1H-Imidazol-4(5H)-ones and thiazol-4(5H)
... substituted at the α-position of the carbonyl and therefore, after reaction with an electrophile, a tetrasubstituted stereocenter is created and, iv) the resulting adducts can be easily hydrolyzed to provide carboxylic acids or their derivatives carrying different functionalities. The most common pr ...
... substituted at the α-position of the carbonyl and therefore, after reaction with an electrophile, a tetrasubstituted stereocenter is created and, iv) the resulting adducts can be easily hydrolyzed to provide carboxylic acids or their derivatives carrying different functionalities. The most common pr ...
aa-2005-38-71-negishi - University of Windsor
... the multimechanistic and multifaceted nature of the Pd-catalyzed cross-coupling, however, it is not practical to compare and rank them on one scale. From a practical viewpoint, synthetic chemists are generally seeking those methods and reactions that satisfy some or most, if not all, of the followin ...
... the multimechanistic and multifaceted nature of the Pd-catalyzed cross-coupling, however, it is not practical to compare and rank them on one scale. From a practical viewpoint, synthetic chemists are generally seeking those methods and reactions that satisfy some or most, if not all, of the followin ...
VITA - Trace: Tennessee Research and Creative Exchange
... case by Hillhouse it was found that although the starting Ni-complex would not activate an organic azide, a Ni-imido could be prepared synthetically (Figure 1.1B) and the nitrene does transfer to an alkene to form an aziridine. 10c Still the lack of a system which can combine both the activation ste ...
... case by Hillhouse it was found that although the starting Ni-complex would not activate an organic azide, a Ni-imido could be prepared synthetically (Figure 1.1B) and the nitrene does transfer to an alkene to form an aziridine. 10c Still the lack of a system which can combine both the activation ste ...
Full-Text PDF
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
C 1 hapter
... The hydroboration reaction is a classical method for synthesising organoboron compounds and one of the most studied reactions in organic synthesis. Introduced by H. C. Brown [1] in 1959, it is based on the syn addition of borane reagent H-BR’2 to alkenes or alkynes. In fact, it is considered to be t ...
... The hydroboration reaction is a classical method for synthesising organoboron compounds and one of the most studied reactions in organic synthesis. Introduced by H. C. Brown [1] in 1959, it is based on the syn addition of borane reagent H-BR’2 to alkenes or alkynes. In fact, it is considered to be t ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... Since the C=C bond forms between the C's of the two C-X groups in the reactant, these reactions precisely place the C=C in the product. However, the dihaloalkane reactant is usually synthesized (Chapter 10) from the same alkene that it forms by elimination of X-X, so dehalogenation reactions are les ...
... Since the C=C bond forms between the C's of the two C-X groups in the reactant, these reactions precisely place the C=C in the product. However, the dihaloalkane reactant is usually synthesized (Chapter 10) from the same alkene that it forms by elimination of X-X, so dehalogenation reactions are les ...
+ H 2 O(l) - Knockhardy
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
No Slide Title
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
chapter-15
... The Carbonyl Group In this and several following chapters, we study the physical and chemical properties of classes of compounds containing the carbonyl group, C=O • aldehydes and ketones (Chapter 13) ...
... The Carbonyl Group In this and several following chapters, we study the physical and chemical properties of classes of compounds containing the carbonyl group, C=O • aldehydes and ketones (Chapter 13) ...
PDF - Nanyang Technological University
... presence of 30 mol % of AcOH, gave product 4 a with low ee values (35 %/37 %; Table 1, entry 5). The ee values were improved by using catalyst C (48 %/51 % ee; Table 1, entry 6). For reactions that were carried out in DMF (Table 1, entry 6), the low ee values were largely caused by the racemic backg ...
... presence of 30 mol % of AcOH, gave product 4 a with low ee values (35 %/37 %; Table 1, entry 5). The ee values were improved by using catalyst C (48 %/51 % ee; Table 1, entry 6). For reactions that were carried out in DMF (Table 1, entry 6), the low ee values were largely caused by the racemic backg ...
aldehydes
... Acid Chlorides + Lithium Dialkyl Copper (Gilman Reagent): produces ketones. The reaction is unique to these two reagents and the mechanism is uncertain. As with DIBAH for aldehyde reductions, a low temperature (78 C) solvent (ether) is used to prevent further alkyl addition to the ketone to form an ...
... Acid Chlorides + Lithium Dialkyl Copper (Gilman Reagent): produces ketones. The reaction is unique to these two reagents and the mechanism is uncertain. As with DIBAH for aldehyde reductions, a low temperature (78 C) solvent (ether) is used to prevent further alkyl addition to the ketone to form an ...
Organic Chemistry - UCR Chemistry
... chemical word nucleus and the Greek word philos which means "loving". A nucleophile wants ("loves") to use one of its unshared electron pairs to bond to a positively polarized nucleus. Nucleophiles always have an unshared electron pair that forms the new chemical bond, but they are not always negati ...
