PowerPoint 演示文稿
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
Aldehydes and Ketones The Carbonyl Group
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
... Other Nomenclature Rules • In cyclic ketones, the carbonyl group is always numbered “1”; this does not need to be included in the name. The numbering continues clockwise or counterclockwise to give the lowest number for the next substituent. • Molecules with more than one ketone group are named by p ...
dr.ebtehal Lec3
... catabolic metabolism of alcohol. It is commonly thought that the flush reaction is caused by an inability to metabolize alcohol. ...
... catabolic metabolism of alcohol. It is commonly thought that the flush reaction is caused by an inability to metabolize alcohol. ...
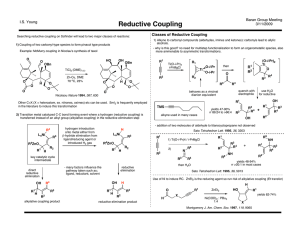
Copper-Catalyzed Coupling Reactions Using Carbon
... Part I - Copper-Catalyzed Oxidative Alkylation of Cyclic Benzyl Ethers Using Oxygen Gas as the Terminal Oxidant ...
... Part I - Copper-Catalyzed Oxidative Alkylation of Cyclic Benzyl Ethers Using Oxygen Gas as the Terminal Oxidant ...
Advanced Practical Organic Chemistry
... The symbols Ca, K, Mn, Na and Cl stand for calcium, potassium, manganese, sodium and chlorine, respectively. Molecules with Carbon Most atoms are only capable of forming small molecules. However, one or two can form larger molecules. By far and away the best atom for making large molecules with, is ...
... The symbols Ca, K, Mn, Na and Cl stand for calcium, potassium, manganese, sodium and chlorine, respectively. Molecules with Carbon Most atoms are only capable of forming small molecules. However, one or two can form larger molecules. By far and away the best atom for making large molecules with, is ...
Alcohols, Phenols, and Thiols
... Alcohols and phenols are also weak bases. They can be protonated on the oxygen by strong acids. This reaction is the first step in the acid-catalyzed dehydration of alcohols to alkenes and in the conversion of alcohols to alkyl halides by reaction with hydrogen halides. Alkyl halides can also be pre ...
... Alcohols and phenols are also weak bases. They can be protonated on the oxygen by strong acids. This reaction is the first step in the acid-catalyzed dehydration of alcohols to alkenes and in the conversion of alcohols to alkyl halides by reaction with hydrogen halides. Alkyl halides can also be pre ...
Lithium Bromide Original Commentary
... acid in a variety of reactions. For example, this reagent was used in the Pictet-Spengler cyclization of a highly functionalized imine (eq 13).33 In this reaction, carbon-carbon bond formation occurs without reaction or loss of stereochemical integrity of the α-amino nitrile functionality. Lithium b ...
... acid in a variety of reactions. For example, this reagent was used in the Pictet-Spengler cyclization of a highly functionalized imine (eq 13).33 In this reaction, carbon-carbon bond formation occurs without reaction or loss of stereochemical integrity of the α-amino nitrile functionality. Lithium b ...
Efficient hydrogenation of organic carbonates, carbamates and
... groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more ...
... groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more ...
Compounds with Oxygen Atoms
... When thiols undergo oxidation, • an H atom is lost from each of two –SH groups. • the product is a disulfide. [O] CH3─SH + HS─CH3 CH3─S─S─CH3 + H2O ...
... When thiols undergo oxidation, • an H atom is lost from each of two –SH groups. • the product is a disulfide. [O] CH3─SH + HS─CH3 CH3─S─S─CH3 + H2O ...
Chapter 14-15 - Bakersfield College
... 3. Low boiling points: hydrocarbons < ethers < alcohols. ...
... 3. Low boiling points: hydrocarbons < ethers < alcohols. ...
Iodine and Lipase Based Green Oxidation Technology
... Oxidation of primary and secondary alcohols to their corresponding carbonyl compounds is widely used in the fine chemicals industry since aldehydes and ketones are the key raw materials for a wide range of pharmaceutical intermediates, fragrances and flavors. 1, ...
... Oxidation of primary and secondary alcohols to their corresponding carbonyl compounds is widely used in the fine chemicals industry since aldehydes and ketones are the key raw materials for a wide range of pharmaceutical intermediates, fragrances and flavors. 1, ...
(omit), and Epoxides
... • Although cyclic ethers have IUPAC names, their common names are more widely used. – IUPAC: prefix ox- shows oxygen in the ring. – The suffixes -irane, irane -etane, etane -olane, olane and -ane show three, four, five, and six atoms in a saturated ring. ...
... • Although cyclic ethers have IUPAC names, their common names are more widely used. – IUPAC: prefix ox- shows oxygen in the ring. – The suffixes -irane, irane -etane, etane -olane, olane and -ane show three, four, five, and six atoms in a saturated ring. ...
