
One step hydroxylation of benzene to phenol using N2O
... to examine the effect of different Si/Al ratios, temperatures and iron content on the selective conversion of benzene to phenol with a desire to achieve high selectivity and minimise catalyst deactivation. Also one of the research objectives was to identify the active sites in the catalyst and desig ...
... to examine the effect of different Si/Al ratios, temperatures and iron content on the selective conversion of benzene to phenol with a desire to achieve high selectivity and minimise catalyst deactivation. Also one of the research objectives was to identify the active sites in the catalyst and desig ...
Lorell Thesis Final Version in PDF S
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
Experimental details
... pressure as well as the very intense ionizing radiation emitted. The relevant materials in nuclear technological applications are required to be stable and durable after having withstood these extreme conditions. The materials stability under high temperature and pressure is primarily controlled by ...
... pressure as well as the very intense ionizing radiation emitted. The relevant materials in nuclear technological applications are required to be stable and durable after having withstood these extreme conditions. The materials stability under high temperature and pressure is primarily controlled by ...
Excited State Reactions of Carbonyl Compounds
... behave similar to an alkene, the fact that the electron distribution in p and p* orbitals is unequal between carbon and oxygen of the C=O bond would make it behave slightly differently. Since neither 1 pp* nor 3 pp* are the lowest excited states in alkyl ketones and aldehydes we will not consider th ...
... behave similar to an alkene, the fact that the electron distribution in p and p* orbitals is unequal between carbon and oxygen of the C=O bond would make it behave slightly differently. Since neither 1 pp* nor 3 pp* are the lowest excited states in alkyl ketones and aldehydes we will not consider th ...
Recent developments in the synthesis of functional poly(olefin)s
... Functionalization of poly(ole®n)s especially poly(propylene), poly(ethylene) and poly(ethylene-copropylene) has been extensively investigated using melt phase free radical grafting. The synthetic technique involves activation of the polymer by shear in presence of free radical initiators or high ene ...
... Functionalization of poly(ole®n)s especially poly(propylene), poly(ethylene) and poly(ethylene-copropylene) has been extensively investigated using melt phase free radical grafting. The synthetic technique involves activation of the polymer by shear in presence of free radical initiators or high ene ...
Lithium Iodide Original Commentary - Groupe Charette
... Heterolytic C–X Bond Cleaving Reactions. In the presence of amine bases, LiI has been extensively used as a mild reagent for the chemoselective cleavage of methyl esters (eq 1).6 Decarboxylation of methyl esters usually occurs when an electronwithdrawing group is present at the α-position of the est ...
... Heterolytic C–X Bond Cleaving Reactions. In the presence of amine bases, LiI has been extensively used as a mild reagent for the chemoselective cleavage of methyl esters (eq 1).6 Decarboxylation of methyl esters usually occurs when an electronwithdrawing group is present at the α-position of the est ...
PowerPoint - Naming Hydrocarbons
... Alkyl Groups -groups that are formed when one hydrogen atom is removed from an alkane - the suffix “-ane” is replaced by “-yl” ...
... Alkyl Groups -groups that are formed when one hydrogen atom is removed from an alkane - the suffix “-ane” is replaced by “-yl” ...
Corrosion studies of LiH thin films
... 640 K in the LiH TPD indicates that there is no free Li metal and that the film has fully hydrided. As a result of these observations we conclude that we have successfully synthesised a thin film of high purity LiH, in agreement with the assertion that the single Li KLL emission at 44.8 eV originates ...
... 640 K in the LiH TPD indicates that there is no free Li metal and that the film has fully hydrided. As a result of these observations we conclude that we have successfully synthesised a thin film of high purity LiH, in agreement with the assertion that the single Li KLL emission at 44.8 eV originates ...
presentation source
... Some molecules with the amide functional group acetaminophen (active ingredient in nonaspirin pain relievers; used to make dyes and photographic chemicals) ...
... Some molecules with the amide functional group acetaminophen (active ingredient in nonaspirin pain relievers; used to make dyes and photographic chemicals) ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
Chem. Soc. Rev., 2015, 44, 2202--2220 - RSC Publishing
... of imines but results were inconclusive.21 Initial attempts to determine the absolute configuration and the enantiomeric purity of the silicon-stereogenic hydrosilane reisolated after the hydrosilylation/Si–N cleavage sequence had failed. Also, the stereogenicity at the silicon atom had not induced ...
... of imines but results were inconclusive.21 Initial attempts to determine the absolute configuration and the enantiomeric purity of the silicon-stereogenic hydrosilane reisolated after the hydrosilylation/Si–N cleavage sequence had failed. Also, the stereogenicity at the silicon atom had not induced ...
- University of Bath Opus
... sulfonates on scale, both of which require an extra step which generates more waste and results in poor atom economy. More importantly, most alkyl halides and sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs ...
... sulfonates on scale, both of which require an extra step which generates more waste and results in poor atom economy. More importantly, most alkyl halides and sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs ...
The Carbonyl Group - Angelo State University
... • Carbonyl compounds cannot hydrogen-bond to each other, but they can hydrogen-bond to water through the carbonyl oxygen. • Low-molecular weight aldehydes and ketones are water-soluble; water solubility decreases as the size of the molecule increases. ...
... • Carbonyl compounds cannot hydrogen-bond to each other, but they can hydrogen-bond to water through the carbonyl oxygen. • Low-molecular weight aldehydes and ketones are water-soluble; water solubility decreases as the size of the molecule increases. ...
