Williamson Ether Synthesis
... or sulfonate ester under SN2 conditions. The parent alcohol of the alkoxide can be used as the solvent, however other polar solvents are often better, such as DMSO (dimethyl sulfoxide) or HMPA (hexamethylphosphoric triamide). ...
... or sulfonate ester under SN2 conditions. The parent alcohol of the alkoxide can be used as the solvent, however other polar solvents are often better, such as DMSO (dimethyl sulfoxide) or HMPA (hexamethylphosphoric triamide). ...
Alcohols - Calderglen High School
... Peeled apples turn brown due to the reaction of compounds called phenols. The first two steps in the reaction of one phenol, A are; ...
... Peeled apples turn brown due to the reaction of compounds called phenols. The first two steps in the reaction of one phenol, A are; ...
Chem 3.5 #3 Alcohols 1
... Explain why the lower molecular weight alcohols are soluble in water while the higher ones are not. ...
... Explain why the lower molecular weight alcohols are soluble in water while the higher ones are not. ...
File - TGHS Level 3 Chemistry
... are soluble in water as they are ionised, but they have a carbon chain end that is soluble in fats and oils. This allows them to dissolve and break down dirt. Sodium laurate is the name of the soap molecule made from coconut oil made by boiling it with sodium hydroxide. ...
... are soluble in water as they are ionised, but they have a carbon chain end that is soluble in fats and oils. This allows them to dissolve and break down dirt. Sodium laurate is the name of the soap molecule made from coconut oil made by boiling it with sodium hydroxide. ...
Derivatives of carboxylic acids - amides, acid anhydrides and nitriles
... Simple compounds containing an -NH2 group such as ammonia, NH3, or a primary amine like methylamine, CH3NH2, are weak bases. A primary amine is a compound where the -NH2 group is attached to a hydrocarbon group. The active lone pair of electrons on the nitrogen atom in ammonia can combine with a hyd ...
... Simple compounds containing an -NH2 group such as ammonia, NH3, or a primary amine like methylamine, CH3NH2, are weak bases. A primary amine is a compound where the -NH2 group is attached to a hydrocarbon group. The active lone pair of electrons on the nitrogen atom in ammonia can combine with a hyd ...
ch08 by dr. Dina
... The hydration of alkenes and the dehydration of alcohols are simply reverse reactions of one other The reaction is governed by the position of all the equilibria Hydration is favored by addition of a small amount of acid and a large amount of water Dehydration is favored by concentrated acid with ve ...
... The hydration of alkenes and the dehydration of alcohols are simply reverse reactions of one other The reaction is governed by the position of all the equilibria Hydration is favored by addition of a small amount of acid and a large amount of water Dehydration is favored by concentrated acid with ve ...
Synthesis of n-Butyl Acetate via Esterification
... Dowex resin as received should be washed with water by decantation to remove much of the yellow color. It is then collected by vacuum filtration on a Buchner funnel before use], 0.61 g (0.58 mL) of acetic acid, 0.74 g (0.91 mL) of 1-butanol, and a 1/2" stirring bar. Set up the Dean-Stark trap as sho ...
... Dowex resin as received should be washed with water by decantation to remove much of the yellow color. It is then collected by vacuum filtration on a Buchner funnel before use], 0.61 g (0.58 mL) of acetic acid, 0.74 g (0.91 mL) of 1-butanol, and a 1/2" stirring bar. Set up the Dean-Stark trap as sho ...
Nucleophilic Substitution Reactions of Epoxides
... aromatic ring has been converted into an epoxide. What happens to aromatic compounds when they enter the body as a foreign substance (such as cigarette smoke, drugs, charcoalbroiled meats or automobile exhaust)? ...
... aromatic ring has been converted into an epoxide. What happens to aromatic compounds when they enter the body as a foreign substance (such as cigarette smoke, drugs, charcoalbroiled meats or automobile exhaust)? ...
Notes on Alkynes
... in the enhanced acidity of the sp-hybridized C-H bond. Upon deprotonating, we now have a great carbon nucleophile that we can use in a SN2 reaction to make a new carbon-carbon bond. Alkynes are very useful in organic synthesis for this reason; they provide a method to make carbon-carbon bonds, and i ...
... in the enhanced acidity of the sp-hybridized C-H bond. Upon deprotonating, we now have a great carbon nucleophile that we can use in a SN2 reaction to make a new carbon-carbon bond. Alkynes are very useful in organic synthesis for this reason; they provide a method to make carbon-carbon bonds, and i ...
Aldehydes and ketones
... – Select as the parent chain the longest continuous chain that involves the carbon of the carbonyl group – Name the parent chain by removing the “e” from the corresponding alkane name and adding “one” – Number the chain to give the carbonyl group the lowest numbering. The number goes before the pare ...
... – Select as the parent chain the longest continuous chain that involves the carbon of the carbonyl group – Name the parent chain by removing the “e” from the corresponding alkane name and adding “one” – Number the chain to give the carbonyl group the lowest numbering. The number goes before the pare ...
Chapter 20: Carboxylic Acids
... reaction is quantitative and very easy…but diazomethane has a tendency to explode…. ...
... reaction is quantitative and very easy…but diazomethane has a tendency to explode…. ...
Παρουσίαση του PowerPoint
... 4.2 The solution to the problems: Guideline 1: When the two groups are of unequal reactivity, the more reactive can be made to react alone. Paracetamol: Analysis H N HO ...
... 4.2 The solution to the problems: Guideline 1: When the two groups are of unequal reactivity, the more reactive can be made to react alone. Paracetamol: Analysis H N HO ...
Παρουσίαση του PowerPoint
... 4.2 The solution to the problems: Guideline 1: When the two groups are of unequal reactivity, the more reactive can be made to react alone. Paracetamol: Analysis H N HO ...
