primary structure
... Proteins (aka polypeptides) 3.11-3.14 A. Polymer of amino acid monomers B. 1000s exist each with unique 3D structure that corresponds to its function C. Role in everything a cell /organism does ...
... Proteins (aka polypeptides) 3.11-3.14 A. Polymer of amino acid monomers B. 1000s exist each with unique 3D structure that corresponds to its function C. Role in everything a cell /organism does ...
Chapter 9 / Energy-Releasing Pathways and Biosynthesis I
... Differ in their final electron acceptor Cellular respiration produces more ATP Pyruvate is a key juncture in catabolism Glycolysis occurs in nearly all organisms ...
... Differ in their final electron acceptor Cellular respiration produces more ATP Pyruvate is a key juncture in catabolism Glycolysis occurs in nearly all organisms ...
Final Exam Review Part 1
... Which of the following is defined as having the same pH with small additions of acids or bases? a. acids polymers bases buffers ...
... Which of the following is defined as having the same pH with small additions of acids or bases? a. acids polymers bases buffers ...
1. (a) - The West Bridgford School
... Saturated – all valencies of C filled / saturated with hydrogen / all (C–C) single bonds / no double bonds; fatty acid 1 is saturated/fatty acids 2 and 3 are unsaturated; ...
... Saturated – all valencies of C filled / saturated with hydrogen / all (C–C) single bonds / no double bonds; fatty acid 1 is saturated/fatty acids 2 and 3 are unsaturated; ...
Answer Sheet
... 15. What are two types of fats and where do they come from? Saturated and unsaturated. Most fats from animals are saturated (meat, cheese, cookies, cakes). Most fats from plants are unsat. (fish, avocados, liquid cooking oils) 16. What is the danger of eating too much fat? High cholesterol, which a ...
... 15. What are two types of fats and where do they come from? Saturated and unsaturated. Most fats from animals are saturated (meat, cheese, cookies, cakes). Most fats from plants are unsat. (fish, avocados, liquid cooking oils) 16. What is the danger of eating too much fat? High cholesterol, which a ...
LS1a Fall 09
... All 19 naturally occurring chiral amino acids (excluding glycine) have the same chirality as shown in the generic amino acid in section activity #1. If the amino-group is to the left and the carboxylgroup is on the right, the side chain will point out of the page (towards you) if the -carbon is poi ...
... All 19 naturally occurring chiral amino acids (excluding glycine) have the same chirality as shown in the generic amino acid in section activity #1. If the amino-group is to the left and the carboxylgroup is on the right, the side chain will point out of the page (towards you) if the -carbon is poi ...
Model Description Sheet
... To someone who has excessive P2X4 receptors, simple gestures like hugs could cause unbearable pain. P2X4, a protein receptor located on the membrane of neurons, plays a large role in neuronal communication and pain perception. Ion channels on dendrites, located on one end of a neuron, allow ions to ...
... To someone who has excessive P2X4 receptors, simple gestures like hugs could cause unbearable pain. P2X4, a protein receptor located on the membrane of neurons, plays a large role in neuronal communication and pain perception. Ion channels on dendrites, located on one end of a neuron, allow ions to ...
Slide () - Anesthesiology - American Society of Anesthesiologists
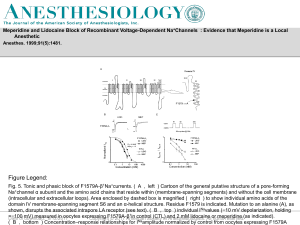
... Fig. 5. Tonic and phasic block of F1579A-β1Na+currents. ( A , left ) Cartoon of the general putative structure of a pore-forming Na+channel α subunit and the amino acid chains that reside within (membrane-spanning segments) and without the cell membrane (intracellular and extracellular loops). Area ...
... Fig. 5. Tonic and phasic block of F1579A-β1Na+currents. ( A , left ) Cartoon of the general putative structure of a pore-forming Na+channel α subunit and the amino acid chains that reside within (membrane-spanning segments) and without the cell membrane (intracellular and extracellular loops). Area ...
Understanding how muscles get energy
... amino acids, joined together. When you eat protein, it is broken down into amino acids during digestion. ...
... amino acids, joined together. When you eat protein, it is broken down into amino acids during digestion. ...
Biology 11, Fall 2002
... MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. The proper order of objects, from simple to complex, is a. atom, molecule, cell, tissue, organ, organism, population, community. b. cell, molecule, atom, tissue, organ, organism, population, com ...
... MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. The proper order of objects, from simple to complex, is a. atom, molecule, cell, tissue, organ, organism, population, community. b. cell, molecule, atom, tissue, organ, organism, population, com ...
Chemical reactions of amino acids:
... like ninhydrin test or fluorescamine, or for detection specific amino acids like: Millon test → for Tyr Salkguchi test → for Arg Nitroprusside test → for Cys Hopkin test → for Trp There are reactions for free α amino group like: {Sanger reaction and Edman reaction}, these used to detect the first ...
... like ninhydrin test or fluorescamine, or for detection specific amino acids like: Millon test → for Tyr Salkguchi test → for Arg Nitroprusside test → for Cys Hopkin test → for Trp There are reactions for free α amino group like: {Sanger reaction and Edman reaction}, these used to detect the first ...
