in a highly specific way – See hemoglobin • The final complex is functional • Individual pieces are not – Toxic truncated peptides ...
... • Some proteins bind (stick) to each other in a highly specific way – See hemoglobin • The final complex is functional • Individual pieces are not – Toxic truncated peptides ...
PROTEINS - ssag.sk
... – Hemoglobin, the iron containing protein in blood, transport oxygen from lungs to other parts of the body (C3032H4816O872N780S9Fe4) – Membrane transport proteins such as channels for potassium and water ...
... – Hemoglobin, the iron containing protein in blood, transport oxygen from lungs to other parts of the body (C3032H4816O872N780S9Fe4) – Membrane transport proteins such as channels for potassium and water ...
Unit 1 - Review Sheet 2010 IB
... 7. Identify the following functional groups: hydroxyl, carboxyl, carbonyl, amino, sulfhydryl and phosphate. 8. Identify the name, structure, chemical properties and biological significance of carbohydrates, fats, proteins and nucleic acids. 9. Define the term isomer and describe the relationship bet ...
... 7. Identify the following functional groups: hydroxyl, carboxyl, carbonyl, amino, sulfhydryl and phosphate. 8. Identify the name, structure, chemical properties and biological significance of carbohydrates, fats, proteins and nucleic acids. 9. Define the term isomer and describe the relationship bet ...
APcarbonandmacromols2015 16
... 1. Is this a condensation or hydrolysis reaction? 2. What are the reactants? What are the products? 3. Is this an anabolic or catabolic reaction? 4. Is energy required or released? ...
... 1. Is this a condensation or hydrolysis reaction? 2. What are the reactants? What are the products? 3. Is this an anabolic or catabolic reaction? 4. Is energy required or released? ...
Keystone Review Packet #2 Answers
... It turns out that hydrogen bonds are important for a few more reasons. Hydrogen bonds give water a high specific heat and also cause water to expand upon freezing. Specific heat is the amount of energy required to raise one gram of water 1 degree Celsius. 4. Can you think of a reason why water can a ...
... It turns out that hydrogen bonds are important for a few more reasons. Hydrogen bonds give water a high specific heat and also cause water to expand upon freezing. Specific heat is the amount of energy required to raise one gram of water 1 degree Celsius. 4. Can you think of a reason why water can a ...
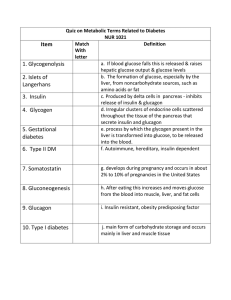
Quiz on Metabolic Terms Related to Diabetes NUR 1021 Item Match
... Quiz on Metabolic Terms Related to Diabetes NUR 1021 Match Definition Item With letter a. If blood glucose falls this is released & raises 1. Glycogenolysis hepatic glucose output & glucose levels b. The formation of glucose, especially by the 2. Islets of liver, from noncarbohydrate sources, such a ...
... Quiz on Metabolic Terms Related to Diabetes NUR 1021 Match Definition Item With letter a. If blood glucose falls this is released & raises 1. Glycogenolysis hepatic glucose output & glucose levels b. The formation of glucose, especially by the 2. Islets of liver, from noncarbohydrate sources, such a ...
Protein_hierarchy
... • “Non essential” amino acids are made by the body. • “Essential” amino acids need to be eaten in our diet. ...
... • “Non essential” amino acids are made by the body. • “Essential” amino acids need to be eaten in our diet. ...
U4L21 fuel oxidation - The University of Sydney
... – It is the ‘rate limiting’ step – The point which determines the overall rate at which people get to Uni ...
... – It is the ‘rate limiting’ step – The point which determines the overall rate at which people get to Uni ...
Chapter 2 Molecules to enzymes Multiple Choice
... D. Some of the enzyme has been consumed and the reaction has reached a plateau. . ...
... D. Some of the enzyme has been consumed and the reaction has reached a plateau. . ...
Biology
... 13. Now, explain what is 1) similar and 2) different between the two diagrams in #11 and #12 and explain how the processes of cellular respiration and photosynthesis use similar setups and similar metabolic pathways, but are fundamentally different in function and overall outcome. You can imagine th ...
... 13. Now, explain what is 1) similar and 2) different between the two diagrams in #11 and #12 and explain how the processes of cellular respiration and photosynthesis use similar setups and similar metabolic pathways, but are fundamentally different in function and overall outcome. You can imagine th ...
