Full-Text PDF
... science-oriented research. In particular, commercial pharmaceuticals and agrochemicals frequently contain single fluorine atoms or trifluoromethyl groups [1–4]. Nevertheless, growing interest in emergent fluorinated substituents, like α-fluorinated ethers, has been observed within recent years [5–8] ...
... science-oriented research. In particular, commercial pharmaceuticals and agrochemicals frequently contain single fluorine atoms or trifluoromethyl groups [1–4]. Nevertheless, growing interest in emergent fluorinated substituents, like α-fluorinated ethers, has been observed within recent years [5–8] ...
The Grob Fragmentation
... 2) Two stepped process, firstly loss of X generating a carbocation then break down to the two olefinic species if following the fragmentation route (similar to E1 or SN1) However the carbonium ion can further react via elimination, substitution, or ring-closure. The rate-determining step is the ioni ...
... 2) Two stepped process, firstly loss of X generating a carbocation then break down to the two olefinic species if following the fragmentation route (similar to E1 or SN1) However the carbonium ion can further react via elimination, substitution, or ring-closure. The rate-determining step is the ioni ...
Transition Metal Catalyzed Selective Oxidation of Sugars and Polyols
... under my laboratory assistance. I learned a lot about assisting and guiding others in research and wish them the best for their future. I also would like to thank Valerie Robertson of the NMR centre of the University of Guelph for her help and technical support with NMR experiments and Prof. John Go ...
... under my laboratory assistance. I learned a lot about assisting and guiding others in research and wish them the best for their future. I also would like to thank Valerie Robertson of the NMR centre of the University of Guelph for her help and technical support with NMR experiments and Prof. John Go ...
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic
... case). Similarly, (CH3 )2 CH+ is a 2 (secondary) carbocation because it has 2 alkyl groups on the C+ center, while CH3 CH2 + with 1 alkyl group on C+ is a 1 (primary) carbocation. Using this general terminology, we can summarize this carbocation stability order as 3° > 2° > 1° > methyl. Other simple ...
... case). Similarly, (CH3 )2 CH+ is a 2 (secondary) carbocation because it has 2 alkyl groups on the C+ center, while CH3 CH2 + with 1 alkyl group on C+ is a 1 (primary) carbocation. Using this general terminology, we can summarize this carbocation stability order as 3° > 2° > 1° > methyl. Other simple ...
中国科学技术大学有机合成讲义-2
... c) Kakis, F. J.; Fétizon, M.; Douchkine, N.; Golfier, M.; Mourgues, P.; Prange, T. Jhttp://doc.xuehai.net/bc956988fd90b0ed78020c467.html. Org. Chem. 1974, 39, 523.] ...
... c) Kakis, F. J.; Fétizon, M.; Douchkine, N.; Golfier, M.; Mourgues, P.; Prange, T. Jhttp://doc.xuehai.net/bc956988fd90b0ed78020c467.html. Org. Chem. 1974, 39, 523.] ...
Amidations of Rosin with Isocyanates
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
Postprint
... Phosphonates and phosphine oxides are highly coordinating groups, and their vinyls should make for good substrates for Ru and Rh catalysis. However these substrates have received rather little attention: while there are reports of Chiraphos type ligands being synthesised by asymmetric hydrogenation, ...
... Phosphonates and phosphine oxides are highly coordinating groups, and their vinyls should make for good substrates for Ru and Rh catalysis. However these substrates have received rather little attention: while there are reports of Chiraphos type ligands being synthesised by asymmetric hydrogenation, ...
Organic Chemistry
... • the carbon skeleton: how can we put it together. Our only method to date for forming new a C-C bond is the alkylation of alkyne anions (Section 7.5) • the functional groups: what are they, how can they be used in forming the carbon-skeleton of the target molecule, and how can they be changed to gi ...
... • the carbon skeleton: how can we put it together. Our only method to date for forming new a C-C bond is the alkylation of alkyne anions (Section 7.5) • the functional groups: what are they, how can they be used in forming the carbon-skeleton of the target molecule, and how can they be changed to gi ...