Alkyl Halides02
... E1 eliminations, like SN1 substitutions, begin with unimolecular dissociation, but the dissociation is followed by loss of a proton from the -carbon (attached to the C+) rather than by substitution. E1 & SN1 normally occur in competition, whenever an alkyl halide is treated in a protic solvent with ...
... E1 eliminations, like SN1 substitutions, begin with unimolecular dissociation, but the dissociation is followed by loss of a proton from the -carbon (attached to the C+) rather than by substitution. E1 & SN1 normally occur in competition, whenever an alkyl halide is treated in a protic solvent with ...
Topic 5: Organic Chemistry - Manitoba Education and Training
... typical reaction of concentrated sulphuric acid with sugar to produce solid carbon and a number of other products that are toxic. This demonstration should be done in a fume hood, with all the necessary safety precautions noted in Appendix 3.8. The Material Safety Data Sheet for concentrated sulphur ...
... typical reaction of concentrated sulphuric acid with sugar to produce solid carbon and a number of other products that are toxic. This demonstration should be done in a fume hood, with all the necessary safety precautions noted in Appendix 3.8. The Material Safety Data Sheet for concentrated sulphur ...
Elimination Reactions
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
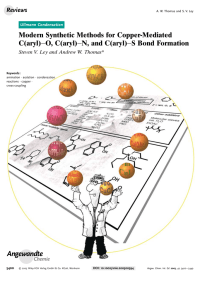
Modern Synthetic Methods for Copper-Mediated C(aryl
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
- University of Bath Opus
... carbonyl compounds are prone to decomposition due to the increased reactivity and are often difficult to form selectively, i.e. without over oxidation. An ideal system would allow the use of stable, non-toxic alcohols while maintaining the reactivity of the carbonyl compound. One way to achieve this ...
... carbonyl compounds are prone to decomposition due to the increased reactivity and are often difficult to form selectively, i.e. without over oxidation. An ideal system would allow the use of stable, non-toxic alcohols while maintaining the reactivity of the carbonyl compound. One way to achieve this ...
CH 2 - ResearchGate
... It has been developed by H.E. Simmons and R.D. Smith of the DuPont Company. During this synthesis diiodomethane and zinc-copper couple are stirred together with alkene. The diiodomethane and zinc react together to produce carbene like species called as carbenoid which brings stereospecific addition ...
... It has been developed by H.E. Simmons and R.D. Smith of the DuPont Company. During this synthesis diiodomethane and zinc-copper couple are stirred together with alkene. The diiodomethane and zinc react together to produce carbene like species called as carbenoid which brings stereospecific addition ...
Alcohols, Phenols, and Ethers
... – in alcohols, a hydroxyl group is connected to a carbon atom. – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional ...
... – in alcohols, a hydroxyl group is connected to a carbon atom. – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional ...
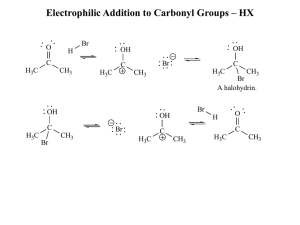
Electrophilic Addition to Carbonyl Groups – HX
... Acetals can be used to protect carbonyl groups (aldehydes, ketones, etc.) They are stable in the presence of strong bases and nucleophiles and the carbonyl can be reformed by addition of aqueous acid. ...
... Acetals can be used to protect carbonyl groups (aldehydes, ketones, etc.) They are stable in the presence of strong bases and nucleophiles and the carbonyl can be reformed by addition of aqueous acid. ...
Iodomethylzinc_iodid.. - Groupe Charette
... Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and co-workers;9 and type 3, an alkyl exchange reaction between an alkyl zinc and a 1,1dihaloalkane, often referred to as the Furukawa procedure.10 Type 1 reagent generation has been used most of ...
... Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and co-workers;9 and type 3, an alkyl exchange reaction between an alkyl zinc and a 1,1dihaloalkane, often referred to as the Furukawa procedure.10 Type 1 reagent generation has been used most of ...
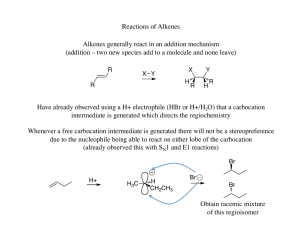
Chapter 11
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
Alcohols - Angelo State University
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. ...
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. ...
Elias James Corey

Elias James ""E.J."" Corey (born July 12, 1928) is an American organic chemist. In 1990, he won the Nobel Prize in Chemistry ""for his development of the theory and methodology of organic synthesis"", specifically retrosynthetic analysis. Regarded by many as one of the greatest living chemists, he has developed numerous synthetic reagents, methodologies and total syntheses and has advanced the science of organic synthesis considerably.