Week 1 Pre-Lecture Slides
... Imagine two massive proteins composed of 10,000 atoms. At the end of one protein is a hydroxyl group, while the other protein has a phosphate group in the same place. How are these massive molecules similar or different? Does it matter? ...
... Imagine two massive proteins composed of 10,000 atoms. At the end of one protein is a hydroxyl group, while the other protein has a phosphate group in the same place. How are these massive molecules similar or different? Does it matter? ...
Bio-Chemistry Course Descriptions
... also be placed on methods of purification and analysis of biological macromolecules and applications of basic biochemical principles to living systems. In addition to lectures and discussions, problem sets will be assigned to equip students with an understanding of basic biochemical principles and ...
... also be placed on methods of purification and analysis of biological macromolecules and applications of basic biochemical principles to living systems. In addition to lectures and discussions, problem sets will be assigned to equip students with an understanding of basic biochemical principles and ...
Cell Benchmark Study Guide 2013
... 8) Fill in the blanks with these words: Mitochondrion / O2 (used twice) / Chloroplast / CO2 / ATP ...
... 8) Fill in the blanks with these words: Mitochondrion / O2 (used twice) / Chloroplast / CO2 / ATP ...
Lecture 19
... that form the backbone of a peptide chain or protein The secondary structures of proteins indicate the three-dimensional spatial arrangements of the polypeptide chains The tertiary structure of a protein gives a specific three-dimensional shape to the polypeptide chain including interactions and cro ...
... that form the backbone of a peptide chain or protein The secondary structures of proteins indicate the three-dimensional spatial arrangements of the polypeptide chains The tertiary structure of a protein gives a specific three-dimensional shape to the polypeptide chain including interactions and cro ...
Chapter 19 Aminoacids and Proteins
... that form the backbone of a peptide chain or protein The secondary structures of proteins indicate the three-dimensional spatial arrangements of the polypeptide chains The tertiary structure of a protein gives a specific three-dimensional shape to the polypeptide chain including interactions and cro ...
... that form the backbone of a peptide chain or protein The secondary structures of proteins indicate the three-dimensional spatial arrangements of the polypeptide chains The tertiary structure of a protein gives a specific three-dimensional shape to the polypeptide chain including interactions and cro ...
Protein Synthesis Notes
... (the “chefs”) to send these recipes to the ribosomes (“kitchen”) so they can be made. 1. The basic unit of a protein is an amino acid a. we use 20 amino acids to make all of our proteins 2. A chain of amino acids together is a protein 3. Types of proteins include: hormones, enzymes, structural prote ...
... (the “chefs”) to send these recipes to the ribosomes (“kitchen”) so they can be made. 1. The basic unit of a protein is an amino acid a. we use 20 amino acids to make all of our proteins 2. A chain of amino acids together is a protein 3. Types of proteins include: hormones, enzymes, structural prote ...
First Exam Study Guide
... between an acid and a base? Can you recognize an example of each? What is a buffer? How does the carbonic acid / bicarbonate buffer respond to added acid or base? 6. Can you recognize straight chains, branched chains and rings of carbon atoms? 7. What are the structures and names of the various func ...
... between an acid and a base? Can you recognize an example of each? What is a buffer? How does the carbonic acid / bicarbonate buffer respond to added acid or base? 6. Can you recognize straight chains, branched chains and rings of carbon atoms? 7. What are the structures and names of the various func ...
SURVEY OF BIOCHEMISTRY - Georgia Institute of Technology
... Glycogen Phosphorylase cAMP activates PKA Glucagon stimulates this process ...
... Glycogen Phosphorylase cAMP activates PKA Glucagon stimulates this process ...
Chemistry of Living Things
... smaller compounds in a process known as polymerization. Organic compounds 4 basic types 1. Carbohydrates: compounds composed of Carbon, Hydrogen and oxygen in a 1:2:1 ratio Ex. Glucose C6H12O6 Monosaccharide: Simple sugar containing one glucose molecule ...
... smaller compounds in a process known as polymerization. Organic compounds 4 basic types 1. Carbohydrates: compounds composed of Carbon, Hydrogen and oxygen in a 1:2:1 ratio Ex. Glucose C6H12O6 Monosaccharide: Simple sugar containing one glucose molecule ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.