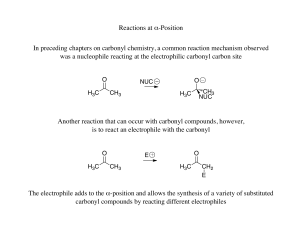
Reactions at α-Position In preceding chapters on carbonyl chemistry
... The dehydration is favored compared to other alcohols dehydrating to alkenes due to the conjugation of the obtained α,β-unsaturated alkene with the carbonyl ...
... The dehydration is favored compared to other alcohols dehydrating to alkenes due to the conjugation of the obtained α,β-unsaturated alkene with the carbonyl ...
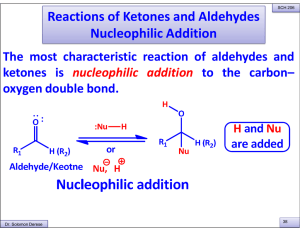
Reactions of Ketones and Aldehydes Nucleophilic Addition
... If the cassava is crushed with water and allowed to stand (‘ferment’), enzymes in the cassava will do the same job and then the HCN can be washed out before the cassava is cooked and eaten. The cassava is now safe to eat but it still contains some glucoside. Some diseases found in eastern Nigeria ca ...
... If the cassava is crushed with water and allowed to stand (‘ferment’), enzymes in the cassava will do the same job and then the HCN can be washed out before the cassava is cooked and eaten. The cassava is now safe to eat but it still contains some glucoside. Some diseases found in eastern Nigeria ca ...
Chapter 3 Alcohols, Phenols, and Ethers
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. R ...
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. R ...
Document
... forming a new OH group with retention of configuration. • The overall result of this two-step sequence is syn addition of the elements of H and OH to a double bond in an “anti-Markovnikov” fashion. ...
... forming a new OH group with retention of configuration. • The overall result of this two-step sequence is syn addition of the elements of H and OH to a double bond in an “anti-Markovnikov” fashion. ...
23.2 Alcohols, Ethers, and Amines
... Halogenation Reactions The addition of bromine to carbon-carbon multiple bonds is often used as a chemical test for unsaturation in an organic molecule. • Bromine has an orange color, but most bromine-substituted organic compounds are colorless. After mixing, if the solution remains colorless, the c ...
... Halogenation Reactions The addition of bromine to carbon-carbon multiple bonds is often used as a chemical test for unsaturation in an organic molecule. • Bromine has an orange color, but most bromine-substituted organic compounds are colorless. After mixing, if the solution remains colorless, the c ...
enantioselective zeolite-catalyzed reactions
... major topic of discussion and research.1 Homogeneous, asymmetry-inducing catalysts have been developed for a wide range of reactions, especially notable are those developed by Knowles, Noyori and Sharpless, for which they jointly received the 2001 Nobel Prize in Chemistry.2 Although these reactions ...
... major topic of discussion and research.1 Homogeneous, asymmetry-inducing catalysts have been developed for a wide range of reactions, especially notable are those developed by Knowles, Noyori and Sharpless, for which they jointly received the 2001 Nobel Prize in Chemistry.2 Although these reactions ...
Palladium and Ruthenium Catalyzed Reactions By Bryan Jaksic
... Part one of this thesis will discuss research which involves the direct comparison of the activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple me ...
... Part one of this thesis will discuss research which involves the direct comparison of the activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple me ...
06_10_13.html
... Two-step reaction sequence called hydroborationoxidation converts alkenes to alcohols with a regiochemistry opposite to Markovnikov's rule. ...
... Two-step reaction sequence called hydroborationoxidation converts alkenes to alcohols with a regiochemistry opposite to Markovnikov's rule. ...
19.7 Reversible Addition Reactions of Aldehydes and Ketones
... 19.6 Basicity of Aldehydes and Ketones 19.7 Reversible Addition Reactions of Aldehydes and Ketones 19.8 Reduction of Aldehydes and Ketones to Alcohols 19.9 Reactions of Aldehydes and Ketones with Grignard and ...
... 19.6 Basicity of Aldehydes and Ketones 19.7 Reversible Addition Reactions of Aldehydes and Ketones 19.8 Reduction of Aldehydes and Ketones to Alcohols 19.9 Reactions of Aldehydes and Ketones with Grignard and ...
Chapter - FIU Faculty Websites
... An approach: (1) Reduction to an aldehyde can be accomplished by using a more reactive carboxylic acid derivatives such as an acyl chloride, ester or nitrile, and (2) Use a less reactive hydride source than LiAlH4 (LAH). The use of a sterically hindered and less reactive aluminum hydride reagent is ...
