Nucleophilic Acyl Substitution
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
Class Notes
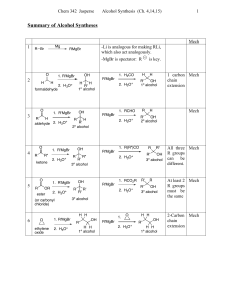
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
Chiral Enolate Equivalents
... confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophiles.3 Intramolecular reactions may tolerate slightly less reactive electrophiles. Wi ...
... confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophiles.3 Intramolecular reactions may tolerate slightly less reactive electrophiles. Wi ...
CHEMKIN Tutorials Manual
... SURFTHERM, TRANSPORT, TWAFER, TWOPNT are all trademarks of Reaction Design or Sandia National Laboratories. All other trademarks are the property of their respective holders. ...
... SURFTHERM, TRANSPORT, TWAFER, TWOPNT are all trademarks of Reaction Design or Sandia National Laboratories. All other trademarks are the property of their respective holders. ...
Boron Reagents in Process Chemistry: Excellent
... trialkylboranes. With more hindered olefins, the hydroboration can be stopped at the mono- or disubstituted organoborane. For example, hydroboration of 2-methyl-2butene proceeds only to the dialkyl stage, generating the corresponding disiamylborane. A very useful regioselective hydroborating agent, ...
... trialkylboranes. With more hindered olefins, the hydroboration can be stopped at the mono- or disubstituted organoborane. For example, hydroboration of 2-methyl-2butene proceeds only to the dialkyl stage, generating the corresponding disiamylborane. A very useful regioselective hydroborating agent, ...
Copper-Catalyzed Coupling Reactions Using Carbon
... Oxidative coupling reactions with C-H bonds are quite challenging due to the relative strong C-H bond, along with the associated selectivity issues with the myriad of C-H bonds that are available for the coupling reaction. ...
... Oxidative coupling reactions with C-H bonds are quite challenging due to the relative strong C-H bond, along with the associated selectivity issues with the myriad of C-H bonds that are available for the coupling reaction. ...
C−C, C−O, C−N Bond Formation on sp2 Carbon by Pd(II)
... the bond susceptible to addition reactions. Many of these are catalyzed by palladium, perhaps the most versatile and widely used transition metal, which can exist in three easily interconvertible oxidation states: Pd(0), Pd(II), and Pd(IV). A wealth of reviews1 and books2 on organopalladium chemistr ...
... the bond susceptible to addition reactions. Many of these are catalyzed by palladium, perhaps the most versatile and widely used transition metal, which can exist in three easily interconvertible oxidation states: Pd(0), Pd(II), and Pd(IV). A wealth of reviews1 and books2 on organopalladium chemistr ...
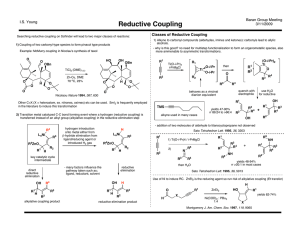
Reductive Couplings
... - desired reaction is three-component alkylative coupling (RC was a competing problem) - imines less electrophilic - need hydroxylic solvent and organoboron reagent - methanol occupies coordination site, hindering β-hydride elimination ...
... - desired reaction is three-component alkylative coupling (RC was a competing problem) - imines less electrophilic - need hydroxylic solvent and organoboron reagent - methanol occupies coordination site, hindering β-hydride elimination ...
Chapter 17 Amines
... the decomposition of aryl diazonium Chlorides to chloroarenes in the presence of copper (I) chloride in 1884. He also worked on the triphenylmethane dyes and the synthesis of isatin. Many years before, he had suggested Traugott Sandmeyer to Victor Meyer an impurity in commerical benzene was responsi ...
... the decomposition of aryl diazonium Chlorides to chloroarenes in the presence of copper (I) chloride in 1884. He also worked on the triphenylmethane dyes and the synthesis of isatin. Many years before, he had suggested Traugott Sandmeyer to Victor Meyer an impurity in commerical benzene was responsi ...
Modern Synthetic Methods for Copper-Mediated C(aryl
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
Forward
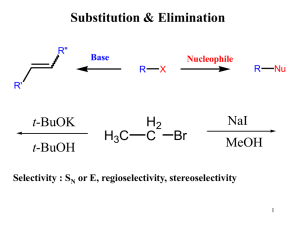
... Secondary and tertiary alkyl halides are not suitable, because they tend to react with alkoxide bases by E2 elimination rather than by SN2 substitution. Whether the alkoxide base is primary, secondary, or tertiary is much less important than the nature of the alkyl halide. Thus benzyl isopropyl ethe ...
... Secondary and tertiary alkyl halides are not suitable, because they tend to react with alkoxide bases by E2 elimination rather than by SN2 substitution. Whether the alkoxide base is primary, secondary, or tertiary is much less important than the nature of the alkyl halide. Thus benzyl isopropyl ethe ...
Elimination Reactions
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
Alcohols
... – they do not react with reducing agents such as H2 in the presence of a transition metal catalyst – they are not affected by most acids or bases at moderate temperatures (but can eliminate) ...