PFC ,RR-86-1 The Kinetics Of Liquid Lithium Reaction With Oxygen
... prototypical fusion reactor conditions under both air and nitrogen atmospheres is performed. And third, a lithium-carbon dioxide reaction model is added to the code allowing pure carbon dioxide environment in which the lithium-CO 2 reaction can take place, and normal air environment in which lithium ...
... prototypical fusion reactor conditions under both air and nitrogen atmospheres is performed. And third, a lithium-carbon dioxide reaction model is added to the code allowing pure carbon dioxide environment in which the lithium-CO 2 reaction can take place, and normal air environment in which lithium ...
New Stereoselective Approaches to Highly Substituted
... In studies towards the synthesis o f substituted pyrroles, the Knight group have adapted an aldol reaction, originally developed by Kazmaier, as a highly diastereoselective method for the synthesis o f the cyclisadon precursors. Both acetylenic and a,f3-unsaturated aldehydes have been successfully u ...
... In studies towards the synthesis o f substituted pyrroles, the Knight group have adapted an aldol reaction, originally developed by Kazmaier, as a highly diastereoselective method for the synthesis o f the cyclisadon precursors. Both acetylenic and a,f3-unsaturated aldehydes have been successfully u ...
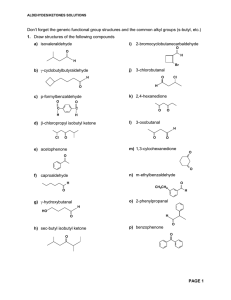
Aldehydes/Ketones Solutions
... The aldehyde is more easily reduced than the ketone so you must first add a protecting group to the aldehyde; reduce the ketone; and finally remove the protecting group to regenerate the aldehyde. ...
... The aldehyde is more easily reduced than the ketone so you must first add a protecting group to the aldehyde; reduce the ketone; and finally remove the protecting group to regenerate the aldehyde. ...
Catalytic Synthesis of Higher Alcohols from Syngas
... member of my research committee, for their help in conducting experiments and useful suggestions throughout my research work. The willingness of the other committee members, Prof. Steven McIntosh, Prof. Mark A. Snyder, and Prof. James T. Hsu, to assist me in their personal fields of expertise is gre ...
... member of my research committee, for their help in conducting experiments and useful suggestions throughout my research work. The willingness of the other committee members, Prof. Steven McIntosh, Prof. Mark A. Snyder, and Prof. James T. Hsu, to assist me in their personal fields of expertise is gre ...
Alkenes and Alkynes I
... 15A. Syn Addition of Hydrogen: Synthesis of cis -Alkenes Semi-hydrogenation of alkynes to alkenes can be achieved using either the Ni2B (P-2) catalyst or the Lindlar’s ...
... 15A. Syn Addition of Hydrogen: Synthesis of cis -Alkenes Semi-hydrogenation of alkynes to alkenes can be achieved using either the Ni2B (P-2) catalyst or the Lindlar’s ...
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... • Draw structural formulas for the following molecules: – 4-chloro-2-phenylpentanal ...
... • Draw structural formulas for the following molecules: – 4-chloro-2-phenylpentanal ...
Document
... • With base, the nucleophile is ¯OH, and the mechanism follows the usual two steps: nucleophilic attack followed by protonation. • The reaction rate increases in the presence of base because the base converts H2O into ¯OH, a stronger ...
... • With base, the nucleophile is ¯OH, and the mechanism follows the usual two steps: nucleophilic attack followed by protonation. • The reaction rate increases in the presence of base because the base converts H2O into ¯OH, a stronger ...
SUPPORTED LIGANDS FOR METAL CATALYZED REACTIONS Rocío Marcos Escartín ISBN:
... Similarly to Brønsted acids, Lewis acids are applied as catalysts in various chemical processes. The wide variety of different Lewis acids available, with significantly different properties, as well as the possibility of tuning them by the use of organic ligands renders them an extremely versatile a ...
... Similarly to Brønsted acids, Lewis acids are applied as catalysts in various chemical processes. The wide variety of different Lewis acids available, with significantly different properties, as well as the possibility of tuning them by the use of organic ligands renders them an extremely versatile a ...
[Ru(Triphos)H2(CO)] Characterisation - Durham e
... low oxidation states through back bonding. The high ligand field strength of phosphines can be important in catalysis, as the stabilisation of the low oxidation states prevents the reducing conditions of reactions, such as hydrogenation or hydroformylation, causing the precipitation of the metal, a ...
... low oxidation states through back bonding. The high ligand field strength of phosphines can be important in catalysis, as the stabilisation of the low oxidation states prevents the reducing conditions of reactions, such as hydrogenation or hydroformylation, causing the precipitation of the metal, a ...
Iodine and Lipase Based Green Oxidation Technology
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
Elementary Steps, the Role of Chemisorbed Oxygen, and the Effects
... relevance. These metal-oxide transformations occur in response to changes in oxygen chemical potential and temperature15!17 as O2 is depleted along the reactor or within porous catalyst pellets. These changes in chemical state can occur concurrently with structural transformations that alter the sha ...
... relevance. These metal-oxide transformations occur in response to changes in oxygen chemical potential and temperature15!17 as O2 is depleted along the reactor or within porous catalyst pellets. These changes in chemical state can occur concurrently with structural transformations that alter the sha ...
Project Overview
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.

















![[Ru(Triphos)H2(CO)] Characterisation - Durham e](http://s1.studyres.com/store/data/017676948_1-4352644236c53cc416f065328f560d26-300x300.png)