... An approach: (1) Reduction to an aldehyde can be accomplished by using a more reactive carboxylic acid derivatives such as an acyl chloride, ester or nitrile, and (2) Use a less reactive hydride source than LiAlH4 (LAH). The use of a sterically hindered and less reactive aluminum hydride reagent is ...
Lorell Thesis Final Version in PDF S
... To my parents for teaching me the real values of life. To my husband, for teaching me how to be a better person. To my daughter, the best thing that has happened in my life. To my sisters and brother for the love that only siblings comprehend. ...
... To my parents for teaching me the real values of life. To my husband, for teaching me how to be a better person. To my daughter, the best thing that has happened in my life. To my sisters and brother for the love that only siblings comprehend. ...
Learning Guide for Chapter 9 - Alkyl Halides I
... do not. The important factor is the hybridization of the C the halogen is attached to. sp3 carbon alkyl halide benzyl, allyl, other aromatic sp2 carbon NOT an alkyl halide aryl, vinyl Label the following as methyl, 1o, 2o, 3o, allyl, and benzyl. ...
... do not. The important factor is the hybridization of the C the halogen is attached to. sp3 carbon alkyl halide benzyl, allyl, other aromatic sp2 carbon NOT an alkyl halide aryl, vinyl Label the following as methyl, 1o, 2o, 3o, allyl, and benzyl. ...
Ether - Clayton State University
... – Phenol first used by Joseph Lister in hospitals in 1800’s. – Phenol derivatives used today in Lysol, mouthwashes and throat lozenges. ...
... – Phenol first used by Joseph Lister in hospitals in 1800’s. – Phenol derivatives used today in Lysol, mouthwashes and throat lozenges. ...
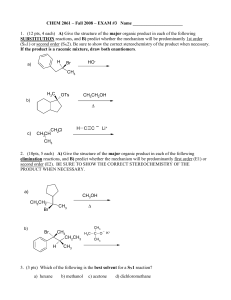
Exam #3
... A) Using mechanistic reasoning, EXPLAIN WHY menthyl chloride gives only 2-menthene as its E2 elimination product. B) Explain why 3-menthene (rather than 2- menthene) is the major product in the E2 elimination of neomenthyl chloride. H H3C ...
... A) Using mechanistic reasoning, EXPLAIN WHY menthyl chloride gives only 2-menthene as its E2 elimination product. B) Explain why 3-menthene (rather than 2- menthene) is the major product in the E2 elimination of neomenthyl chloride. H H3C ...
Part I Carbohydrate Auxiliaries - Wiley-VCH
... (modified Strecker reaction) are fundamental carbon–carbon bond-forming processes [3], which are efficient methods for preparing α-amino acids (Scheme 1.1). In 1987 Kunz and coworkers first reported pivaloyl protected d-galactosyl amine 3 as a very useful tool for asymmetric aminonitrile syntheses [4]. ...
... (modified Strecker reaction) are fundamental carbon–carbon bond-forming processes [3], which are efficient methods for preparing α-amino acids (Scheme 1.1). In 1987 Kunz and coworkers first reported pivaloyl protected d-galactosyl amine 3 as a very useful tool for asymmetric aminonitrile syntheses [4]. ...
Neuman Chapter - Department of Chemistry
... Since the C=C bond forms between the C's of the two C-X groups in the reactant, these reactions precisely place the C=C in the product. However, the dihaloalkane reactant is usually synthesized (Chapter 10) from the same alkene that it forms by elimination of X-X, so dehalogenation reactions are les ...
... Since the C=C bond forms between the C's of the two C-X groups in the reactant, these reactions precisely place the C=C in the product. However, the dihaloalkane reactant is usually synthesized (Chapter 10) from the same alkene that it forms by elimination of X-X, so dehalogenation reactions are les ...
Elias James Corey

Elias James ""E.J."" Corey (born July 12, 1928) is an American organic chemist. In 1990, he won the Nobel Prize in Chemistry ""for his development of the theory and methodology of organic synthesis"", specifically retrosynthetic analysis. Regarded by many as one of the greatest living chemists, he has developed numerous synthetic reagents, methodologies and total syntheses and has advanced the science of organic synthesis considerably.