... 4.2 The solution to the problems: Guideline 1: When the two groups are of unequal reactivity, the more reactive can be made to react alone. Paracetamol: Analysis H N HO ...
Chapter Sixteen Aldehydes and Ketones
... ► Aldehydes and ketones establish equilibria with alcohols to form hemiacetals or acetals. ► Hemiacetals, which have an -OH and an -OR on what was the carbonyl carbon, result from addition of one alcohol molecule to the C=O bond. ► The more stable acetals, which have two -OR groups on what was the c ...
... ► Aldehydes and ketones establish equilibria with alcohols to form hemiacetals or acetals. ► Hemiacetals, which have an -OH and an -OR on what was the carbonyl carbon, result from addition of one alcohol molecule to the C=O bond. ► The more stable acetals, which have two -OR groups on what was the c ...
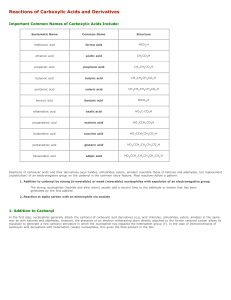
Reactions of Carboxylic Acids and Derivatives
... In the first step, nucleophiles generally attack the carbonyl of carboxylic acid derivatives (e.g. acid chlorides, anhydrides, esters, amides) in the same way as with ketones and aldehydes. However, the presence of an electron withdrawing atom directly attached to the former carbonyl carbon allows i ...
... In the first step, nucleophiles generally attack the carbonyl of carboxylic acid derivatives (e.g. acid chlorides, anhydrides, esters, amides) in the same way as with ketones and aldehydes. However, the presence of an electron withdrawing atom directly attached to the former carbonyl carbon allows i ...
Chapter 6: Alkynes, reactions of alkynes, and multistep synthesis
... c. easly get aldehyde 5. Hydrogenation a. usually can’t stop at alkene b. can stop only if using “poison” catalyst (Lindlar catalyst) c. get cis alkene for syn addition with Lindlar d. to get trans, use Na or Li in liquid ammonia (-78ºC) e. this is radical addition ...
... c. easly get aldehyde 5. Hydrogenation a. usually can’t stop at alkene b. can stop only if using “poison” catalyst (Lindlar catalyst) c. get cis alkene for syn addition with Lindlar d. to get trans, use Na or Li in liquid ammonia (-78ºC) e. this is radical addition ...
Synthesis of Methyl Benzoate by Fisher Esterification
... equilibrium. To upset the equilibrium we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling p ...
... equilibrium. To upset the equilibrium we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling p ...
- Iranian Journal of Science and Technology (Sciences)
... immiscibility of ionic liquids with a number of organic solvents provides a solution for biphasic separation of the desired products. Therefore, here high conversions have been achieved with a convenience of separation of the ester product from the reaction mixture. This makes the workup much easier ...
... immiscibility of ionic liquids with a number of organic solvents provides a solution for biphasic separation of the desired products. Therefore, here high conversions have been achieved with a convenience of separation of the ester product from the reaction mixture. This makes the workup much easier ...
chapter 8 lecture
... • Two elimination reactions are needed to remove two moles of HX from a dihalide substrate. • Two different starting materials can be used—a vicinal dihalide or a geminal dihalide. ...
... • Two elimination reactions are needed to remove two moles of HX from a dihalide substrate. • Two different starting materials can be used—a vicinal dihalide or a geminal dihalide. ...
Direct production of hydrogen peroxide from CO, O2, and H2O over
... Table 1 shows the catalytic results for H2O2 production from CO/O2/H2O over several types of metal nanoparticles dispersed on alumina prepared by the wet reduction (WR) method, which has recently been shown to be an effective method for the preparation of various amorphous alloy catalysts for versati ...
... Table 1 shows the catalytic results for H2O2 production from CO/O2/H2O over several types of metal nanoparticles dispersed on alumina prepared by the wet reduction (WR) method, which has recently been shown to be an effective method for the preparation of various amorphous alloy catalysts for versati ...
Carbon-Carbon Bond Forming Reactions
... - mixed aldol: reaction of two different aldehydes and/or ketones - requires quantitative deprotonation of one carbonyl component (LDA) - low temperature reaction typically gives aldol product ...
... - mixed aldol: reaction of two different aldehydes and/or ketones - requires quantitative deprotonation of one carbonyl component (LDA) - low temperature reaction typically gives aldol product ...
Organic Halides
... hemiacetal, and its acetal all exist in solution. Hemiacetal results from addition of the alcohol’s hydroxyl group to the carbon in the C=O bond. A cetals are products of substitution reactions catalyzed by acid. The presence of acid improves the leaving capacity of the hydroxyl group and enables it ...
... hemiacetal, and its acetal all exist in solution. Hemiacetal results from addition of the alcohol’s hydroxyl group to the carbon in the C=O bond. A cetals are products of substitution reactions catalyzed by acid. The presence of acid improves the leaving capacity of the hydroxyl group and enables it ...
Chapter 9-Additions to Alkenes I
... circumstances you will learn in CHE 232). 9.2. Addition of Strong Acids HX to Alkenes to give Alkyl ...
... circumstances you will learn in CHE 232). 9.2. Addition of Strong Acids HX to Alkenes to give Alkyl ...
lab 12 Multistep Synthesis of Benzilic acid
... dissolved and heated for a few minutes, the mixture will turn blue-‐black in color. As the reaction proceeds, the reaction product will turn brown and the solid may, or ...
... dissolved and heated for a few minutes, the mixture will turn blue-‐black in color. As the reaction proceeds, the reaction product will turn brown and the solid may, or ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.