Cell Respiration Notes (Honors)
... modified before the citric acid cycle begins. These new molecules are broken down to form ATP and CO2. One ATP per cycle is produced, two cycles occur per glucose molecule – therefore 2 ATP’s are produced by Krebs Cycle. *Also generates high energy electrons carried by NADH and FADH2. ...
... modified before the citric acid cycle begins. These new molecules are broken down to form ATP and CO2. One ATP per cycle is produced, two cycles occur per glucose molecule – therefore 2 ATP’s are produced by Krebs Cycle. *Also generates high energy electrons carried by NADH and FADH2. ...
Organic Chemistry Fifth Edition
... Zwitterionic Compounds/Zwitterions neutral compounds having formal unit electrical charges of opposite sign. Some chemists restrict the term to compounds with the charges on nonadjacent atoms. Sometimes referred to as inner salts, dipolar ions (a misnomer), e.g. +H3N-CH2CO2ammonioacetate (glycine). ...
... Zwitterionic Compounds/Zwitterions neutral compounds having formal unit electrical charges of opposite sign. Some chemists restrict the term to compounds with the charges on nonadjacent atoms. Sometimes referred to as inner salts, dipolar ions (a misnomer), e.g. +H3N-CH2CO2ammonioacetate (glycine). ...
Energy Review - MrsAllisonMagee
... and physically separate where the Calvin Cycle takes place • Corn, sugarcane ...
... and physically separate where the Calvin Cycle takes place • Corn, sugarcane ...
Bacterial Physiology Lec-7 Energy Release and Conservation
... such as pyruvate that act as the electron acceptor. Fermentation occurs under anaerobic conditions , but sometimes occur when oxygen is present. The amount of energy that resulted from respiration is high while limited energy released during fermentation, so fermentation can be defined: energy-yield ...
... such as pyruvate that act as the electron acceptor. Fermentation occurs under anaerobic conditions , but sometimes occur when oxygen is present. The amount of energy that resulted from respiration is high while limited energy released during fermentation, so fermentation can be defined: energy-yield ...
2.3 Carbon-Based Molecules
... heme, which binds & transports oxygen in the body – Skin, hair & nails contain the protein keratin which provides structure, strength & water-proofing – Enzymes are protein catalysts for chemical reactions in all living things ...
... heme, which binds & transports oxygen in the body – Skin, hair & nails contain the protein keratin which provides structure, strength & water-proofing – Enzymes are protein catalysts for chemical reactions in all living things ...
1 Chapter 3: Chemistry of Water Polar covalent bonds within water
... Hydrocarbons consist of only carbon and hydrogen The nonpolar C – H bonds in hydrocarbon chains result in their hydrophobic properties -Many cells have hydrocarbon regions, like the hydrophobic fatty acid tails Hydrocarbons can undergo reactions that release a large amount of energy. -In fats, hydro ...
... Hydrocarbons consist of only carbon and hydrogen The nonpolar C – H bonds in hydrocarbon chains result in their hydrophobic properties -Many cells have hydrocarbon regions, like the hydrophobic fatty acid tails Hydrocarbons can undergo reactions that release a large amount of energy. -In fats, hydro ...
IV. -Amino Acids: carboxyl and amino groups bonded to
... 2. Polypeptide contains many amino acids and if there are very many amino acids one can call it protein C. Proteins have molecular weights > several thousand and have 3-4 levels of structure 1. Primary Structure (1°) sequence of amino acids connected by peptide bo n d s 2. Secondary Structure (2°) l ...
... 2. Polypeptide contains many amino acids and if there are very many amino acids one can call it protein C. Proteins have molecular weights > several thousand and have 3-4 levels of structure 1. Primary Structure (1°) sequence of amino acids connected by peptide bo n d s 2. Secondary Structure (2°) l ...
Microbial Origins of Life and Energy Conversions
... • NO3- (nitrate) can be used as terminal electron acceptor • under anaerobic conditions • Results in conversion of NO3- to atmospheric nitrogen N2 • Three reactions involved in process ...
... • NO3- (nitrate) can be used as terminal electron acceptor • under anaerobic conditions • Results in conversion of NO3- to atmospheric nitrogen N2 • Three reactions involved in process ...
COURSE SYLLABUS CHM 521 Biochemistry I 3(3
... Biochemistry by Stryer (4th edition), Freeman and Co., 1995 Principles of Biochemistry by Horton, et al, Neil Patterson Publishers, 1993 Biochemistry by Rawn, Neil Patterson Publishers, 1989 Biochemistry by Voet and Voet, (2nd edition), John Wiley & Sons, 1995 Biochemistry by Zubay (3rd edition), Wm ...
... Biochemistry by Stryer (4th edition), Freeman and Co., 1995 Principles of Biochemistry by Horton, et al, Neil Patterson Publishers, 1993 Biochemistry by Rawn, Neil Patterson Publishers, 1989 Biochemistry by Voet and Voet, (2nd edition), John Wiley & Sons, 1995 Biochemistry by Zubay (3rd edition), Wm ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.