... chemical word nucleus and the Greek word philos which means "loving". A nucleophile wants ("loves") to use one of its unshared electron pairs to bond to a positively polarized nucleus. Nucleophiles always have an unshared electron pair that forms the new chemical bond, but they are not always negati ...
Organic Chemistry
... • Study of carbon compounds – Composed of carbon, hydrogen, oxygen, nitrogen, sulfur, phosphorus, and the halogens • More than 95% of known compounds are organic compounds (Carbon-containing) • Recall – Carbons can have 4 bonds – Nitrogen can have 3 bonds – Oxygen can have 2 bonds – Hydrogen can hav ...
... • Study of carbon compounds – Composed of carbon, hydrogen, oxygen, nitrogen, sulfur, phosphorus, and the halogens • More than 95% of known compounds are organic compounds (Carbon-containing) • Recall – Carbons can have 4 bonds – Nitrogen can have 3 bonds – Oxygen can have 2 bonds – Hydrogen can hav ...
Oxidation
... Some oxidation reactions of alkenes give cyclic ethers in which both carbons of a double bond become bonded to the same oxygen atom. These products are called epoxides or oxiranes. An important method for preparing epoxides is by reaction with peracids, RCO3H. The oxygen-oxygen bond of such peroxide ...
... Some oxidation reactions of alkenes give cyclic ethers in which both carbons of a double bond become bonded to the same oxygen atom. These products are called epoxides or oxiranes. An important method for preparing epoxides is by reaction with peracids, RCO3H. The oxygen-oxygen bond of such peroxide ...
12_chemistry_impq_CH10_haloalkanes_and_haloarenes_02
... Ans. Nitration is an electrophilic substitution. The –OH group in phenol increases the electron density at ortho and para position as follows Since phenol has higher electron density due to electron releasing nature of -OH group , compared to benzene , therefore nitration is easy in phenol than benz ...
... Ans. Nitration is an electrophilic substitution. The –OH group in phenol increases the electron density at ortho and para position as follows Since phenol has higher electron density due to electron releasing nature of -OH group , compared to benzene , therefore nitration is easy in phenol than benz ...
15alcpp - Knockhardy
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
Scope and Limitations - Organic Reactions Wiki
... Osmium tetroxide has since established itself as the reagent of choice for the syndihydroxylation of olefins, primarily because of its inertness toward other functional groups and lack of over-oxidation products.4 Researchers from the UpJohn company reported a convenient and reliable procedure for ...
... Osmium tetroxide has since established itself as the reagent of choice for the syndihydroxylation of olefins, primarily because of its inertness toward other functional groups and lack of over-oxidation products.4 Researchers from the UpJohn company reported a convenient and reliable procedure for ...
Rhenium Nanochemistry for Catalyst Preparation
... The methoxide derivatives of this family are poorly soluble but their thermal decomposition under thoroughly controlled conditions offered an attractive approach to organized porous solids with the prospect of incorporating rhenium oxide into a simultaneously formed hierarchical matrix [51]. Unfortu ...
... The methoxide derivatives of this family are poorly soluble but their thermal decomposition under thoroughly controlled conditions offered an attractive approach to organized porous solids with the prospect of incorporating rhenium oxide into a simultaneously formed hierarchical matrix [51]. Unfortu ...
Organic Chemistry
... Multiple bonds affect, physical and chemical properties (reactivity of organic molecules). ...
... Multiple bonds affect, physical and chemical properties (reactivity of organic molecules). ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... abundant, and readily available catalysts for chemical synthesis (Figure 2).23 Importantly, these carbocatalysts merge the benefits of green synthesis with heterogeneous reaction conditions, which greatly simplifies workup conditions and is particularly attractive from an industrial standpoint.24 The ...
... abundant, and readily available catalysts for chemical synthesis (Figure 2).23 Importantly, these carbocatalysts merge the benefits of green synthesis with heterogeneous reaction conditions, which greatly simplifies workup conditions and is particularly attractive from an industrial standpoint.24 The ...
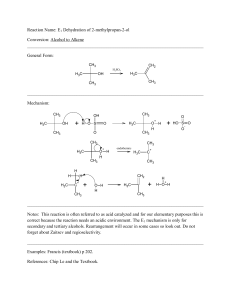
1990-Spring-Exam-2-student
... 633 (1990), two steps you would be able to describe. Fill in the necessary reagents, putting them in order if required. Ask yourself: What has changed in the reaction? What remains the same? ...
... 633 (1990), two steps you would be able to describe. Fill in the necessary reagents, putting them in order if required. Ask yourself: What has changed in the reaction? What remains the same? ...
Elias James Corey

Elias James ""E.J."" Corey (born July 12, 1928) is an American organic chemist. In 1990, he won the Nobel Prize in Chemistry ""for his development of the theory and methodology of organic synthesis"", specifically retrosynthetic analysis. Regarded by many as one of the greatest living chemists, he has developed numerous synthetic reagents, methodologies and total syntheses and has advanced the science of organic synthesis considerably.