... – they do not react with reducing agents such as H2 in the presence of a transition metal catalyst – they are not affected by most acids or bases at moderate temperatures (but can eliminate) ...
Matrix Isolation Spectroscopic and Theoretical Study of Carbon
... as well as simple metal oxide molecules with CO2 serve as the simplest model in understanding the intrinsic mechanism of catalytic CO2 activation processes. The reactions of atomic transition metal cations and neutrals with carbon dioxide have been intensively studied both experimentally and theoret ...
... as well as simple metal oxide molecules with CO2 serve as the simplest model in understanding the intrinsic mechanism of catalytic CO2 activation processes. The reactions of atomic transition metal cations and neutrals with carbon dioxide have been intensively studied both experimentally and theoret ...
Chapter 10:Alcohols, Phenols and Ethers
... hydroxyl group is strongly activating and ortho, para-directing. Phenols are excellent substrates for halogention, nitration, sulfonation, and Friedel-Crafts reactions. Becaues they are highly reactive, phenols are usually alkylated or acylated using relatively weak Friedel-Crafts catalysts (such as ...
... hydroxyl group is strongly activating and ortho, para-directing. Phenols are excellent substrates for halogention, nitration, sulfonation, and Friedel-Crafts reactions. Becaues they are highly reactive, phenols are usually alkylated or acylated using relatively weak Friedel-Crafts catalysts (such as ...
Document
... • Because the functional group is at C1, the 1 is usually omitted from the name. • The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lowest number. ...
... • Because the functional group is at C1, the 1 is usually omitted from the name. • The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lowest number. ...
Alcohols, Phenols, and Ethers
... Classify the following alcohols as primary, secondary, or tertiary The –OH group is polar and capable of hydrogen bonding. This makes low molecular weight alcohols highly soluble in water. Hydrogen bonding in a water-methanol solution: ...
... Classify the following alcohols as primary, secondary, or tertiary The –OH group is polar and capable of hydrogen bonding. This makes low molecular weight alcohols highly soluble in water. Hydrogen bonding in a water-methanol solution: ...
Module I Oxidation Reactions
... MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, Inc., L. A. Paquette Ed., N ...
... MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, Inc., L. A. Paquette Ed., N ...
Chapter 7. Alcohols, Thiols, Phenols, Ethers
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
OChem1 Course Pack
... produces an anion in which the charge is localized on O and therefore unstable. The carboxylic acid produces a conjugate base in which the charge is delocalized onto two O atoms, which is quite stable. The third molecule also has a resonance-stabilized conjugate base, however here the charge is delo ...
... produces an anion in which the charge is localized on O and therefore unstable. The carboxylic acid produces a conjugate base in which the charge is delocalized onto two O atoms, which is quite stable. The third molecule also has a resonance-stabilized conjugate base, however here the charge is delo ...
Efficient hydrogenation of organic carbonates, carbamates and
... reactions8. Moreover, the popular amine protecting groups, benzyl carbamates, undergo deprotection by heterogeneous hydrogenation, which involves cleavage of the benzyl–O bond, but with the carbonyl group not being reduced 9. In addition, hydrogenation of cyclic N-acylcarbamates leads to cleavage of ...
... reactions8. Moreover, the popular amine protecting groups, benzyl carbamates, undergo deprotection by heterogeneous hydrogenation, which involves cleavage of the benzyl–O bond, but with the carbonyl group not being reduced 9. In addition, hydrogenation of cyclic N-acylcarbamates leads to cleavage of ...
Zn(BH4)2/Al2O3: A new synthetic method for the efficient
... method is fast for the reduction of aldehydes, in the case of ketones, the need for large amounts of NaBH4 (eightfold) and the moderate yields of the products are major limitations.5c–d On the other hand, zinc tetrahydroborate, Zn(BH4)2, as a non-conventional hydride transfer agent, has been reporte ...
... method is fast for the reduction of aldehydes, in the case of ketones, the need for large amounts of NaBH4 (eightfold) and the moderate yields of the products are major limitations.5c–d On the other hand, zinc tetrahydroborate, Zn(BH4)2, as a non-conventional hydride transfer agent, has been reporte ...
aa-2005-38-71-negishi - University of Windsor
... alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalization may have to be significantly modified in the future, as discussed in Section 2.6. Another group of categorically difficul ...
... alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalization may have to be significantly modified in the future, as discussed in Section 2.6. Another group of categorically difficul ...
lec-3- 211( Elim+ Re..
... • Drug metabolism can occur in every tissue (e.g. gut, lung and kidney). However, the major drug metabolizing enzymes (DMEs) are expressed at the highest levels in the liver, which thus serves as the major organ of metabolic clearance • Drug metabolism serves to control the exposure of a potentially ...
... • Drug metabolism can occur in every tissue (e.g. gut, lung and kidney). However, the major drug metabolizing enzymes (DMEs) are expressed at the highest levels in the liver, which thus serves as the major organ of metabolic clearance • Drug metabolism serves to control the exposure of a potentially ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.