Microsoft Word
... dispersion, (ii) enlarging oxygen storage capacity of the catalyst and (iv) high thermal stability. To better understand the catalytic nature of supported Pd catalysts, an in-depth study of their surface chemistry would be necessary. The supported metal systems are usually characterized by several ...
... dispersion, (ii) enlarging oxygen storage capacity of the catalyst and (iv) high thermal stability. To better understand the catalytic nature of supported Pd catalysts, an in-depth study of their surface chemistry would be necessary. The supported metal systems are usually characterized by several ...
Forward
... (b) formaldehyde. Both molecules have the same number of electrons, and carbon is sp2-hybridized in both. In formaldehyde, one of the carbons is replaced by an sp2hybridized oxygen (shown in red). Oxygen has two unshared electron pairs; each pair occupies an sp2hybridized orbital. Like the carbon–ca ...
... (b) formaldehyde. Both molecules have the same number of electrons, and carbon is sp2-hybridized in both. In formaldehyde, one of the carbons is replaced by an sp2hybridized oxygen (shown in red). Oxygen has two unshared electron pairs; each pair occupies an sp2hybridized orbital. Like the carbon–ca ...
1 Q. What are Saturated and unsaturated hydrocarbons?imp
... Staggered conformations has the lease torsional strain while eclipsed having maximum. Due to torsional strain, certain energy called torsional energy, is required to allow the rotation around the C-C single bond. in other words ethane molecule having staggered conformation will have to come cross an ...
... Staggered conformations has the lease torsional strain while eclipsed having maximum. Due to torsional strain, certain energy called torsional energy, is required to allow the rotation around the C-C single bond. in other words ethane molecule having staggered conformation will have to come cross an ...
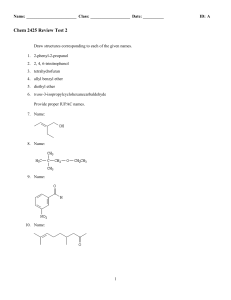
Chem 2425-Test 2 Review
... Consider the data below to answer the following question(s). Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed t ...
... Consider the data below to answer the following question(s). Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed t ...
1 Bite Angle Effects of Diphosphines in Carbonylation Reactions
... the selectivity, but the selectivity is determined in the alkene coordination to RhH(diphosphine)(CO) or in the hydride migration step. A plausible explanation of the bite angle effect is that in these steps, an augmentation of the steric congestion around the metal center is produced when the bite ...
... the selectivity, but the selectivity is determined in the alkene coordination to RhH(diphosphine)(CO) or in the hydride migration step. A plausible explanation of the bite angle effect is that in these steps, an augmentation of the steric congestion around the metal center is produced when the bite ...
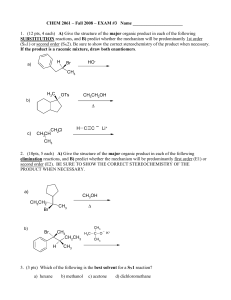
Exam #3
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
Chapter 22 and 23 Study Guide
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
Chapter 1 Structure and Bonding
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
Si(OR - am Lehrstuhl für Bauchemie
... Alkoxy silanes react readily with water by reaction called hydrolysis. The hydrolysis reaction, through the addition of water, replaces alkoxide group (OR) with hydroxyl group (OH-) which becomes attached to the silicon atom. Depending on the amount of water and catalysts present, hydrolysis may go ...
... Alkoxy silanes react readily with water by reaction called hydrolysis. The hydrolysis reaction, through the addition of water, replaces alkoxide group (OR) with hydroxyl group (OH-) which becomes attached to the silicon atom. Depending on the amount of water and catalysts present, hydrolysis may go ...
Alcohols
... Reaction is unusually broad and useful method of alcohol synthesis Reaction does have an indirect biological counterpart • The addition of stabilized carbon nucleophiles to carbonyl compounds is used in almost all metabolic pathways as the major process for forming carboncarbon bonds ...
... Reaction is unusually broad and useful method of alcohol synthesis Reaction does have an indirect biological counterpart • The addition of stabilized carbon nucleophiles to carbonyl compounds is used in almost all metabolic pathways as the major process for forming carboncarbon bonds ...
BIS(CYCLOPENTADIENYL TRANSITION METAL) COMPLEXES
... (C,Me,),Cr,S, and (C,Me,),Cr,S,, but they react under H2 pressure with formation of the cubane-like cluster (C,Me,),Cr,S,.’2 A mechanism for this reaction has not yet been established, but it is very likely, that the presence of H, is essential for this reaction. In this regard it may be of interest ...
... (C,Me,),Cr,S, and (C,Me,),Cr,S,, but they react under H2 pressure with formation of the cubane-like cluster (C,Me,),Cr,S,.’2 A mechanism for this reaction has not yet been established, but it is very likely, that the presence of H, is essential for this reaction. In this regard it may be of interest ...
Methanol Oxidative Dehydrogenation on Oxide Catalysts: Molecular
... involving CH2OH radical species formed via direct Habstraction from molecular CH3OH.19 Direct and rigorous comparisons between molecular and dissociative routes require the assessment of enthalpic and entropic factors that determine the relative contributions of these routes to turnover rates; this ...
... involving CH2OH radical species formed via direct Habstraction from molecular CH3OH.19 Direct and rigorous comparisons between molecular and dissociative routes require the assessment of enthalpic and entropic factors that determine the relative contributions of these routes to turnover rates; this ...
Ch 9 Lecture 2
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
13: Carbonyl Compounds: Ketones, Aldehydes, Carboxylic Acids
... We systematically name aldehydes as "alkanals" and we show some simple examples in Figure 13.9. Figure 13.9 The systematic nomenclature for aldehydes derives from the systematic nomenclature for 1° alcohols because oxidation of 1° alcohols gives aldehydes. The ending "ol" in the name of the precurso ...
... We systematically name aldehydes as "alkanals" and we show some simple examples in Figure 13.9. Figure 13.9 The systematic nomenclature for aldehydes derives from the systematic nomenclature for 1° alcohols because oxidation of 1° alcohols gives aldehydes. The ending "ol" in the name of the precurso ...
HMDS - Sigma
... is readily lost from the transition state during reaction, but possesses sufficient chemical stability in combination with the alkyl silyl group to allow long term storage of the derivatizing agent for use as required. As the formation of the transition state is reversible, the derivatization will o ...
... is readily lost from the transition state during reaction, but possesses sufficient chemical stability in combination with the alkyl silyl group to allow long term storage of the derivatizing agent for use as required. As the formation of the transition state is reversible, the derivatization will o ...
Melt Modification of Poly(styrene-co-maleic anhydride)
... acids as shown in Figure 1 (pathway b). Consequently, C11OXA addition converted free carboxylic acid. In order to quantify the anhydride conversion, the ANH1780 signal of the products is compared with the ANH1780 signal of the educt SMA17. For the binary system, S17A1, an anhydride conversion of onl ...
... acids as shown in Figure 1 (pathway b). Consequently, C11OXA addition converted free carboxylic acid. In order to quantify the anhydride conversion, the ANH1780 signal of the products is compared with the ANH1780 signal of the educt SMA17. For the binary system, S17A1, an anhydride conversion of onl ...
幻灯片 1
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... produce valuable diarylalkane products21 in high yields and excellent regioselectivity. This process constitutes the first general application of graphenes to promote direct C−C bond formation, utilizing polar functional groups anchored on the GO surface,23−30 which may open the door for industrial a ...
... produce valuable diarylalkane products21 in high yields and excellent regioselectivity. This process constitutes the first general application of graphenes to promote direct C−C bond formation, utilizing polar functional groups anchored on the GO surface,23−30 which may open the door for industrial a ...
Assessing the Potential for the Reactions of
... primary amine addition product, an alcohol group would be formed at the secondary carbon (which has one proton) and for a secondary amine addition product, an alcohol group would be formed at the primary carbon (which has two distinguishable protons). In addition to the epoxide reactant peaks at 3.0 ...
... primary amine addition product, an alcohol group would be formed at the secondary carbon (which has one proton) and for a secondary amine addition product, an alcohol group would be formed at the primary carbon (which has two distinguishable protons). In addition to the epoxide reactant peaks at 3.0 ...
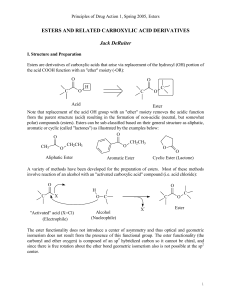
esters and related carboxylic acid derivatives
... Thus the esters carbonyl carbon is susceptible to "attack" by electron rich atoms (nucleophiles) including the oxygen of water and the nucleophilic residues at the active sites of esterase enzymes. When in the presence of a nucleophile, an ester may undergo reaction leading to cleavage of the carbon ...
... Thus the esters carbonyl carbon is susceptible to "attack" by electron rich atoms (nucleophiles) including the oxygen of water and the nucleophilic residues at the active sites of esterase enzymes. When in the presence of a nucleophile, an ester may undergo reaction leading to cleavage of the carbon ...
Amidations of Rosin with Isocyanates
... that of triethylamine. No significant difference in catalysis efficiency could be observed for the addition amounts of the catalysts between 10 and 20 mg/g abietic acid. The results demonstrate that the tertiary amines can greatly accelerate the reaction rate of abietic acid with phenyl isocyanate. ...
... that of triethylamine. No significant difference in catalysis efficiency could be observed for the addition amounts of the catalysts between 10 and 20 mg/g abietic acid. The results demonstrate that the tertiary amines can greatly accelerate the reaction rate of abietic acid with phenyl isocyanate. ...
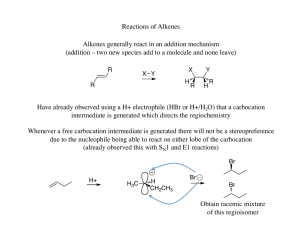
Chapter 11
... The electrophilic mercury reacts with an alkene to form a mercurinium ion which is similar to bromonium ions in that a three membered ring is formed with a partial bond to the carbon that can best handle the partial positive charge ...
... The electrophilic mercury reacts with an alkene to form a mercurinium ion which is similar to bromonium ions in that a three membered ring is formed with a partial bond to the carbon that can best handle the partial positive charge ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... carbinols were obtained in high yields. A formal synthesis of carbinoxamine, an antihistamine drug used for the treatment of seasonal allergies and hay fever, emerges from this methodology. As an update, the [1,2]-anionic rearrangement of benzyl 2-pyridyl ethers can also be accessed by a distinct an ...
... carbinols were obtained in high yields. A formal synthesis of carbinoxamine, an antihistamine drug used for the treatment of seasonal allergies and hay fever, emerges from this methodology. As an update, the [1,2]-anionic rearrangement of benzyl 2-pyridyl ethers can also be accessed by a distinct an ...
Alkenes 3 - ChemWeb (UCC)
... in the same plane to overlap in a sideways-on fashion and form the component of the double bond. This developing -overlap is most efficient if the C-H and C-X bonds - and hence the developing p-orbitals - are already in the same plane in the transition state. A syn periplanar conformation also pr ...
... in the same plane to overlap in a sideways-on fashion and form the component of the double bond. This developing -overlap is most efficient if the C-H and C-X bonds - and hence the developing p-orbitals - are already in the same plane in the transition state. A syn periplanar conformation also pr ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.